About the Report
Base Year 2024APAC Live Cell Encapsulation Market Overview
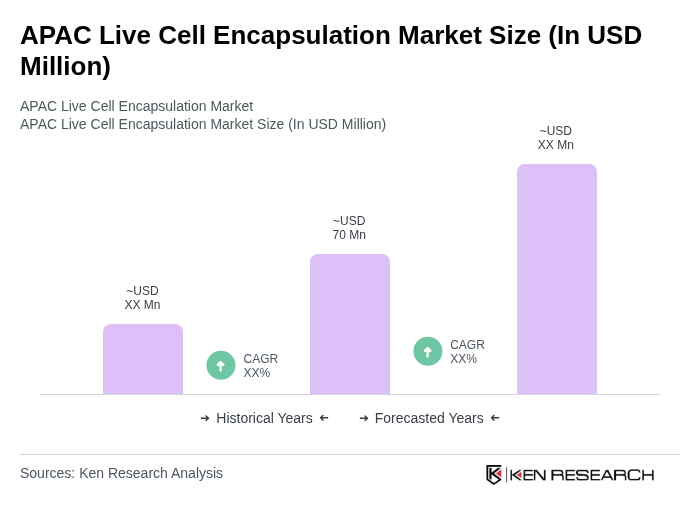
- The APAC Live Cell Encapsulation Market is valued at USD 70 million, based on a five-year historical analysis. This valuation is consistent with regional shares reported for Asia-Pacific within the global live cell encapsulation and broader cell encapsulation markets, where Asia-Pacific is identified as the fastest-growing region but still represents a minority share of a global market valued in the low hundreds of millions of USD. This growth is primarily driven by advancements in biotechnology, increasing demand for cell-based therapies, and rising investments in research and development, particularly in diabetes, oncology, and regenerative medicine applications. The market is also supported by the growing prevalence of chronic diseases such as diabetes and cardiovascular disorders across Asia-Pacific, which necessitate innovative treatment solutions and support adoption of encapsulated cell-based drug delivery and regenerative therapies.
- Key players in this market include China, Japan, and South Korea, which dominate due to their robust healthcare infrastructure, significant investments in biotechnology, and a strong focus on research and development. These countries have established themselves as leaders in the field of cell therapy and regenerative medicine, with strong pipelines of cell and gene therapy clinical trials, advanced biologics manufacturing capabilities, and active government support programs, thereby contributing to the overall growth of the live cell encapsulation market in APAC.
- In India, the regulatory environment for cell-based products is guided by instruments such as the “National Guidelines for Stem Cell Research, 2017” issued by the Indian Council of Medical Research (ICMR) and the Department of Biotechnology, and by the Central Drugs Standard Control Organization (CDSCO) guidance that classifies most stem cell-based products as “new drugs” under the New Drugs and Clinical Trials Rules, 2019, requiring marketing authorization and clinical trial approval. These regulations are designed to enhance patient safety and ensure the quality, efficacy, and traceability of cell-based therapies, including encapsulated live cell products where applicable, thereby fostering innovation and structured growth in the live cell encapsulation market.
APAC Live Cell Encapsulation Market Segmentation
By Type:
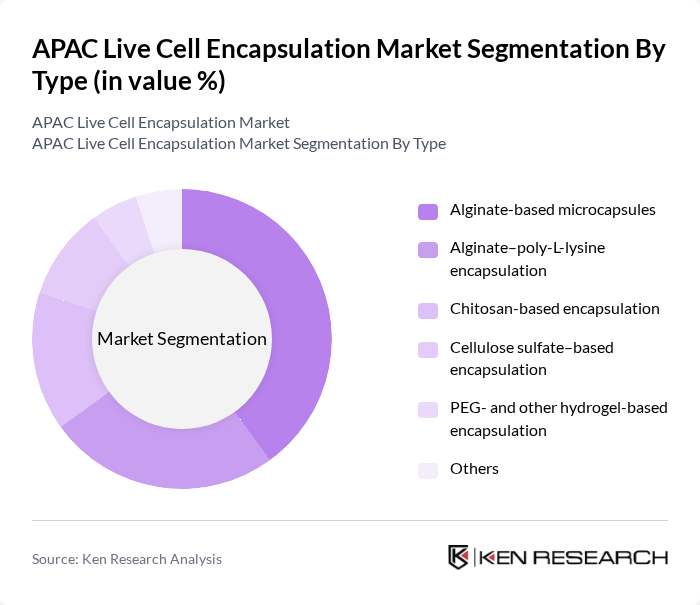
The subsegments under this category include Alginate-based microcapsules, Alginate–poly-L-lysine encapsulation, Chitosan-based encapsulation, Cellulose sulfate–based encapsulation, PEG- and other hydrogel-based encapsulation, and Others. Among these, Alginate-based microcapsules are leading the market due to their biocompatibility, mild gelation conditions, and established use in encapsulating islet cells and other therapeutic cells for controlled drug release and immune isolation. The growing demand for targeted therapies and the increasing focus on personalized medicine and advanced therapy medicinal products (ATMPs) are driving the adoption of this subsegment across APAC research institutes and biopharmaceutical developers. Additionally, advancements in encapsulation technologies—such as improved alginate purification, multilayer coatings, and microfluidic-based production—are enhancing the mechanical stability, cell viability, and functional performance of alginate-based microcapsules, making them a preferred choice for researchers and healthcare providers.
By End-User:
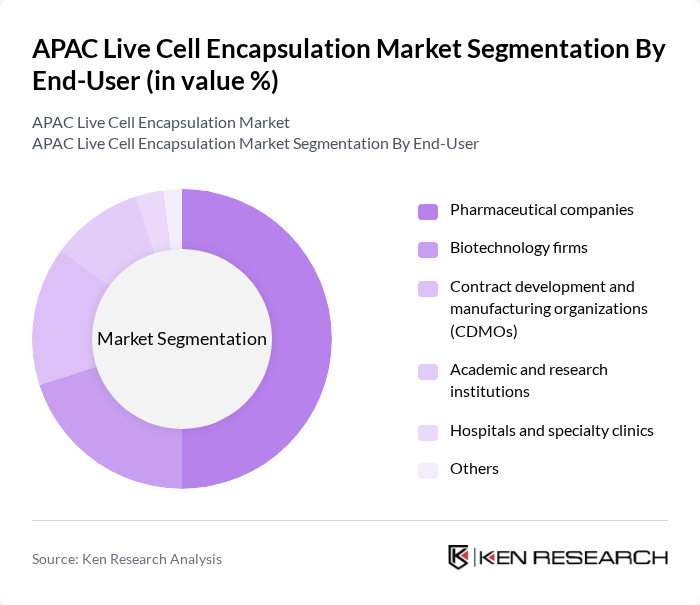
This category includes Pharmaceutical companies, Biotechnology firms, Contract development and manufacturing organizations (CDMOs), Academic and research institutions, Hospitals and specialty clinics, and Others. The Pharmaceutical companies segment is currently dominating the market due to their extensive research and development capabilities, significant financial resources, and established distribution networks, which enable them to integrate live cell encapsulation into pipelines for regenerative medicine, implantable cell therapies, and long-acting biologic delivery. These companies are increasingly investing in live cell encapsulation technologies to enhance drug delivery systems, extend therapeutic duration, and improve safety profiles by localizing cell-based therapies. The rising trend of collaboration between pharmaceutical companies, biotechnology firms, and CDMOs in APAC—covering process development, GMP manufacturing of encapsulated products, and clinical trial support—is also contributing to the growth of this segment.
APAC Live Cell Encapsulation Market Competitive Landscape
The APAC Live Cell Encapsulation Market is characterized by a dynamic mix of regional and international players. Leading participants such as Viacyte, Inc., Sigilon Therapeutics, Inc., Blacktrace Holdings Ltd (Dolomite Microfluidics), Sernova Corp., Evonik Industries AG, Merck KGaA (Merck Millipore / Sigma-Aldrich), Lonza Group Ltd, Becton, Dickinson and Company, 3M Company, Ballex Corporation, Kadimastem Ltd., PharmaCell B.V. (part of Lonza), Cellamo Co., Ltd. (South Korea), NIPRO Corporation, Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space.
APAC Live Cell Encapsulation Market Industry Analysis
Growth Drivers
- Increasing Demand for Cell-Based Therapies:The APAC region is witnessing a surge in demand for cell-based therapies, driven by the rising incidence of chronic diseases. According to the World Health Organization, chronic diseases account for approximately 74% of all global deaths, with Asia-Pacific countries reporting significant increases. The market for cell-based therapies is projected to reach $25 billion in future, highlighting the urgent need for effective treatment options, thereby propelling the live cell encapsulation market.
- Advancements in Encapsulation Technologies:Technological innovations in encapsulation methods are enhancing the efficacy and safety of cell-based therapies. For instance, the development of microencapsulation techniques has improved the viability of cells during storage and transportation. A report by Research and Markets indicates that the global microencapsulation market is expected to grow to $35 billion in future, indicating a strong correlation with advancements in encapsulation technologies that support live cell applications in APAC.
- Rising Prevalence of Chronic Diseases:The increasing prevalence of chronic diseases such as diabetes and cardiovascular disorders is a significant growth driver for the live cell encapsulation market. The International Diabetes Federation reported that approximately 463 million adults were living with diabetes, a number projected to rise to 700 million in future. This alarming trend necessitates innovative treatment solutions, including cell-based therapies, thereby boosting the demand for encapsulation technologies in the region.
Market Challenges
- High Production Costs:One of the primary challenges facing the live cell encapsulation market is the high production costs associated with advanced encapsulation technologies. The cost of raw materials and sophisticated manufacturing processes can exceed $1.2 million per batch, making it difficult for smaller firms to compete. This financial barrier limits market entry and innovation, hindering the overall growth of the industry in the APAC region.
- Regulatory Hurdles:Regulatory challenges pose significant obstacles to the live cell encapsulation market. The approval processes for new encapsulation technologies can be lengthy and complex, often taking several years. For example, the U.S. FDA's Biologics License Application process can take up to 14 months for review. These stringent regulations can delay product launches and increase costs, creating a challenging environment for companies operating in the APAC region.
APAC Live Cell Encapsulation Market Future Outlook
The future of the live cell encapsulation market in the APAC region appears promising, driven by ongoing advancements in technology and increasing investments in regenerative medicine. As healthcare providers and researchers focus on personalized medicine, the demand for innovative encapsulation solutions is expected to rise. Furthermore, emerging markets in APAC are likely to adopt these technologies, creating new opportunities for growth and collaboration among industry stakeholders, ultimately enhancing patient outcomes and treatment efficacy.
Market Opportunities
- Expansion of Research and Development Activities:Increased funding for research and development in regenerative medicine presents a significant opportunity for the live cell encapsulation market. In future, R&D spending in the healthcare sector in APAC is projected to reach $160 billion, fostering innovation and the development of new encapsulation technologies that can improve treatment outcomes.
- Collaborations with Biotech Firms:Strategic partnerships between pharmaceutical companies and biotech firms can enhance the development of live cell encapsulation technologies. Collaborations are expected to increase, with over 250 partnerships anticipated in future, facilitating knowledge sharing and resource pooling, which can accelerate the commercialization of innovative therapies in the APAC region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Alginate-based microcapsules Alginate–poly-L-lysine encapsulation Chitosan-based encapsulation Cellulose sulfate–based encapsulation PEG- and other hydrogel-based encapsulation Others |
| By End-User | Pharmaceutical companies Biotechnology firms Contract development and manufacturing organizations (CDMOs) Academic and research institutions Hospitals and specialty clinics Others |
| By Application | Drug delivery and controlled release Regenerative medicine and tissue engineering Cell transplantation (e.g., islet and stem cell therapy) Oncology and immunotherapy Others |
| By Material | Natural polymers (alginate, chitosan, cellulose derivatives, others) Synthetic polymers (PEG, PLA, PLGA, others) Inorganic and hybrid materials Others |
| By Technology | Microencapsulation Nanoencapsulation Macroencapsulation (devices, capsules) D bioprinting-enabled encapsulation Others |
| By Region | East Asia (China, Japan, South Korea) Southeast Asia (Singapore, Malaysia, Thailand, Indonesia, others) South Asia (India, Pakistan, Bangladesh, others) Oceania (Australia, New Zealand) Rest of APAC |
| By Regulatory Compliance | PMDA (Japan) regulations on cell-based products NMPA (China) guidelines for advanced therapies CDSCO and ICMR (India) frameworks for cell therapy MFDS (South Korea) and TGA (Australia) standards Other APAC regional standards |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Institutes of Health, Food and Drug Administration)
Biotechnology and Pharmaceutical Companies
Medical Device Manufacturers
Healthcare Providers and Hospitals
Research and Development Organizations
Industry Associations and Trade Groups
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Viacyte, Inc.
Sigilon Therapeutics, Inc.
Blacktrace Holdings Ltd (Dolomite Microfluidics)
Sernova Corp.
Evonik Industries AG
Merck KGaA (Merck Millipore / Sigma-Aldrich)
Lonza Group Ltd
Becton, Dickinson and Company
3M Company
Ballex Corporation
Kadimastem Ltd.
PharmaCell B.V. (part of Lonza)
Cellamo Co., Ltd. (South Korea)
NIPRO Corporation
Terumo Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Live Cell Encapsulation Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Live Cell Encapsulation Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Live Cell Encapsulation Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for cell-based therapies
3.1.2 Advancements in encapsulation technologies
3.1.3 Rising prevalence of chronic diseases
3.1.4 Growing investment in regenerative medicine
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Regulatory hurdles
3.2.3 Limited awareness among healthcare providers
3.2.4 Technical complexities in encapsulation processes
3.3 Market Opportunities
3.3.1 Expansion of research and development activities
3.3.2 Collaborations with biotech firms
3.3.3 Increasing focus on personalized medicine
3.3.4 Emerging markets in APAC
3.4 Market Trends
3.4.1 Shift towards biocompatible materials
3.4.2 Integration of AI in encapsulation processes
3.4.3 Growth of microencapsulation techniques
3.4.4 Rising demand for targeted drug delivery systems
3.5 Government Regulation
3.5.1 Guidelines for cell therapy products
3.5.2 Approval processes for new encapsulation technologies
3.5.3 Compliance requirements for manufacturing
3.5.4 Funding programs for research in regenerative medicine
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Live Cell Encapsulation Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Live Cell Encapsulation Market Segmentation
8.1 By Type
8.1.1 Alginate-based microcapsules
8.1.2 Alginate–poly-L-lysine encapsulation
8.1.3 Chitosan-based encapsulation
8.1.4 Cellulose sulfate–based encapsulation
8.1.5 PEG- and other hydrogel-based encapsulation
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical companies
8.2.2 Biotechnology firms
8.2.3 Contract development and manufacturing organizations (CDMOs)
8.2.4 Academic and research institutions
8.2.5 Hospitals and specialty clinics
8.2.6 Others
8.3 By Application
8.3.1 Drug delivery and controlled release
8.3.2 Regenerative medicine and tissue engineering
8.3.3 Cell transplantation (e.g., islet and stem cell therapy)
8.3.4 Oncology and immunotherapy
8.3.5 Others
8.4 By Material
8.4.1 Natural polymers (alginate, chitosan, cellulose derivatives, others)
8.4.2 Synthetic polymers (PEG, PLA, PLGA, others)
8.4.3 Inorganic and hybrid materials
8.4.4 Others
8.5 By Technology
8.5.1 Microencapsulation
8.5.2 Nanoencapsulation
8.5.3 Macroencapsulation (devices, capsules)
8.5.4 3D bioprinting-enabled encapsulation
8.5.5 Others
8.6 By Region
8.6.1 East Asia (China, Japan, South Korea)
8.6.2 Southeast Asia (Singapore, Malaysia, Thailand, Indonesia, others)
8.6.3 South Asia (India, Pakistan, Bangladesh, others)
8.6.4 Oceania (Australia, New Zealand)
8.6.5 Rest of APAC
8.7 By Regulatory Compliance
8.7.1 PMDA (Japan) regulations on cell-based products
8.7.2 NMPA (China) guidelines for advanced therapies
8.7.3 CDSCO and ICMR (India) frameworks for cell therapy
8.7.4 MFDS (South Korea) and TGA (Australia) standards
8.7.5 Other APAC regional standards
9. APAC Live Cell Encapsulation Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company name and headquarters (APAC/global)
9.2.2 Group size (Large, Medium, or Small as per industry convention)
9.2.3 APAC live cell encapsulation revenue and revenue growth rate
9.2.4 R&D intensity in cell encapsulation (% of revenue)
9.2.5 Number of active encapsulation-related clinical trials in APAC
9.2.6 Pipeline breadth (number of encapsulated cell therapy programs)
9.2.7 Installed manufacturing capacity for encapsulated cell products
9.2.8 Number of strategic collaborations and licensing deals in APAC
9.2.9 Geographic footprint in APAC (countries served, facilities)
9.2.10 Time-to-market and regulatory approval track record in APAC
9.2.11 Intellectual property strength (patent families in encapsulation)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Viacyte, Inc.
9.5.2 Sigilon Therapeutics, Inc.
9.5.3 Blacktrace Holdings Ltd (Dolomite Microfluidics)
9.5.4 Sernova Corp.
9.5.5 Evonik Industries AG
9.5.6 Merck KGaA (Merck Millipore / Sigma-Aldrich)
9.5.7 Lonza Group Ltd
9.5.8 Becton, Dickinson and Company
9.5.9 3M Company
9.5.10 Ballex Corporation
9.5.11 Kadimastem Ltd.
9.5.12 PharmaCell B.V. (part of Lonza)
9.5.13 Cellamo Co., Ltd. (South Korea)
9.5.14 NIPRO Corporation
9.5.15 Terumo Corporation
10. APAC Live Cell Encapsulation Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for healthcare
10.1.2 Decision-making processes
10.1.3 Evaluation criteria for suppliers
10.1.4 Contract management practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment trends in healthcare infrastructure
10.2.2 Spending on innovative technologies
10.2.3 Budgeting for R&D
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges faced by hospitals
10.3.2 Issues in supply chain management
10.3.3 Regulatory compliance difficulties
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of encapsulation benefits
10.4.2 Training and education needs
10.4.3 Infrastructure readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of success metrics
10.5.2 Case studies of successful implementations
10.5.3 Future expansion opportunities
10.5.4 Others
11. APAC Live Cell Encapsulation Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Risk assessment
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience definition
2.4 Communication strategies
2.5 Digital marketing tactics
2.6 Event marketing opportunities
2.7 Feedback mechanisms
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 E-commerce channels
3.4 Direct sales approaches
3.5 Distribution partnerships
3.6 Logistics and supply chain management
3.7 Performance metrics
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
4.5 Value-based pricing models
4.6 Discounting strategies
4.7 Price elasticity considerations
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends exploration
5.4 Product development opportunities
5.5 Market entry barriers
5.6 Customer feedback analysis
5.7 Future demand forecasting
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Retention strategies
6.7 Customer satisfaction measurement
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-saving measures
7.4 Quality assurance practices
7.5 Innovation in product offerings
7.6 Customer-centric approaches
7.7 Competitive advantages
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Market entry timelines
10.7 Success factors
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Financial projections
11.4 Funding sources
11.5 Budget allocation
11.6 Cost management strategies
11.7 Investment return expectations
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnership dynamics
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Long-term sustainability
12.6 Exit strategies
12.7 Performance evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin expectations
13.4 Revenue growth projections
13.5 Cost structure analysis
13.6 Financial health indicators
13.7 Market share growth
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaboration opportunities
14
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from biotechnology and pharmaceutical associations in the APAC region
- Market analysis publications focusing on live cell encapsulation technologies
- Regulatory frameworks and guidelines from health authorities across APAC countries
Primary Research
- Interviews with R&D heads at leading biotechnology firms specializing in cell therapies
- Surveys with healthcare professionals involved in regenerative medicine
- Field interviews with production managers at facilities utilizing live cell encapsulation
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert opinions
- Triangulation of market trends with sales data and clinical trial outcomes
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of the overall biotechnology market size in the APAC region
- Segmentation by application areas such as drug delivery and tissue engineering
- Incorporation of growth rates from related sectors like pharmaceuticals and healthcare
Bottom-up Modeling
- Volume estimates based on production capacities of key manufacturers in the region
- Cost analysis derived from pricing models of encapsulation technologies
- Estimation of market share based on sales data from leading firms
Forecasting & Scenario Analysis
- Multi-variable regression analysis considering factors like technological advancements and market demand
- Scenario planning based on potential regulatory changes and healthcare trends
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biotechnology Firms | 90 | R&D Directors, Product Managers |
| Healthcare Providers | 80 | Clinical Researchers, Medical Directors |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Academic Institutions | 70 | Professors, Research Scientists |
| Manufacturing Facilities | 60 | Production Managers, Quality Control Analysts |
Frequently Asked Questions
What is the current value of the APAC Live Cell Encapsulation Market?
The APAC Live Cell Encapsulation Market is valued at approximately USD 70 million, reflecting its position as the fastest-growing region in the global live cell encapsulation market, which is valued in the low hundreds of millions of USD.