About the Report
Base Year 2024APAC Single Use Bioprocessing Market Overview
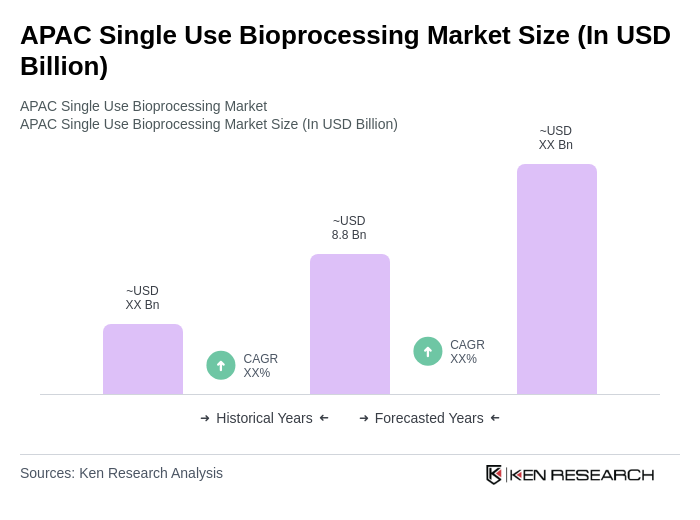
- The APAC Single Use Bioprocessing Market is valued at USD 8.8 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for biopharmaceuticals, advancements in bioprocessing technologies, and the rising adoption of single-use systems for their cost-effectiveness and reduced risk of contamination. The market is also supported by the growing trend of personalized medicine, government-backed capacity expansion initiatives, and the need for rapid facility deployment in response to global health challenges.
- Key players in this market include China, Japan, and India, which dominate due to their robust pharmaceutical industries, significant investments in bioprocessing technologies, and a large pool of skilled labor. China is the fastest-growing country in the region and has seen rapid growth in biopharmaceutical manufacturing capacity and CDMO capabilities, while Japan is known for its advanced research and development capabilities. India's cost-effective production, increasing focus on biopharmaceutical exports, and expanding manufacturing infrastructure further enhance its market position.
- The Indian regulatory framework is guided by the Drugs and Cosmetics Act, 1940 and its associated rules, administered by the Central Drugs Standard Control Organization (CDSCO). This comprehensive legislation governs the manufacturing, distribution, and quality standards of pharmaceutical and biopharmaceutical products in India. The framework includes specific provisions for licensing requirements, good manufacturing practices compliance, and quality control standards for bioprocessing facilities. It establishes clear thresholds for facility inspections, mandates adherence to international quality standards, and requires manufacturers to obtain appropriate licenses before commencing commercial production. The CDSCO oversees the implementation of these regulations and ensures that biopharmaceutical manufacturing facilities meet stringent safety and efficacy requirements, thereby enhancing the competitiveness of the Indian biopharmaceutical sector in the global market.
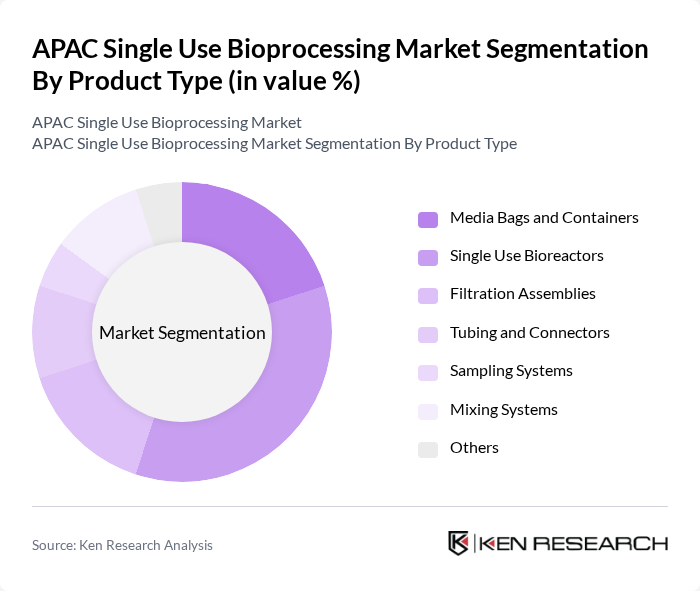
APAC Single Use Bioprocessing Market Segmentation
By Product Type:The product type segmentation includes various components essential for single-use bioprocessing. The subsegments are Media Bags and Containers, Single Use Bioreactors, Filtration Assemblies, Tubing and Connectors, Sampling Systems, Mixing Systems, and Others. Among these, Simple and Peripheral Elements (which include media bags, containers, tubing, and connectors) represent the largest and fastest-growing segment due to their widespread application across all bioprocessing workflows and the continuous demand for disposable components that minimize cross-contamination risks. Single Use Bioreactors continue to gain significant traction due to their efficiency in bioprocessing and the growing trend towards disposable systems, with commercial systems now reaching capacities above 5,000 liters while maintaining mixing and sterility performance.
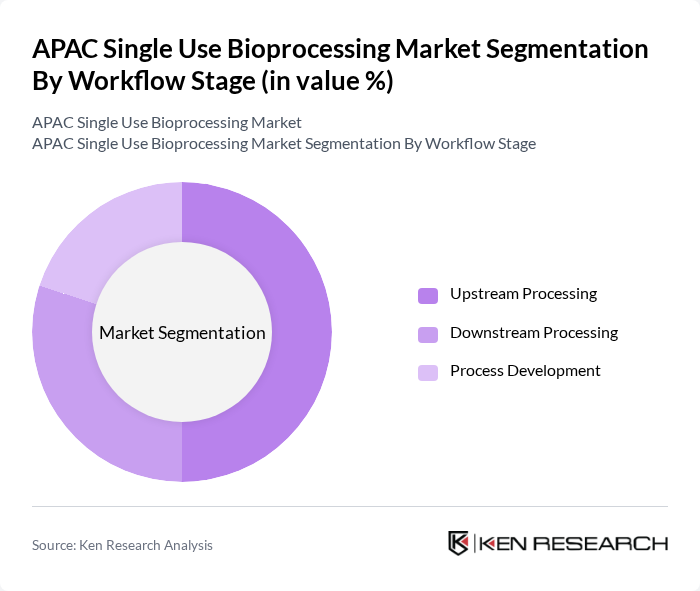
By Workflow Stage:The workflow stage segmentation encompasses Upstream Processing, Downstream Processing, and Process Development. Upstream Processing is currently the dominant segment, driven by the increasing need for efficient cell culture and fermentation processes in biopharmaceutical production. The trend towards automation and the integration of single-use technologies in upstream processes further enhance its market leadership. Downstream Processing is experiencing accelerated growth as purification technology advances and manufacturers seek to match the flexibility gains achieved in upstream operations.
APAC Single Use Bioprocessing Market Competitive Landscape
The APAC Single Use Bioprocessing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sartorius AG, Danaher Corporation (Cytiva), Thermo Fisher Scientific Inc., Merck KGaA, Avantor, Inc., Repligen Corporation, Entegris, Inc., Getinge AB, Parker Hannifin Corporation, Alfa Laval Corporate AB, Saint-Gobain, Eppendorf SE, Corning Incorporated, Lonza Group AG, ABEC, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
APAC Single Use Bioprocessing Market Industry Analysis
Growth Drivers
- Increasing Demand for Biopharmaceuticals:The biopharmaceutical sector in the APAC region is projected to reach $300 billion in future, driven by rising healthcare needs and an aging population. The World Health Organization reports that the prevalence of chronic diseases is increasing, necessitating innovative therapies. This surge in demand for biopharmaceuticals directly correlates with the adoption of single-use bioprocessing systems, which offer flexibility and efficiency in production, thus supporting market growth.
- Cost-Effectiveness of Single Use Systems:Single-use bioprocessing systems can reduce operational costs by up to 30% compared to traditional systems, according to industry reports. This cost efficiency is particularly crucial in the APAC region, where many biopharmaceutical companies are looking to optimize their production processes. The reduction in cleaning and validation times further enhances the appeal of single-use systems, making them a preferred choice for manufacturers aiming to improve their bottom line.
- Technological Advancements in Bioprocessing:The APAC region is witnessing significant technological innovations in bioprocessing, with investments exceeding $1 billion in future. These advancements include the development of more efficient single-use components and automation technologies that streamline production. As companies adopt these technologies, they enhance productivity and reduce time-to-market for biopharmaceutical products, thereby driving the growth of the single-use bioprocessing market.
Market Challenges
- Regulatory Compliance Issues:The biopharmaceutical industry in APAC faces stringent regulatory requirements, with compliance costs averaging around $2 million per product. These regulations can delay product launches and increase operational complexities for companies utilizing single-use systems. Navigating these regulatory landscapes is crucial for market players, as non-compliance can lead to significant financial penalties and reputational damage.
- High Initial Investment Costs:The initial setup costs for single-use bioprocessing systems can range from $500,000 to $1 million, posing a barrier for smaller companies. This financial hurdle can limit market entry and expansion opportunities, particularly in developing economies within the APAC region. Companies must weigh these costs against the long-term benefits of operational efficiency and flexibility offered by single-use technologies.
APAC Single Use Bioprocessing Market Future Outlook
The future of the APAC single-use bioprocessing market appears promising, driven by ongoing innovations and increasing demand for biopharmaceuticals. As companies continue to invest in advanced technologies, the efficiency and scalability of bioprocessing will improve. Additionally, the growing trend towards personalized medicine will further propel the adoption of single-use systems, as they offer the flexibility needed for small-batch production. Overall, the market is poised for significant growth, supported by favorable economic conditions and technological advancements.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets in Southeast Asia are expected to see a 15% increase in biopharmaceutical investments in future. This growth presents opportunities for single-use bioprocessing companies to establish a foothold in these regions, catering to the rising demand for affordable healthcare solutions and innovative therapies.
- Development of Advanced Materials:The development of new materials for single-use systems is projected to grow by 20% annually in future. Innovations in biocompatible materials can enhance product safety and performance, creating opportunities for manufacturers to differentiate their offerings and meet the evolving needs of the biopharmaceutical industry.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Media Bags and Containers Single Use Bioreactors Filtration Assemblies Tubing and Connectors Sampling Systems Mixing Systems Others |
| By Workflow Stage | Upstream Processing Downstream Processing Process Development |
| By Application | Monoclonal Antibody Production Vaccine Manufacturing Cell and Gene Therapy Biosimilars Production Others |
| By End-User | Biopharmaceutical Companies Contract Manufacturing Organizations (CMOs) Research and Academic Institutes Others |
| By Country | China India Japan South Korea Australia Singapore Thailand Malaysia Others |
| By Scale of Operation | Small-Scale (?500L) Mid-Scale (500L-2000L) Large-Scale (>2000L) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Institute of Health, Food and Drug Administration, Ministry of Health)
Biopharmaceutical Manufacturers
Contract Manufacturing Organizations (CMOs)
Biotechnology Companies
Equipment Suppliers and Manufacturers
Pharmaceutical Research Organizations
Healthcare Providers and Hospitals
Players Mentioned in the Report:
Sartorius AG
Danaher Corporation (Cytiva)
Thermo Fisher Scientific Inc.
Merck KGaA
Avantor, Inc.
Repligen Corporation
Entegris, Inc.
Getinge AB
Parker Hannifin Corporation
Alfa Laval Corporate AB
Saint-Gobain
Eppendorf SE
Corning Incorporated
Lonza Group AG
ABEC, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Single Use Bioprocessing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Single Use Bioprocessing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Single Use Bioprocessing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Biopharmaceuticals
3.1.2 Cost-Effectiveness of Single Use Systems
3.1.3 Technological Advancements in Bioprocessing
3.1.4 Rising Focus on Biomanufacturing Efficiency
3.2 Market Challenges
3.2.1 Regulatory Compliance Issues
3.2.2 High Initial Investment Costs
3.2.3 Limited Availability of Skilled Workforce
3.2.4 Concerns Over Product Quality and Safety
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Development of Advanced Materials
3.3.3 Strategic Partnerships and Collaborations
3.3.4 Increasing Adoption of Personalized Medicine
3.4 Market Trends
3.4.1 Shift Towards Modular Bioprocessing Solutions
3.4.2 Growing Interest in Sustainable Practices
3.4.3 Integration of Digital Technologies
3.4.4 Rise of Contract Manufacturing Organizations (CMOs)
3.5 Government Regulation
3.5.1 Stringent Quality Control Standards
3.5.2 Guidelines for Single Use Systems
3.5.3 Environmental Regulations on Waste Disposal
3.5.4 Incentives for Biopharmaceutical Innovation
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Single Use Bioprocessing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Single Use Bioprocessing Market Segmentation
8.1 By Product Type
8.1.1 Media Bags and Containers
8.1.2 Single Use Bioreactors
8.1.3 Filtration Assemblies
8.1.4 Tubing and Connectors
8.1.5 Sampling Systems
8.1.6 Mixing Systems
8.1.7 Others
8.2 By Workflow Stage
8.2.1 Upstream Processing
8.2.2 Downstream Processing
8.2.3 Process Development
8.3 By Application
8.3.1 Monoclonal Antibody Production
8.3.2 Vaccine Manufacturing
8.3.3 Cell and Gene Therapy
8.3.4 Biosimilars Production
8.3.5 Others
8.4 By End-User
8.4.1 Biopharmaceutical Companies
8.4.2 Contract Manufacturing Organizations (CMOs)
8.4.3 Research and Academic Institutes
8.4.4 Others
8.5 By Country
8.5.1 China
8.5.2 India
8.5.3 Japan
8.5.4 South Korea
8.5.5 Australia
8.5.6 Singapore
8.5.7 Thailand
8.5.8 Malaysia
8.5.9 Others
8.6 By Scale of Operation
8.6.1 Small-Scale (?500L)
8.6.2 Mid-Scale (500L-2000L)
8.6.3 Large-Scale (>2000L)
9. APAC Single Use Bioprocessing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, or Small)
9.2.3 APAC Revenue Growth Rate (%)
9.2.4 Product Portfolio Breadth
9.2.5 Manufacturing Capacity in APAC
9.2.6 R&D Investment Intensity (%)
9.2.7 Local Partnership Strength
9.2.8 Time-to-Market for New Products (Months)
9.2.9 Regulatory Compliance Score
9.2.10 Customer Adoption Rate in Biologics Manufacturing
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sartorius AG
9.5.2 Danaher Corporation (Cytiva)
9.5.3 Thermo Fisher Scientific Inc.
9.5.4 Merck KGaA
9.5.5 Avantor, Inc.
9.5.6 Repligen Corporation
9.5.7 Entegris, Inc.
9.5.8 Getinge AB
9.5.9 Parker Hannifin Corporation
9.5.10 Alfa Laval Corporate AB
9.5.11 Saint-Gobain
9.5.12 Eppendorf SE
9.5.13 Corning Incorporated
9.5.14 Lonza Group AG
9.5.15 ABEC, Inc.
10. APAC Single Use Bioprocessing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Funding for Bioprocessing
10.1.2 Procurement Policies for Biopharmaceuticals
10.1.3 Collaboration with Private Sector
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Bioprocessing Facilities
10.2.2 Budget Allocation for R&D
10.2.3 Expenditure on Sustainable Practices
10.3 Pain Point Analysis by End-User Category
10.3.1 Supply Chain Disruptions
10.3.2 Quality Assurance Challenges
10.3.3 Cost Management Issues
10.4 User Readiness for Adoption
10.4.1 Training and Development Needs
10.4.2 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI in Bioprocessing
10.5.2 Expansion into New Applications
11. APAC Single Use Bioprocessing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from bioprocessing associations and market research firms
- Published articles and white papers on single-use bioprocessing technologies
- Regulatory guidelines and updates from health authorities across APAC countries
Primary Research
- Interviews with R&D heads at biopharmaceutical companies utilizing single-use systems
- Surveys with procurement managers in the bioprocessing supply chain
- Field interviews with engineers and technicians involved in bioprocessing operations
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market size estimates using sales data and production capacity
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of overall bioprocessing market size and growth trends in APAC
- Segmentation by application areas such as vaccines, monoclonal antibodies, and cell therapies
- Incorporation of government funding and initiatives supporting bioprocessing innovation
Bottom-up Modeling
- Volume estimates based on production capacities of leading biopharmaceutical firms
- Cost analysis of single-use systems versus traditional bioprocessing methods
- Estimation of market share based on sales data from key suppliers of single-use technologies
Forecasting & Scenario Analysis
- Multi-variable regression analysis considering factors like healthcare expenditure and biopharma growth
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical Manufacturing | 100 | Production Managers, Quality Assurance Leads |
| Research & Development in Bioprocessing | 60 | R&D Directors, Process Development Scientists |
| Supply Chain Management for Single-Use Systems | 50 | Supply Chain Managers, Procurement Specialists |
| Regulatory Affairs in Bioprocessing | 40 | Regulatory Affairs Managers, Compliance Officers |
| End-User Feedback on Single-Use Technologies | 50 | Bioprocess Engineers, Facility Managers |
Frequently Asked Questions
What is the current value of the APAC Single Use Bioprocessing Market?
The APAC Single Use Bioprocessing Market is valued at approximately USD 8.8 billion, driven by the increasing demand for biopharmaceuticals and advancements in bioprocessing technologies. This market is expected to grow significantly due to the rising adoption of single-use systems.