About the Report
Base Year 2024Bahrain Oncology Companion Diagnostics Market Overview
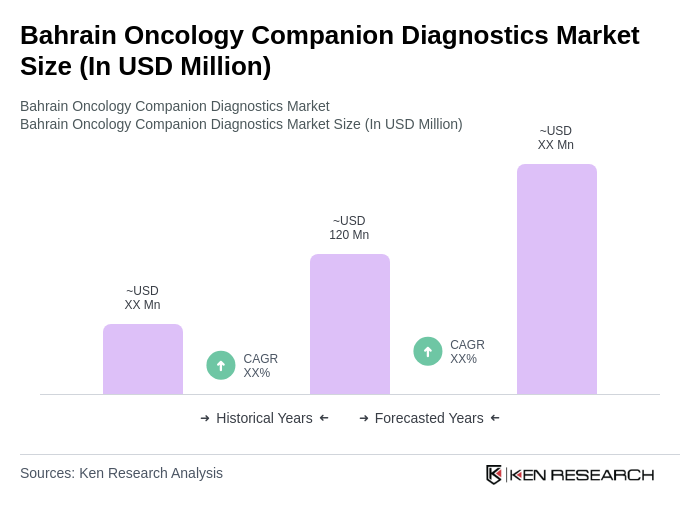
- The Bahrain Oncology Companion Diagnostics Market is valued at USD 120 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cancer, with over 1,380 new cancer cases and more than 4,600 five-year prevalent cases reported in the most recent period. Additional growth factors include advancements in personalized medicine, rising demand for targeted therapies, and the integration of companion diagnostics with therapeutic drugs to optimize treatment outcomes. The adoption of innovative diagnostic technologies and the expansion of oncology services in leading healthcare facilities further propel market expansion.
- Key players in this market are concentrated in Manama, Riffa, and Muharraq, which dominate due to their advanced healthcare infrastructure, availability of specialized oncology services, and collaboration with international pharmaceutical companies. These cities serve as hubs for medical research and innovation, attracting investments and fostering a competitive environment for companion diagnostics.
- In 2023, the Bahrain government implemented the “Medical Devices Regulation, 2023” issued by the National Health Regulatory Authority (NHRA), which mandates that all companion diagnostics must receive NHRA approval prior to marketing. This regulation establishes comprehensive requirements for clinical evidence, safety, and performance, ensuring that diagnostic tests meet international standards and enhancing patient care across the country.
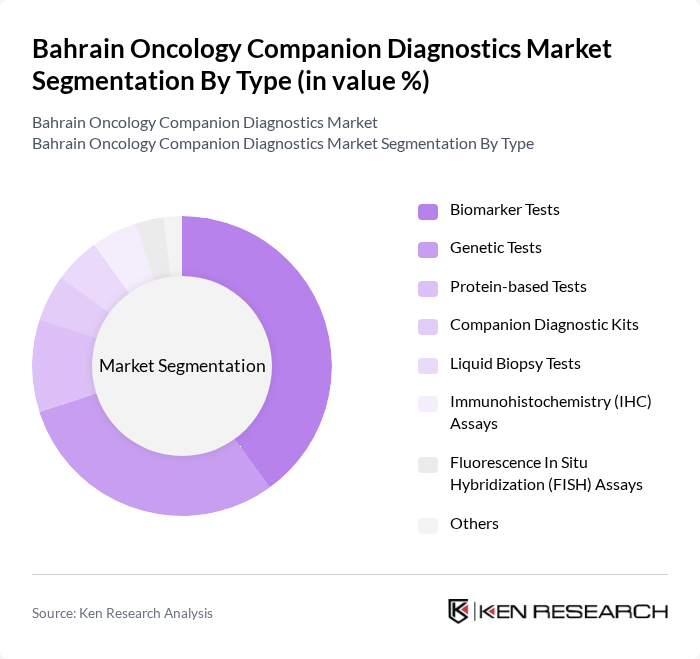
Bahrain Oncology Companion Diagnostics Market Segmentation
By Type:The market is segmented into various types of diagnostics, including biomarker tests, genetic tests, protein-based tests, companion diagnostic kits, liquid biopsy tests, immunohistochemistry (IHC) assays, fluorescence in situ hybridization (FISH) assays, and others. Among these, biomarker tests and genetic tests are particularly prominent due to their critical role in identifying specific cancer types and guiding treatment decisions. The increasing focus on personalized medicine and the adoption of molecular profiling in oncology have led to a surge in demand for these tests, making them the leading subsegments in the market.
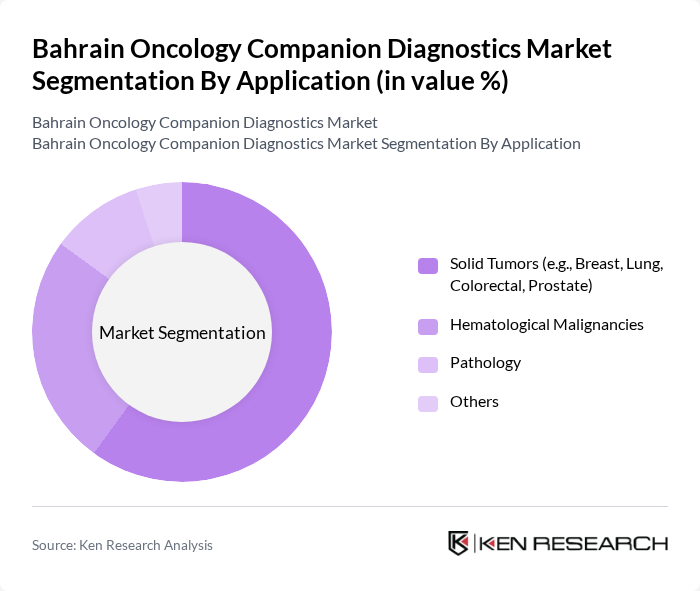
By Application:The applications of companion diagnostics in Bahrain include solid tumors, hematological malignancies, pathology, and others. Solid tumors, such as breast, lung, colorectal, and prostate cancers, dominate the market due to their high incidence rates and the need for targeted therapies. The increasing awareness of early detection, national cancer screening programs, and personalized treatment options has further fueled the demand for companion diagnostics in this application area.
Bahrain Oncology Companion Diagnostics Market Competitive Landscape
The Bahrain Oncology Companion Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Agilent Technologies, Thermo Fisher Scientific, Illumina, Inc., QIAGEN N.V., Abbott Laboratories, Myriad Genetics, Inc., Bio-Rad Laboratories, Inc., Hologic, Inc., Siemens Healthineers, Genomic Health, Inc., Foundation Medicine, Inc., Guardant Health, Inc., Novartis AG, Merck KGaA, BioMérieux S.A., Sysmex Corporation, Becton, Dickinson and Company (BD), Danaher Corporation (Leica Biosystems), Thermo Fisher Scientific Middle East FZE contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain Oncology Companion Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer:Bahrain has witnessed a significant rise in cancer cases, with the Ministry of Health reporting over 3,500 new cancer diagnoses in future. This increase is attributed to lifestyle changes and an aging population, with cancer becoming the second leading cause of death in the country. The growing patient population necessitates advanced diagnostic tools, driving demand for companion diagnostics that can tailor treatment options effectively.
- Advancements in Genomic Testing:The genomic testing market in Bahrain is projected to reach $20 million in future, reflecting a growing interest in precision medicine. Innovations in next-generation sequencing (NGS) technologies have made genomic testing more accessible and affordable. This trend is crucial for companion diagnostics, as it enables healthcare providers to identify specific biomarkers, leading to more effective and personalized treatment plans for cancer patients.
- Rising Demand for Personalized Medicine:The shift towards personalized medicine is evident in Bahrain, where healthcare expenditure on personalized therapies is expected to exceed $70 million in future. Patients and healthcare providers increasingly prefer treatments tailored to individual genetic profiles. This demand is propelling the growth of companion diagnostics, which play a vital role in determining the most effective therapies based on a patient’s unique genetic makeup, thereby improving treatment outcomes.
Market Challenges
- High Cost of Companion Diagnostics:The average cost of companion diagnostics in Bahrain can reach up to $6,000 per test, which poses a significant barrier for widespread adoption. Many healthcare providers and patients struggle to afford these tests, limiting access to essential diagnostic tools. This high cost is compounded by the limited insurance coverage for such diagnostics, further hindering their integration into standard cancer care practices.
- Limited Awareness Among Healthcare Providers:Despite advancements in oncology, many healthcare providers in Bahrain lack adequate knowledge about companion diagnostics. A recent survey indicated that over 65% of oncologists are unfamiliar with the latest diagnostic technologies. This knowledge gap can lead to underutilization of companion diagnostics, resulting in suboptimal treatment decisions and poorer patient outcomes, ultimately stalling market growth.
Bahrain Oncology Companion Diagnostics Market Future Outlook
The future of the Bahrain oncology companion diagnostics market appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the government enhances healthcare infrastructure, the integration of innovative diagnostic tools is expected to improve patient outcomes significantly. Additionally, collaborations between diagnostic companies and pharmaceutical firms are likely to foster the development of new therapies, further propelling market growth and enhancing the overall quality of cancer care in Bahrain.
Market Opportunities
- Expansion of Healthcare Infrastructure:Bahrain's government is investing approximately $1.5 billion in healthcare infrastructure in future. This investment will enhance access to advanced diagnostic services, creating opportunities for companion diagnostics to become integral to cancer treatment protocols, ultimately improving patient care and outcomes.
- Growth in Clinical Trials:The number of clinical trials in Bahrain is projected to increase by 35% in future, driven by partnerships with international research organizations. This growth presents opportunities for companion diagnostics to be utilized in trial settings, facilitating the development of targeted therapies and expanding their application in clinical practice.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Biomarker Tests Genetic Tests Protein-based Tests Companion Diagnostic Kits Liquid Biopsy Tests Immunohistochemistry (IHC) Assays Fluorescence In Situ Hybridization (FISH) Assays Others |
| By Application | Solid Tumors (e.g., Breast, Lung, Colorectal, Prostate) Hematological Malignancies Pathology Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Oncology Clinics Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Technology | Next-Generation Sequencing (NGS) PCR-based Testing Microarray Technology Immunohistochemistry (IHC) Fluorescence In Situ Hybridization (FISH) Digital Pathology Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-based Pricing Others |
| By Regulatory Approval Status | FDA Approved CE Marked Under Review NHRA Bahrain Approved Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Bahrain National Health Regulatory Authority)
Pharmaceutical Companies
Biotechnology Firms
Diagnostic Test Manufacturers
Healthcare Providers and Hospitals
Health Insurance Companies
Clinical Research Organizations
Players Mentioned in the Report:
Roche Diagnostics
Agilent Technologies
Thermo Fisher Scientific
Illumina, Inc.
QIAGEN N.V.
Abbott Laboratories
Myriad Genetics, Inc.
Bio-Rad Laboratories, Inc.
Hologic, Inc.
Siemens Healthineers
Genomic Health, Inc.
Foundation Medicine, Inc.
Guardant Health, Inc.
Novartis AG
Merck KGaA
BioMerieux S.A.
Sysmex Corporation
Becton, Dickinson and Company (BD)
Danaher Corporation (Leica Biosystems)
Thermo Fisher Scientific Middle East FZE
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Oncology Companion Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Oncology Companion Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Oncology Companion Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cancer
3.1.2 Advancements in genomic testing
3.1.3 Rising demand for personalized medicine
3.1.4 Government initiatives for cancer care
3.2 Market Challenges
3.2.1 High cost of companion diagnostics
3.2.2 Limited awareness among healthcare providers
3.2.3 Regulatory hurdles in market entry
3.2.4 Fragmented healthcare system
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with pharmaceutical companies
3.3.3 Growth in clinical trials
3.3.4 Increasing investment in R&D
3.4 Market Trends
3.4.1 Shift towards targeted therapies
3.4.2 Integration of AI in diagnostics
3.4.3 Rise of telemedicine in oncology
3.4.4 Focus on patient-centric care
3.5 Government Regulation
3.5.1 Approval processes for diagnostics
3.5.2 Reimbursement policies for companion diagnostics
3.5.3 Guidelines for clinical use
3.5.4 Data protection regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Oncology Companion Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Oncology Companion Diagnostics Market Segmentation
8.1 By Type
8.1.1 Biomarker Tests
8.1.2 Genetic Tests
8.1.3 Protein-based Tests
8.1.4 Companion Diagnostic Kits
8.1.5 Liquid Biopsy Tests
8.1.6 Immunohistochemistry (IHC) Assays
8.1.7 Fluorescence In Situ Hybridization (FISH) Assays
8.1.8 Others
8.2 By Application
8.2.1 Solid Tumors (e.g., Breast, Lung, Colorectal, Prostate)
8.2.2 Hematological Malignancies
8.2.3 Pathology
8.2.4 Others
8.3 By End-User
8.3.1 Hospitals
8.3.2 Diagnostic Laboratories
8.3.3 Research Institutions
8.3.4 Oncology Clinics
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Others
8.5 By Technology
8.5.1 Next-Generation Sequencing (NGS)
8.5.2 PCR-based Testing
8.5.3 Microarray Technology
8.5.4 Immunohistochemistry (IHC)
8.5.5 Fluorescence In Situ Hybridization (FISH)
8.5.6 Digital Pathology
8.5.7 Others
8.6 By Pricing Model
8.6.1 Premium Pricing
8.6.2 Competitive Pricing
8.6.3 Value-based Pricing
8.6.4 Others
8.7 By Regulatory Approval Status
8.7.1 FDA Approved
8.7.2 CE Marked
8.7.3 Under Review
8.7.4 NHRA Bahrain Approved
8.7.5 Others
9. Bahrain Oncology Companion Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Bahrain Oncology CDx Segment)
9.2.4 Market Penetration Rate (Share of Bahrain Oncology CDx Market)
9.2.5 Number of Oncology CDx Products Approved in Bahrain
9.2.6 R&D Investment in Oncology CDx (USD, % of Revenue)
9.2.7 Local Partnerships/Distribution Agreements
9.2.8 Product Development Cycle Time (Oncology CDx)
9.2.9 Sales Conversion Rate (Bahrain Oncology CDx)
9.2.10 Brand Awareness Index (Bahrain Healthcare Sector)
9.2.11 Distribution Efficiency (Time to Market, Coverage)
9.2.12 Regulatory Approval Success Rate (Bahrain/NHRA)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Agilent Technologies
9.5.3 Thermo Fisher Scientific
9.5.4 Illumina, Inc.
9.5.5 QIAGEN N.V.
9.5.6 Abbott Laboratories
9.5.7 Myriad Genetics, Inc.
9.5.8 Bio-Rad Laboratories, Inc.
9.5.9 Hologic, Inc.
9.5.10 Siemens Healthineers
9.5.11 Genomic Health, Inc.
9.5.12 Foundation Medicine, Inc.
9.5.13 Guardant Health, Inc.
9.5.14 Novartis AG
9.5.15 Merck KGaA
9.5.16 BioMérieux S.A.
9.5.17 Sysmex Corporation
9.5.18 Becton, Dickinson and Company (BD)
9.5.19 Danaher Corporation (Leica Biosystems)
9.5.20 Thermo Fisher Scientific Middle East FZE
10. Bahrain Oncology Companion Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Industry and Commerce
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research and Development
10.2.3 Budget Allocation for Cancer Treatment
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Laboratories
10.3.3 Research Institutions
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Infrastructure Readiness
10.4.3 Technology Acceptance
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Clinical Outcomes
10.5.2 Cost Savings Analysis
10.5.3 Expansion into New Applications
11. Bahrain Oncology Companion Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from Bahrain's Ministry of Health regarding oncology and diagnostics
- Review of academic journals and articles focusing on companion diagnostics in oncology
- Examination of market reports from healthcare industry associations and regulatory bodies in Bahrain
Primary Research
- Interviews with oncologists and pathologists practicing in Bahrain's leading hospitals
- Surveys conducted with laboratory managers and diagnostic service providers
- Focus group discussions with healthcare policymakers and industry experts
Validation & Triangulation
- Cross-validation of findings through multiple data sources including government and private sector reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks performed through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall oncology market size in Bahrain and its growth trajectory
- Segmentation of the market by types of companion diagnostics and therapeutic areas
- Incorporation of demographic data and cancer prevalence rates in Bahrain
Bottom-up Modeling
- Collection of sales data from leading diagnostic companies operating in Bahrain
- Estimation of market share based on product offerings and pricing strategies
- Volume and revenue projections based on historical data and growth rates
Forecasting & Scenario Analysis
- Development of predictive models using historical growth rates and market trends
- Scenario analysis based on potential regulatory changes and healthcare funding shifts
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists in Private Practice | 60 | Medical Oncologists, Surgical Oncologists |
| Pathology Laboratories | 50 | Laboratory Directors, Chief Pathologists |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Regulatory Affairs Managers |
| Diagnostic Equipment Suppliers | 45 | Sales Managers, Product Specialists |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Patient Support Coordinators |
Frequently Asked Questions
What is the current value of the Bahrain Oncology Companion Diagnostics Market?
The Bahrain Oncology Companion Diagnostics Market is valued at approximately USD 120 million, reflecting significant growth driven by the increasing prevalence of cancer and advancements in personalized medicine and targeted therapies.