About the Report
Base Year 2024GCC Ligation Devices Market Overview
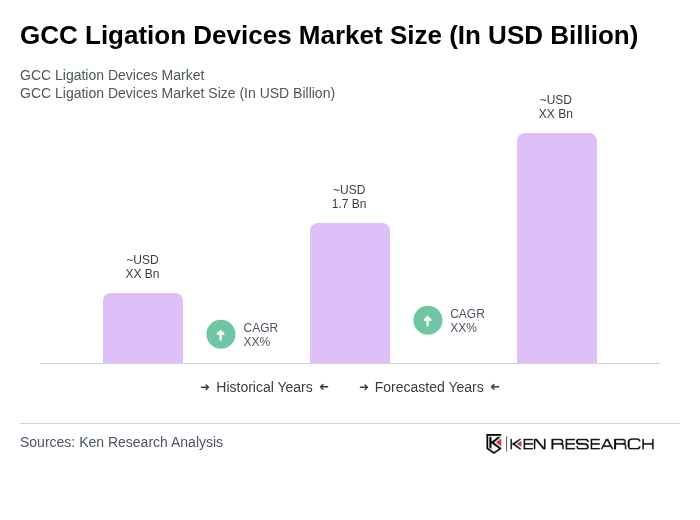
- The GCC Ligation Devices Market is valued at USD 1.7 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, advancements in minimally invasive surgical techniques, and a rising number of surgical procedures across the region. The demand for ligation devices is further supported by the expansion of endoscopic and laparoscopic procedures, which improve patient recovery times and reduce hospital stays. Vendors are increasingly offering single-use and prepackaged ligation systems to streamline workflows and enhance procedural safety .
- Key players in this market include Saudi Arabia, the UAE, and Qatar, which dominate due to their advanced healthcare infrastructure, high healthcare expenditure, and a growing population requiring surgical interventions. The presence of leading medical device manufacturers and a strategic focus on healthcare modernization in these countries further contribute to their market leadership .
- The Medical Devices Interim Regulation, issued by the Saudi Food and Drug Authority (SFDA) in 2021, mandates that all ligation devices used in public and private healthcare facilities must be certified and registered with the SFDA. This regulation enhances patient safety and ensures the quality of surgical procedures, thereby driving the demand for high-quality ligation devices in the market. The regulation covers product registration, conformity assessment, and post-market surveillance requirements .
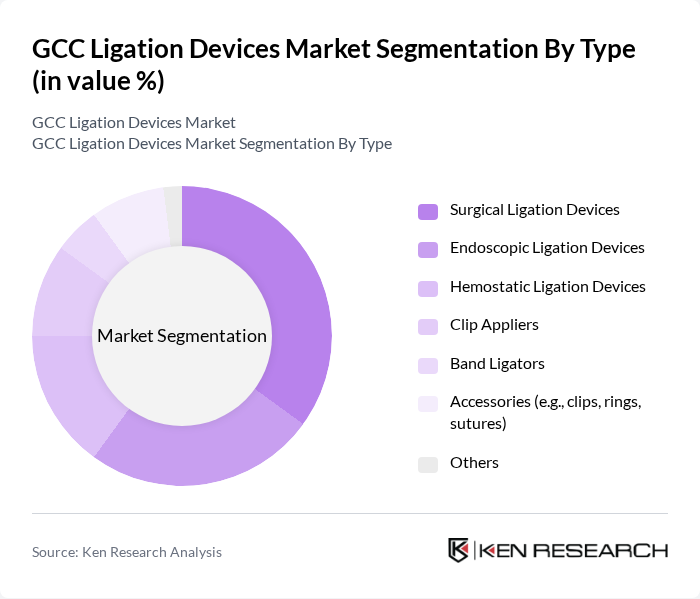
GCC Ligation Devices Market Segmentation
By Type:The market is segmented into various types of ligation devices, including Surgical Ligation Devices, Endoscopic Ligation Devices, Hemostatic Ligation Devices, Clip Appliers, Band Ligators, Accessories (e.g., clips, rings, sutures), and Others. Among these, Surgical Ligation Devices hold the largest share due to their extensive use in gastrointestinal, cardiovascular, and urological surgeries. The increasing adoption of minimally invasive and endoscopic procedures is driving significant demand for Endoscopic Ligation Devices, which are gaining traction as hospitals and ambulatory surgical centers expand their advanced surgical offerings .
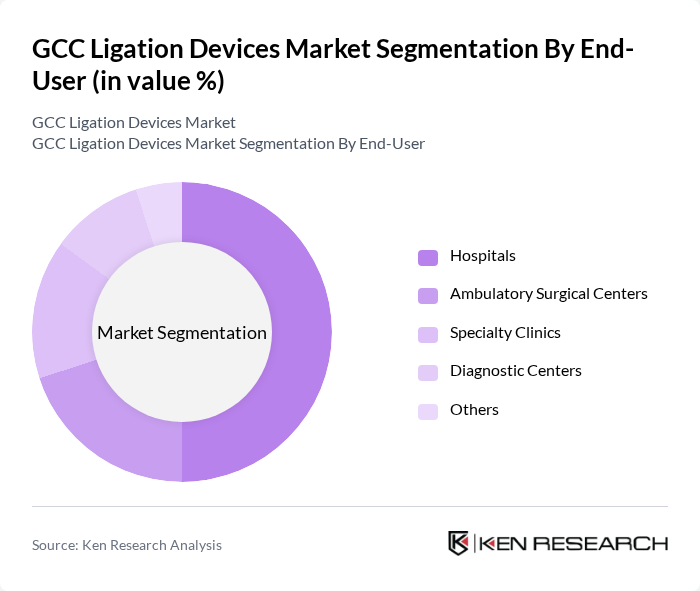
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Diagnostic Centers, and Others. Hospitals remain the dominant end-user segment, accounting for the majority of procedures due to their comprehensive surgical capabilities and higher patient throughput. Ambulatory Surgical Centers are experiencing rapid growth as they offer cost-effective and efficient surgical options, contributing to the overall demand for ligation devices as more procedures shift to outpatient settings .
GCC Ligation Devices Market Competitive Landscape
The GCC Ligation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson (Ethicon), Boston Scientific Corporation, Stryker Corporation, B. Braun Melsungen AG, Teleflex Incorporated, Olympus Corporation, ConMed Corporation, Merit Medical Systems, Inc., Applied Medical Resources Corporation, Grena Ltd., CooperSurgical, Inc., Genicom Co., Ltd., Smith & Nephew plc, and 3M Company contribute to innovation, geographic expansion, and service delivery in this space .
GCC Ligation Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The GCC region is witnessing a significant rise in chronic diseases, with diabetes affecting approximately 17.3% of the adult population in countries like Saudi Arabia and the UAE. This increase drives the demand for ligation devices, as surgical interventions become necessary for managing complications. The World Health Organization reported that chronic diseases account for about 70% of deaths in the region, highlighting the urgent need for effective medical solutions, including ligation devices.
- Advancements in Surgical Techniques:The GCC healthcare sector is rapidly adopting advanced surgical techniques, with a reported 15% increase in minimally invasive surgeries over the past three years. Innovations in ligation devices, such as improved materials and designs, enhance surgical outcomes and reduce recovery times. The integration of robotic-assisted surgeries, which have grown by 20% annually, further supports the demand for sophisticated ligation devices, positioning them as essential tools in modern surgical practices.
- Rising Demand for Minimally Invasive Procedures:There is a notable shift towards minimally invasive procedures in the GCC, with a 30% increase in such surgeries reported in future. Patients prefer these methods due to shorter recovery times and reduced hospital stays. This trend is supported by healthcare investments, which reached USD 89 billion in future, aimed at enhancing surgical capabilities. Consequently, the demand for ligation devices that facilitate these procedures is expected to grow significantly, aligning with patient preferences and healthcare provider capabilities.
Market Challenges
- High Cost of Advanced Ligation Devices:The high cost of advanced ligation devices poses a significant barrier to market growth, with prices ranging from $500 to $2,000 per unit. Many healthcare facilities in the GCC face budget constraints, limiting their ability to invest in these technologies. As a result, hospitals may opt for less effective alternatives, which can compromise patient outcomes and hinder the overall adoption of advanced ligation solutions in the region.
- Stringent Regulatory Requirements:The regulatory landscape for medical devices in the GCC is complex, with stringent requirements for approval and compliance. The average time for device approval can exceed 12 months, creating delays in market entry for new ligation devices. This regulatory burden can deter manufacturers from introducing innovative products, ultimately limiting the options available to healthcare providers and impacting patient care in the region.
GCC Ligation Devices Market Future Outlook
The future of the GCC ligation devices market appears promising, driven by ongoing advancements in medical technology and increasing healthcare investments. As the region's healthcare infrastructure expands, the integration of smart technologies into ligation devices is expected to enhance surgical precision and patient outcomes. Additionally, the rising geriatric population will further fuel demand for effective surgical solutions, creating a favorable environment for market growth and innovation in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure:The GCC countries are investing heavily in healthcare infrastructure, with over USD 89 billion allocated for new hospitals and clinics in future. This expansion will increase the availability of ligation devices, providing manufacturers with a larger market to target and enhancing patient access to advanced surgical options.
- Increasing Investment in Medical Technology:The GCC region is projected to invest approximately $5 billion in medical technology in future. This investment will foster innovation and development of advanced ligation devices, creating opportunities for companies to introduce cutting-edge solutions that meet the evolving needs of healthcare providers and patients alike.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Surgical Ligation Devices Endoscopic Ligation Devices Hemostatic Ligation Devices Clip Appliers Band Ligators Accessories (e.g., clips, rings, sutures) Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Diagnostic Centers Others |
| By Application | Gastrointestinal Surgery Cardiovascular Surgery Urological Surgery Gynecological Surgery General Surgery Bariatric Surgery Others |
| By Material | Absorbable Materials Non-absorbable Materials Others |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Pricing Model | Premium Pricing Competitive Pricing Value-based Pricing Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Gulf Cooperation Council, Ministry of Health and Prevention)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Regulatory Authorities (e.g., Saudi Food and Drug Authority)
Industry Associations and Trade Organizations
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Johnson & Johnson (Ethicon)
Boston Scientific Corporation
Stryker Corporation
B. Braun Melsungen AG
Teleflex Incorporated
Olympus Corporation
ConMed Corporation
Merit Medical Systems, Inc.
Applied Medical Resources Corporation
Grena Ltd.
CooperSurgical, Inc.
Genicom Co., Ltd.
Smith & Nephew plc
3M Company
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. GCC Ligation Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 GCC Ligation Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. GCC Ligation Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Advancements in surgical techniques
3.1.3 Rising demand for minimally invasive procedures
3.1.4 Growing healthcare expenditure in GCC countries
3.2 Market Challenges
3.2.1 High cost of advanced ligation devices
3.2.2 Stringent regulatory requirements
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative treatment options
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in medical technology
3.3.3 Collaborations with local distributors
3.3.4 Rising geriatric population
3.4 Market Trends
3.4.1 Shift towards single-use ligation devices
3.4.2 Integration of smart technology in devices
3.4.3 Focus on patient safety and outcomes
3.4.4 Growth of telemedicine and remote surgeries
3.5 Government Regulation
3.5.1 Compliance with international safety standards
3.5.2 Approval processes for new devices
3.5.3 Monitoring and reporting of device performance
3.5.4 Incentives for local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. GCC Ligation Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. GCC Ligation Devices Market Segmentation
8.1 By Type
8.1.1 Surgical Ligation Devices
8.1.2 Endoscopic Ligation Devices
8.1.3 Hemostatic Ligation Devices
8.1.4 Clip Appliers
8.1.5 Band Ligators
8.1.6 Accessories (e.g., clips, rings, sutures)
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory Surgical Centers
8.2.3 Specialty Clinics
8.2.4 Diagnostic Centers
8.2.5 Others
8.3 By Application
8.3.1 Gastrointestinal Surgery
8.3.2 Cardiovascular Surgery
8.3.3 Urological Surgery
8.3.4 Gynecological Surgery
8.3.5 General Surgery
8.3.6 Bariatric Surgery
8.3.7 Others
8.4 By Material
8.4.1 Absorbable Materials
8.4.2 Non-absorbable Materials
8.4.3 Others
8.5 By Region
8.5.1 Saudi Arabia
8.5.2 UAE
8.5.3 Qatar
8.5.4 Kuwait
8.5.5 Oman
8.5.6 Bahrain
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Distributors
8.6.3 Online Sales
8.6.4 Others
8.7 By Pricing Model
8.7.1 Premium Pricing
8.7.2 Competitive Pricing
8.7.3 Value-based Pricing
8.7.4 Others
9. GCC Ligation Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (GCC-specific, 3-year CAGR)
9.2.4 Market Penetration Rate (Share of GCC ligation device sales)
9.2.5 Product Portfolio Breadth (Number of ligation device SKUs)
9.2.6 Regulatory Approval Track Record (MOH/GCC certifications)
9.2.7 Distribution Network Strength (Number of GCC distributors/partners)
9.2.8 Pricing Competitiveness (Average price vs. market median)
9.2.9 Innovation Index (Patents, new launches in GCC)
9.2.10 Customer Satisfaction Score (GCC end-user feedback)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Johnson & Johnson (Ethicon)
9.5.3 Boston Scientific Corporation
9.5.4 Stryker Corporation
9.5.5 B. Braun Melsungen AG
9.5.6 Teleflex Incorporated
9.5.7 Olympus Corporation
9.5.8 ConMed Corporation
9.5.9 Merit Medical Systems, Inc.
9.5.10 Applied Medical Resources Corporation
9.5.11 Grena Ltd.
9.5.12 CooperSurgical, Inc.
9.5.13 Genicom Co., Ltd.
9.5.14 Smith & Nephew plc
9.5.15 3M Company
10. GCC Ligation Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Key Projects
10.2.3 Funding Sources
10.2.4 Strategic Partnerships
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Ambulatory Surgical Centers
10.3.3 Concerns of Specialty Clinics
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Acceptance
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success
10.5.2 Case Studies
10.5.3 Future Use Cases
10.5.4 Others
11. GCC Ligation Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from healthcare associations and medical device regulatory bodies
- Market analysis publications focusing on surgical and ligation devices
- Statistical data from government health departments and international health organizations
Primary Research
- Interviews with surgeons and healthcare professionals specializing in minimally invasive procedures
- Surveys targeting hospital procurement managers and medical device distributors
- Field interviews with product managers from leading ligation device manufacturers
Validation & Triangulation
- Cross-validation of findings through multiple data sources including sales data and clinical studies
- Triangulation of insights from primary interviews and secondary research findings
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of overall healthcare expenditure trends in the GCC region
- Segmentation of the ligation devices market by product type and application area
- Incorporation of demographic data and disease prevalence rates affecting market demand
Bottom-up Modeling
- Volume estimates based on surgical procedure statistics and ligation device usage rates
- Cost analysis derived from pricing models of various ligation devices
- Estimation of market share based on sales data from key manufacturers
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors such as population growth and healthcare advancements
- Scenario modeling based on potential regulatory changes and technological innovations
- Development of baseline, optimistic, and pessimistic market forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| General Surgery Departments | 100 | Surgeons, Surgical Coordinators |
| Cardiovascular Surgery Units | 60 | Cardiothoracic Surgeons, Nurse Practitioners |
| Gastrointestinal Surgery Clinics | 50 | Gastrointestinal Surgeons, Surgical Assistants |
| Medical Device Procurement Teams | 70 | Procurement Managers, Supply Chain Analysts |
| Healthcare Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the GCC Ligation Devices Market?
The GCC Ligation Devices Market is valued at approximately USD 1.7 billion, reflecting a significant growth driven by the rising prevalence of chronic diseases and advancements in surgical techniques across the region.