About the Report
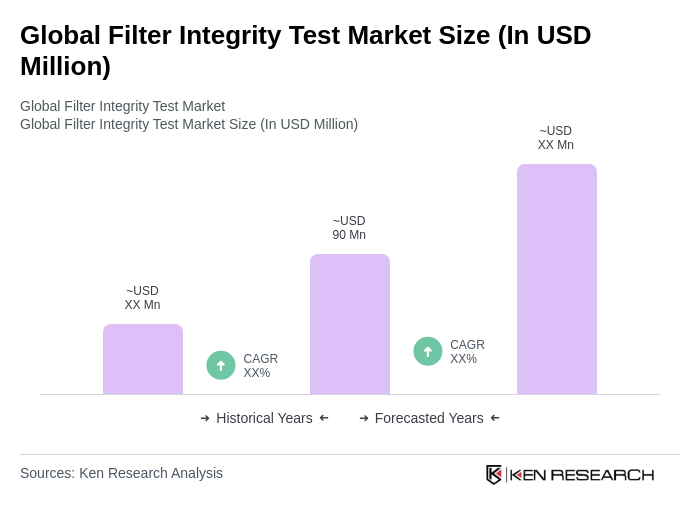
Base Year 2024Global Filter Integrity Test Market Overview
- The Global Filter Integrity Test Market is valued at USD 90 million, based on a five-year historical analysis. Growth is primarily driven by quality assurance needs in pharmaceutical and biopharmaceutical manufacturing, where integrity testing of sterilizing and critical process filters (e.g., bubble point, diffusion, pressure hold, water intrusion tests) is required to ensure sterility assurance, product safety, and regulatory compliance. Rising biologics and vaccine production, stricter data integrity/compliance needs, and adoption of automated testers are key drivers.
- Key regions dominating the market include North America and Europe due to strong pharma/biotech bases and stringent regulatory frameworks that mandate integrity testing in sterile manufacturing; Asia-Pacific is expanding with investments in bioprocessing capacity and healthcare infrastructure.
- In 2023, the U.S. FDA emphasized filter integrity testing as a current good manufacturing practice expectation for sterile drug production through existing guidance and inspections; integrity testing before and after use is an established requirement to ensure sterilizing filter performance, supported by industry consensus (e.g., PDA technical reports) and widely enforced in FDA-regulated facilities.
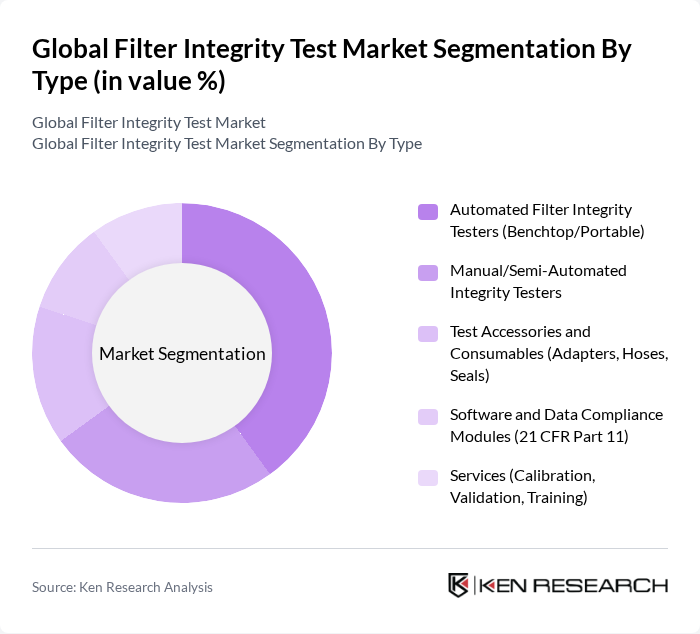
Global Filter Integrity Test Market Segmentation
By Type:The market is segmented into various types, including Automated Filter Integrity Testers (Benchtop/Portable), Manual/Semi-Automated Integrity Testers, Test Accessories and Consumables (Adapters, Hoses, Seals), Software and Data Compliance Modules (21 CFR Part 11), and Services (Calibration, Validation, Training). Among these, Automated Filter Integrity Testers are gaining traction due to their efficiency and accuracy in testing, which is crucial for maintaining compliance in regulated industries.
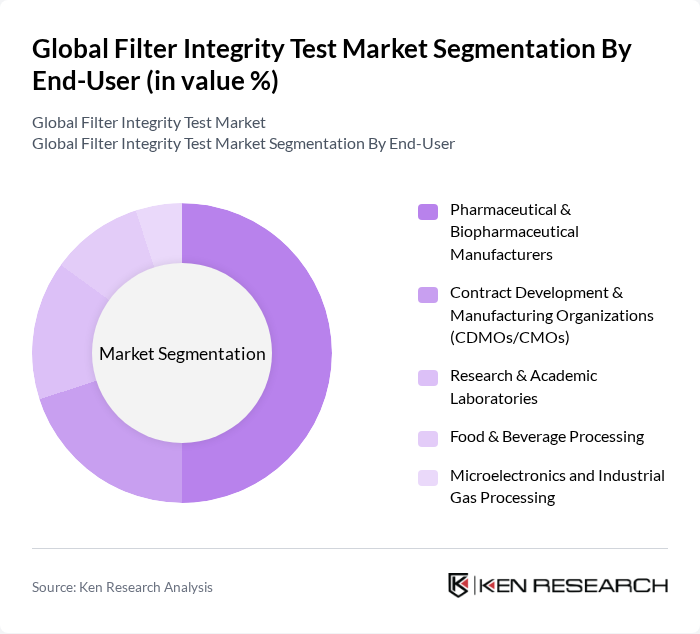
By End-User:The end-user segmentation includes Pharmaceutical & Biopharmaceutical Manufacturers, Contract Development & Manufacturing Organizations (CDMOs/CMOs), Research & Academic Laboratories, Food & Beverage Processing, and Microelectronics and Industrial Gas Processing. The pharmaceutical and biopharmaceutical sectors are the largest consumers of filter integrity testing solutions, driven by stringent regulatory requirements and the need for high-quality production processes.
Global Filter Integrity Test Market Competitive Landscape
The Global Filter Integrity Test Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sartorius AG, Merck KGaA (Merck Life Science), Pall Corporation (Danaher), Parker Hannifin Corporation (Parker Bioscience), Donaldson Company, Inc., Thermo Fisher Scientific Inc., 3M Company (3M Separation and Purification Sciences), Porvair plc (Porvair Filtration Group), Graver Technologies, LLC, Meissner Filtration Products, Inc., Parker domnick hunter Ltd., Saint-Gobain Life Sciences, Donaldson Ultrafilter GmbH, Cobetter Filtration Co., Ltd., Eaton Corporation plc (Eaton Filtration) contribute to innovation, geographic expansion, and service delivery in this space.
Global Filter Integrity Test Market Industry Analysis
Growth Drivers
- Increasing Demand for Quality Assurance:The pharmaceutical and biopharmaceutical industries are projected to reach a combined value of $1.6 trillion in future, driving the need for stringent quality assurance measures. With over 50% of pharmaceutical companies reporting contamination issues, the demand for filter integrity testing is critical. This heightened focus on quality assurance is expected to propel the market for filter integrity tests, as companies seek to ensure product safety and compliance with industry standards.
- Rising Regulatory Requirements:Regulatory bodies, including the FDA and EMA, have established rigorous guidelines for filter integrity testing, with over 75% of pharmaceutical manufacturers reporting increased compliance costs. In future, the global regulatory landscape is expected to tighten further, necessitating advanced testing methods. This trend is anticipated to drive the adoption of filter integrity tests, as companies strive to meet evolving regulatory demands and avoid costly penalties associated with non-compliance.
- Technological Advancements in Testing Methods:The filter integrity testing market is witnessing significant technological innovations, with investments in R&D projected to exceed $6 billion in future. New testing methods, such as automated systems and real-time monitoring technologies, are enhancing accuracy and efficiency. These advancements are expected to attract more companies to adopt filter integrity testing solutions, as they seek to improve operational efficiency and reduce the risk of contamination in sterile environments.
Market Challenges
- High Costs of Advanced Testing Equipment:The initial investment for advanced filter integrity testing equipment can exceed $120,000, posing a significant barrier for smaller companies. With over 45% of industry players citing budget constraints, the high costs associated with acquiring and maintaining sophisticated testing systems can limit market growth. This challenge is particularly pronounced in emerging markets, where financial resources are often more restricted.
- Limited Availability of Skilled Professionals:The filter integrity testing sector faces a shortage of qualified professionals, with an estimated 35% of companies reporting difficulties in hiring skilled technicians. This talent gap is exacerbated by the rapid technological advancements in testing methods, which require specialized training. As the market grows, the lack of skilled labor may hinder the effective implementation of filter integrity testing protocols, impacting overall industry performance.
Global Filter Integrity Test Market Future Outlook
The future of the filter integrity test market appears promising, driven by ongoing advancements in technology and increasing regulatory scrutiny. As the biopharmaceutical sector continues to expand, the demand for reliable testing solutions is expected to rise. Companies are likely to invest in automated systems and IoT integration to enhance testing efficiency. Furthermore, the focus on sustainability will encourage the development of eco-friendly testing methods, aligning with global environmental goals and fostering innovation in the industry.
Market Opportunities
- Expansion of the Biopharmaceutical Sector:The biopharmaceutical market is projected to grow to $650 billion in future, creating significant opportunities for filter integrity testing providers. As more biopharmaceutical companies emerge, the demand for reliable testing solutions will increase, driving market growth and innovation in testing technologies.
- Development of Innovative Filter Technologies:The ongoing research and development in filter technologies present a lucrative opportunity for market players. With investments in innovative solutions expected to reach $2.5 billion in future, companies that focus on developing advanced filter technologies will likely capture a significant share of the growing market, meeting the evolving needs of the pharmaceutical industry.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Automated Filter Integrity Testers (Benchtop/Portable) Manual/Semi-Automated Integrity Testers Test Accessories and Consumables (Adapters, Hoses, Seals) Software and Data Compliance Modules (21 CFR Part 11) Services (Calibration, Validation, Training) |
| By End-User | Pharmaceutical & Biopharmaceutical Manufacturers Contract Development & Manufacturing Organizations (CDMOs/CMOs) Research & Academic Laboratories Food & Beverage Processing Microelectronics and Industrial Gas Processing |
| By Application | Sterile Liquid Filtration (Fill-Finish, Parenterals, Vaccines) Gas/Vent Filter Integrity (Air, Nitrogen, CO?) Water and WFI/Buffer Filtration Single-Use System Integrity Testing Others |
| By Distribution Channel | Direct Sales (OEM) Authorized Distributors/Systems Integrators Online/Inside Sales Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Filter Material | Polyethersulfone (PES) Polyvinylidene Fluoride (PVDF) Polytetrafluoroethylene (PTFE) Nylon Polypropylene (PP) Others |
| By Testing Method | Bubble Point Test Diffusion/Forward-Flow Test Pressure Hold/Decay Test Water Intrusion Test (WIT) Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Filter Integrity Testing Equipment
Pharmaceutical and Biotechnology Companies
Quality Control and Assurance Departments
Healthcare and Medical Device Companies
Industry Associations (e.g., Parenteral Drug Association)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Sartorius AG
Merck KGaA (Merck Life Science)
Pall Corporation (Danaher)
Parker Hannifin Corporation (Parker Bioscience)
Donaldson Company, Inc.
Thermo Fisher Scientific Inc.
3M Company (3M Separation and Purification Sciences)
Porvair plc (Porvair Filtration Group)
Graver Technologies, LLC
Meissner Filtration Products, Inc.
Parker domnick hunter Ltd.
Saint-Gobain Life Sciences
Donaldson Ultrafilter GmbH
Cobetter Filtration Co., Ltd.
Eaton Corporation plc (Eaton Filtration)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Filter Integrity Test Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Filter Integrity Test Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Filter Integrity Test Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for quality assurance in pharmaceutical and biopharmaceutical industries
3.1.2 Rising regulatory requirements for filter integrity testing
3.1.3 Technological advancements in filter testing methods
3.1.4 Growing awareness of contamination risks in sterile environments
3.2 Market Challenges
3.2.1 High costs associated with advanced testing equipment
3.2.2 Limited availability of skilled professionals for testing
3.2.3 Variability in regulatory standards across regions
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of the biopharmaceutical sector
3.3.2 Development of innovative filter technologies
3.3.3 Increasing investments in healthcare infrastructure
3.3.4 Growing demand for environmental monitoring solutions
3.4 Market Trends
3.4.1 Shift towards automated filter integrity testing solutions
3.4.2 Increasing adoption of single-use systems in manufacturing
3.4.3 Focus on sustainability and eco-friendly testing methods
3.4.4 Integration of IoT in filter testing processes
3.5 Government Regulation
3.5.1 FDA guidelines on filter integrity testing
3.5.2 EU regulations on pharmaceutical manufacturing standards
3.5.3 ISO standards for filter testing procedures
3.5.4 Environmental regulations impacting filter disposal
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Filter Integrity Test Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Filter Integrity Test Market Segmentation
8.1 By Type
8.1.1 Automated Filter Integrity Testers (Benchtop/Portable)
8.1.2 Manual/Semi-Automated Integrity Testers
8.1.3 Test Accessories and Consumables (Adapters, Hoses, Seals)
8.1.4 Software and Data Compliance Modules (21 CFR Part 11)
8.1.5 Services (Calibration, Validation, Training)
8.2 By End-User
8.2.1 Pharmaceutical & Biopharmaceutical Manufacturers
8.2.2 Contract Development & Manufacturing Organizations (CDMOs/CMOs)
8.2.3 Research & Academic Laboratories
8.2.4 Food & Beverage Processing
8.2.5 Microelectronics and Industrial Gas Processing
8.3 By Application
8.3.1 Sterile Liquid Filtration (Fill-Finish, Parenterals, Vaccines)
8.3.2 Gas/Vent Filter Integrity (Air, Nitrogen, CO?)
8.3.3 Water and WFI/Buffer Filtration
8.3.4 Single-Use System Integrity Testing
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales (OEM)
8.4.2 Authorized Distributors/Systems Integrators
8.4.3 Online/Inside Sales
8.4.4 Others
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Filter Material
8.6.1 Polyethersulfone (PES)
8.6.2 Polyvinylidene Fluoride (PVDF)
8.6.3 Polytetrafluoroethylene (PTFE)
8.6.4 Nylon
8.6.5 Polypropylene (PP)
8.6.6 Others
8.7 By Testing Method
8.7.1 Bubble Point Test
8.7.2 Diffusion/Forward-Flow Test
8.7.3 Pressure Hold/Decay Test
8.7.4 Water Intrusion Test (WIT)
8.7.5 Others
9. Global Filter Integrity Test Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Product Portfolio Breadth (Test types supported: bubble point, diffusion, pressure hold, WIT)
9.2.3 Installed Base of Testers (units in field)
9.2.4 Biopharma Penetration (share of revenue from pharma/biotech)
9.2.5 Compliance & Certifications (21 CFR Part 11, GMP data integrity, GAMP 5)
9.2.6 R&D Intensity (% of revenue; patents/publications)
9.2.7 Global Service Coverage (calibration/validation sites, regions served)
9.2.8 Integration Capabilities (MES/LIMS/SCADA connectivity)
9.2.9 Average Selling Price Band (manual vs automated systems)
9.2.10 Innovation Cadence (new product releases/firmware updates per year)
9.2.11 Lead Time and Delivery Reliability
9.2.12 Customer Satisfaction/Net Promoter Score (enterprise accounts)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sartorius AG
9.5.2 Merck KGaA (Merck Life Science)
9.5.3 Pall Corporation (Danaher)
9.5.4 Parker Hannifin Corporation (Parker Bioscience)
9.5.5 Donaldson Company, Inc.
9.5.6 Thermo Fisher Scientific Inc.
9.5.7 3M Company (3M Separation and Purification Sciences)
9.5.8 Porvair plc (Porvair Filtration Group)
9.5.9 Graver Technologies, LLC
9.5.10 Meissner Filtration Products, Inc.
9.5.11 Parker domnick hunter Ltd.
9.5.12 Saint-Gobain Life Sciences
9.5.13 Donaldson Ultrafilter GmbH
9.5.14 Cobetter Filtration Co., Ltd.
9.5.15 Eaton Corporation plc (Eaton Filtration)
10. Global Filter Integrity Test Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government procurement policies
10.1.2 Budget allocation for healthcare and pharmaceuticals
10.1.3 Compliance with international standards
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in laboratory infrastructure
10.2.2 Funding for research and development
10.2.3 Expenditure on quality control measures
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in meeting regulatory compliance
10.3.2 Issues with filter performance and reliability
10.3.3 Cost constraints in testing procedures
10.4 User Readiness for Adoption
10.4.1 Awareness of filter integrity testing importance
10.4.2 Training and skill development needs
10.4.3 Technology adoption barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of testing efficiency improvements
10.5.2 Expansion into new applications
10.5.3 Long-term cost savings analysis
11. Global Filter Integrity Test Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Key partnerships and resources
1.5 Customer segments analysis
1.6 Cost structure evaluation
1.7 Channels and customer relationships
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target market identification
2.4 Communication strategies
2.5 Digital marketing initiatives
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing strategies
4.4 Customer willingness to pay
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends and needs
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service strategies
6.3 Customer feedback mechanisms
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Competitive advantages
8. Key Activities
8.1 Regulatory compliance measures
8.2 Branding efforts
8.3 Distribution setup activities
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategies
9.1.3 Packaging approaches
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model evaluation
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors identification
14.2 Joint Ventures opportunities
14.3 Acquisition targets analysis
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity timelines
15.2.2 Milestone tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from leading market research firms focusing on filter integrity testing
- Published articles and white papers from filtration technology journals
- Regulatory guidelines and standards from organizations such as ISO and ASTM
Primary Research
- Interviews with laboratory managers in pharmaceutical and biotechnology companies
- Surveys with quality assurance professionals in the food and beverage sector
- Field interviews with engineers specializing in filtration systems
Validation & Triangulation
- Cross-validation of findings through multiple industry sources and expert opinions
- Triangulation of data from market reports, expert interviews, and regulatory insights
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of global filtration market size and growth trends
- Segmentation by application areas such as pharmaceuticals, food & beverage, and water treatment
- Incorporation of emerging trends in filter integrity testing technologies
Bottom-up Modeling
- Estimation of market size based on sales data from leading filter manufacturers
- Operational cost analysis of filter integrity testing services
- Volume estimates based on the number of tests conducted across various industries
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors like regulatory changes and technological advancements
- Scenario modeling based on market dynamics and potential disruptions
- Baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Filter Integrity Testing | 110 | Quality Control Managers, Laboratory Directors |
| Food & Beverage Filtration Processes | 80 | Production Managers, Quality Assurance Specialists |
| Water Treatment Filtration Systems | 70 | Environmental Engineers, Operations Supervisors |
| Industrial Filtration Applications | 60 | Process Engineers, Maintenance Managers |
| Research & Development in Filtration Technologies | 90 | R&D Managers, Product Development Engineers |
Frequently Asked Questions
What is the current value of the Global Filter Integrity Test Market?
The Global Filter Integrity Test Market is valued at approximately USD 90 million, based on a five-year historical analysis. This valuation reflects the increasing demand for quality assurance in pharmaceutical and biopharmaceutical manufacturing.