About the Report
Base Year 2024Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Overview
- The Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market is valued at USD 4.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of IPF, advancements in drug development—particularly the adoption of antifibrotic therapies such as pirfenidone and nintedanib—and rising awareness regarding the disease. The market is also supported by the growing geriatric population, which is more susceptible to respiratory diseases, thereby increasing the demand for effective treatment options. Recent trends include a focus on early diagnosis, improved long-term patient management, and the development of novel therapeutic agents to address unmet clinical needs .
- Key regions dominating the market include North America and Europe, primarily due to the presence of advanced healthcare infrastructure, high healthcare expenditure, and significant investments in research and development. The United States, in particular, leads the market due to its robust pharmaceutical industry and a high number of clinical trials focused on IPF treatments. The Asia-Pacific region is emerging as the fastest-growing market, driven by rising disease prevalence, expanding elderly populations, and increased adoption of antifibrotic drugs .
- In 2023, the U.S. Food and Drug Administration (FDA) issued the “Pulmonary Fibrosis Drug Development Guidance for Industry,” which provides a framework to expedite the approval process for innovative therapies targeting IPF. This regulation, issued by the U.S. Food and Drug Administration, outlines requirements for clinical trial design, endpoints, and patient selection criteria, aiming to enhance patient access to novel treatments and encourage pharmaceutical investment in IPF therapies .
 Treatment Market.png)
Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Segmentation
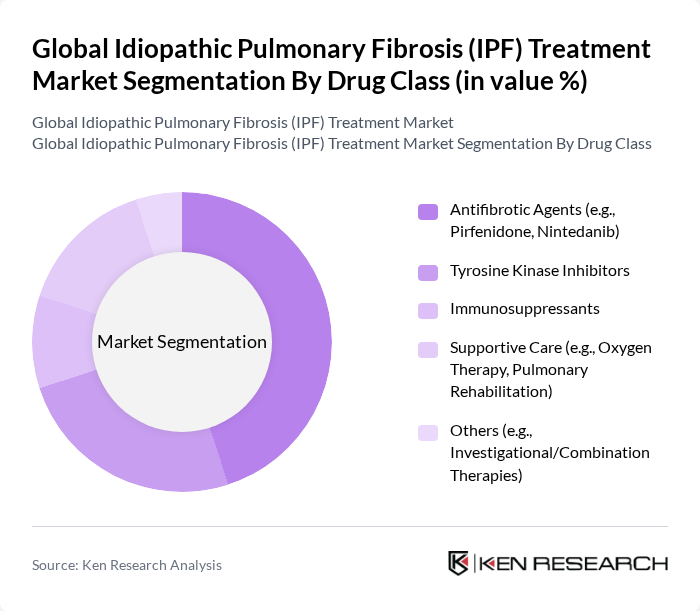
By Drug Class:The drug class segmentation includes various therapeutic agents used in the treatment of IPF. The primary subsegments are Antifibrotic Agents, Tyrosine Kinase Inhibitors, Immunosuppressants, Supportive Care, and Others. Among these, Antifibrotic Agents, such as pirfenidone and nintedanib, dominate the market due to their proven efficacy in slowing disease progression and improving lung function. The increasing adoption of these agents in clinical practice is driven by their favorable safety profiles and the growing body of clinical evidence supporting their use. Recent developments also highlight ongoing research into novel tyrosine kinase inhibitors and combination therapies to address unmet needs .
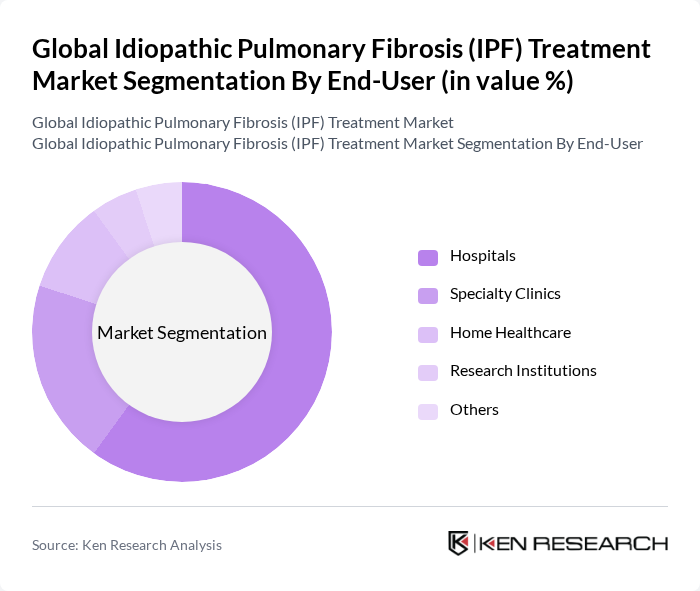
By End-User:The end-user segmentation encompasses various healthcare settings where IPF treatments are administered. This includes Hospitals, Specialty Clinics, Home Healthcare, Research Institutions, and Others. Hospitals are the leading end-user segment, primarily due to their comprehensive facilities and access to advanced medical technologies. The increasing number of hospital admissions for respiratory diseases and the availability of specialized care in hospitals further contribute to their dominance in the market. Specialty clinics and home healthcare are also witnessing growth as patient-centric care models and remote monitoring solutions expand .
Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Competitive Landscape
The Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boehringer Ingelheim, F. Hoffmann-La Roche Ltd, Bristol-Myers Squibb Company, Galapagos NV, FibroGen, Inc., AstraZeneca plc, Ark Biopharmaceutical Co., Ltd., MediciNova, Inc., Gilead Sciences, Inc., Sanofi, Pfizer Inc., Amgen Inc., Teva Pharmaceutical Industries Ltd., Eli Lilly and Company, Vertex Pharmaceuticals Incorporated contribute to innovation, geographic expansion, and service delivery in this space.
Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Industry Analysis
Growth Drivers
- Increasing Prevalence of IPF:The prevalence of idiopathic pulmonary fibrosis (IPF) is rising, with approximately 200,000 cases reported annually in the United States alone. The World Health Organization (WHO) estimates that the global incidence of IPF is around 3-9 cases per 100,000 people, indicating a significant health burden. This increasing prevalence drives demand for effective treatment options, as healthcare systems strive to manage the growing number of patients requiring specialized care and therapies.
- Advancements in Treatment Options:Recent advancements in treatment options, including antifibrotic therapies like nintedanib and pirfenidone, have shown efficacy in slowing disease progression. In future, the FDA approved two new therapies, expanding the treatment landscape. The global market for IPF treatments is projected to reach $3.5 billion in future, driven by these innovations. Such advancements not only improve patient outcomes but also stimulate market growth as healthcare providers adopt these new therapies.
- Rising Awareness and Diagnosis Rates:Increased awareness of IPF among healthcare professionals and the general public has led to improved diagnosis rates. In future, the American Thoracic Society reported a 25% increase in diagnosed cases compared to previous years. Enhanced diagnostic tools, such as high-resolution CT scans, have contributed to this trend. As more patients are diagnosed, the demand for effective treatments rises, further propelling market growth in the IPF treatment sector.
Market Challenges
- High Treatment Costs:The cost of IPF treatments remains a significant barrier, with annual treatment expenses exceeding $100,000 per patient for some therapies. This high financial burden limits access for many patients, particularly in regions with less robust healthcare funding. According to the National Institutes of Health (NIH), only 30% of patients can afford these treatments, leading to disparities in care and outcomes across different demographics and regions.
- Limited Patient Access to Therapies:Access to effective IPF therapies is often restricted due to geographic and economic factors. In rural areas, only 40% of patients have access to specialized pulmonary care, according to the American Lung Association. This limited access can delay treatment initiation, worsening patient outcomes. Additionally, insurance coverage varies significantly, with many plans imposing stringent criteria for therapy approval, further complicating patient access to necessary treatments.
Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Future Outlook
The future of the IPF treatment market appears promising, driven by ongoing research and development efforts aimed at discovering novel therapies. As the healthcare landscape evolves, the integration of personalized medicine and telehealth solutions is expected to enhance patient care. Furthermore, increased collaboration between pharmaceutical companies and research institutions will likely accelerate the development of innovative treatments, addressing unmet needs in the IPF patient population and improving overall health outcomes.
Market Opportunities
- Development of Novel Therapies:The ongoing research into novel therapies presents a significant opportunity for market growth. With over 50 clinical trials currently underway, the potential for breakthrough treatments could reshape the IPF landscape. Successful development and approval of these therapies could lead to improved patient outcomes and increased market share for innovative pharmaceutical companies.
- Expansion into Emerging Markets:Emerging markets, particularly in Asia and Latin America, represent a substantial growth opportunity for IPF treatments. With rising healthcare expenditures projected to reach $1.5 trillion in future in these regions, pharmaceutical companies can capitalize on the increasing demand for effective therapies. Establishing a presence in these markets can enhance accessibility and affordability for patients, driving overall market expansion.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Class | Antifibrotic Agents (e.g., Pirfenidone, Nintedanib) Tyrosine Kinase Inhibitors Immunosuppressants Supportive Care (e.g., Oxygen Therapy, Pulmonary Rehabilitation) Others (e.g., Investigational/Combination Therapies) |
| By End-User | Hospitals Specialty Clinics Home Healthcare Research Institutions Others |
| By Stage of Disease | Early Stage Moderate Stage Advanced Stage Others |
| By Route of Administration | Oral Injectable Inhalation Others |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospital Pharmacies Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Patient Demographics | Age Group Gender Comorbid Conditions Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Insurance Companies and Payers
Clinical Research Organizations
Patient Advocacy Groups
Players Mentioned in the Report:
Boehringer Ingelheim
F. Hoffmann-La Roche Ltd
Bristol-Myers Squibb Company
Galapagos NV
FibroGen, Inc.
AstraZeneca plc
Ark Biopharmaceutical Co., Ltd.
MediciNova, Inc.
Gilead Sciences, Inc.
Sanofi
Pfizer Inc.
Amgen Inc.
Teva Pharmaceutical Industries Ltd.
Eli Lilly and Company
Vertex Pharmaceuticals Incorporated
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of IPF
3.1.2 Advancements in treatment options
3.1.3 Rising awareness and diagnosis rates
3.1.4 Supportive government initiatives
3.2 Market Challenges
3.2.1 High treatment costs
3.2.2 Limited patient access to therapies
3.2.3 Regulatory hurdles
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Development of novel therapies
3.3.2 Expansion into emerging markets
3.3.3 Collaborations and partnerships
3.3.4 Increased investment in research and development
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth of telemedicine in treatment delivery
3.4.3 Focus on patient-centric care
3.4.4 Integration of digital health technologies
3.5 Government Regulation
3.5.1 Approval processes for new drugs
3.5.2 Pricing regulations
3.5.3 Reimbursement policies
3.5.4 Clinical trial regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Segmentation
8.1 By Drug Class
8.1.1 Antifibrotic Agents (e.g., Pirfenidone, Nintedanib)
8.1.2 Tyrosine Kinase Inhibitors
8.1.3 Immunosuppressants
8.1.4 Supportive Care (e.g., Oxygen Therapy, Pulmonary Rehabilitation)
8.1.5 Others (e.g., Investigational/Combination Therapies)
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty Clinics
8.2.3 Home Healthcare
8.2.4 Research Institutions
8.2.5 Others
8.3 By Stage of Disease
8.3.1 Early Stage
8.3.2 Moderate Stage
8.3.3 Advanced Stage
8.3.4 Others
8.4 By Route of Administration
8.4.1 Oral
8.4.2 Injectable
8.4.3 Inhalation
8.4.4 Others
8.5 By Distribution Channel
8.5.1 Retail Pharmacies
8.5.2 Online Pharmacies
8.5.3 Hospital Pharmacies
8.5.4 Others
8.6 By Geography
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa
8.7 By Patient Demographics
8.7.1 Age Group
8.7.2 Gender
8.7.3 Comorbid Conditions
8.7.4 Others
9. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Global IPF Treatment Revenue
9.2.4 R&D Investment in IPF Pipeline (USD Millions)
9.2.5 Number of Approved IPF Therapies
9.2.6 Clinical Pipeline Strength (Number of IPF Candidates in Development)
9.2.7 Geographic Market Presence (Regions/Countries)
9.2.8 Year-on-Year IPF Revenue Growth Rate (%)
9.2.9 Market Share in IPF Segment (%)
9.2.10 Time-to-Market for New IPF Therapies (Months)
9.2.11 Strategic Partnerships/Collaborations in IPF
9.2.12 Patient Access/Distribution Reach
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Boehringer Ingelheim
9.5.2 F. Hoffmann-La Roche Ltd
9.5.3 Bristol-Myers Squibb Company
9.5.4 Galapagos NV
9.5.5 FibroGen, Inc.
9.5.6 AstraZeneca plc
9.5.7 Ark Biopharmaceutical Co., Ltd.
9.5.8 MediciNova, Inc.
9.5.9 Gilead Sciences, Inc.
9.5.10 Sanofi
9.5.11 Pfizer Inc.
9.5.12 Amgen Inc.
9.5.13 Teva Pharmaceutical Industries Ltd.
9.5.14 Eli Lilly and Company
9.5.15 Vertex Pharmaceuticals Incorporated
10. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for IPF Treatments
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Compliance Requirements
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Infrastructure
10.2.2 Spending on Research and Development
10.2.3 Budget for Patient Support Programs
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability of Medications
10.3.3 Quality of Care
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Technology Adoption
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Long-term Cost Savings
10.5.3 Patient Satisfaction Metrics
10.5.4 Others
11. Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and medical journals on IPF treatment trends
- Review of clinical trial registries and outcomes related to IPF therapies
- Examination of market reports and white papers from pharmaceutical industry analysts
Primary Research
- Interviews with pulmonologists and respiratory specialists to gather insights on treatment preferences
- Surveys with healthcare providers involved in the management of IPF patients
- Focus groups with patients diagnosed with IPF to understand treatment experiences and outcomes
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through feedback from a panel of medical professionals and industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the global IPF treatment market size based on healthcare expenditure data
- Segmentation of the market by drug class, including antifibrotic agents and supportive therapies
- Incorporation of demographic data on IPF prevalence and incidence rates across regions
Bottom-up Modeling
- Analysis of sales data from leading pharmaceutical companies producing IPF treatments
- Estimation of treatment costs based on pricing models and reimbursement frameworks
- Volume estimates derived from patient population data and treatment adherence rates
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as aging population and healthcare access
- Scenario modeling based on potential drug approvals and market entry of new therapies
- Baseline, optimistic, and pessimistic forecasts through 2030 based on market dynamics
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pulmonology Clinics | 120 | Pulmonologists, Nurse Practitioners |
| Respiratory Therapy Centers | 90 | Respiratory Therapists, Clinical Coordinators |
| Pharmaceutical Distributors | 50 | Sales Managers, Product Managers |
| Patient Advocacy Groups | 40 | Patient Advocates, Program Directors |
| Healthcare Payers | 45 | Health Insurance Analysts, Medical Directors |
Frequently Asked Questions
What is the current value of the Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market?
The Global Idiopathic Pulmonary Fibrosis (IPF) Treatment Market is valued at approximately USD 4.5 billion, driven by the increasing prevalence of IPF and advancements in drug development, particularly antifibrotic therapies like pirfenidone and nintedanib.