
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Outlook to 2030
Region:Asia
Author(s):Meenakshi Bisht
Product Code:KROD7406
November 2024
86
About the Report
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Overview
- The Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market is valued at USD 1.37 billion, backed by a consistent five-year analysis reflecting the rapid adoption of outsourcing services. The market's expansion is driven by the increasing complexity of pharmaceutical products, strict regulatory requirements for quality control, and the need to reduce time-to-market for new drugs.
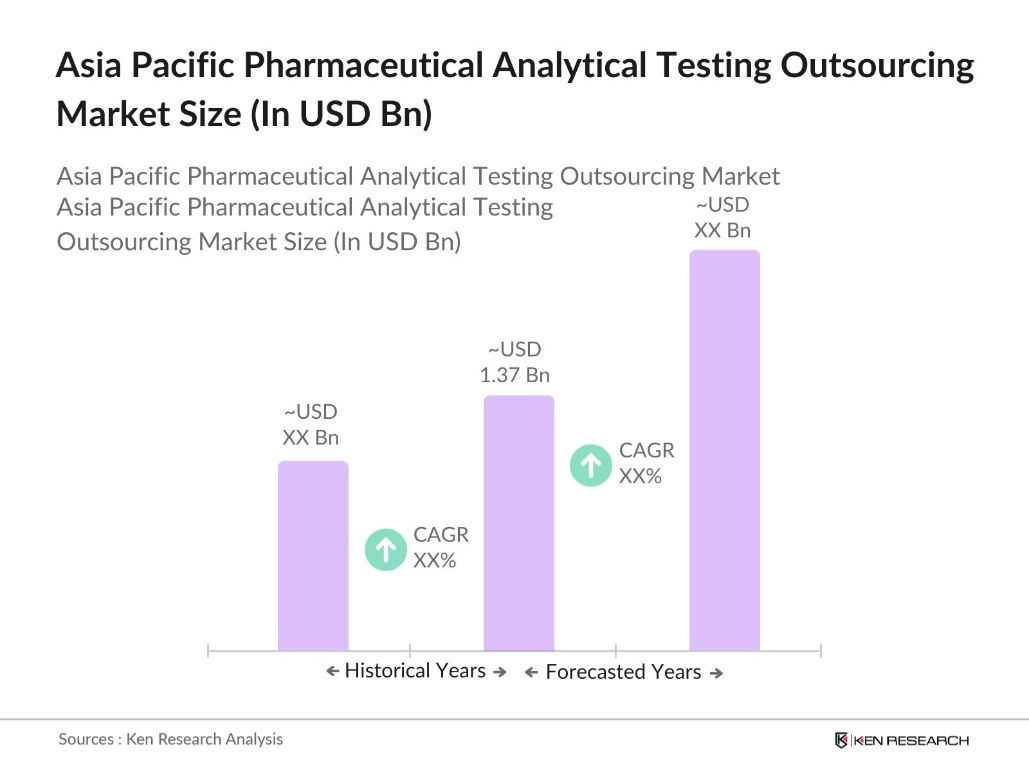
- Dominant countries in the Asia Pacific region include China, Japan, and India, which lead due to their established pharmaceutical sectors, robust manufacturing bases, and substantial investments in R&D. China and India, in particular, benefit from lower operational costs and large, skilled labor pools, while Japans stringent regulatory standards make it a key player for high-quality testing and analysis services in pharmaceutical manufacturing and R&D.
- The Asia-Pacific region adheres to various regulatory standards set by the International Council for Harmonisation (ICH) and national health authorities. In 2023, Asia-Pacific countries harmonized several drug regulations based on ICH guidelines, ensuring consistent standards for pharmaceutical products across the region. Such harmonization promotes smoother cross-border trade and supports the global competitiveness of APAC pharmaceutical companies, although it requires ongoing compliance monitoring by analytical testing providers.
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Segmentation
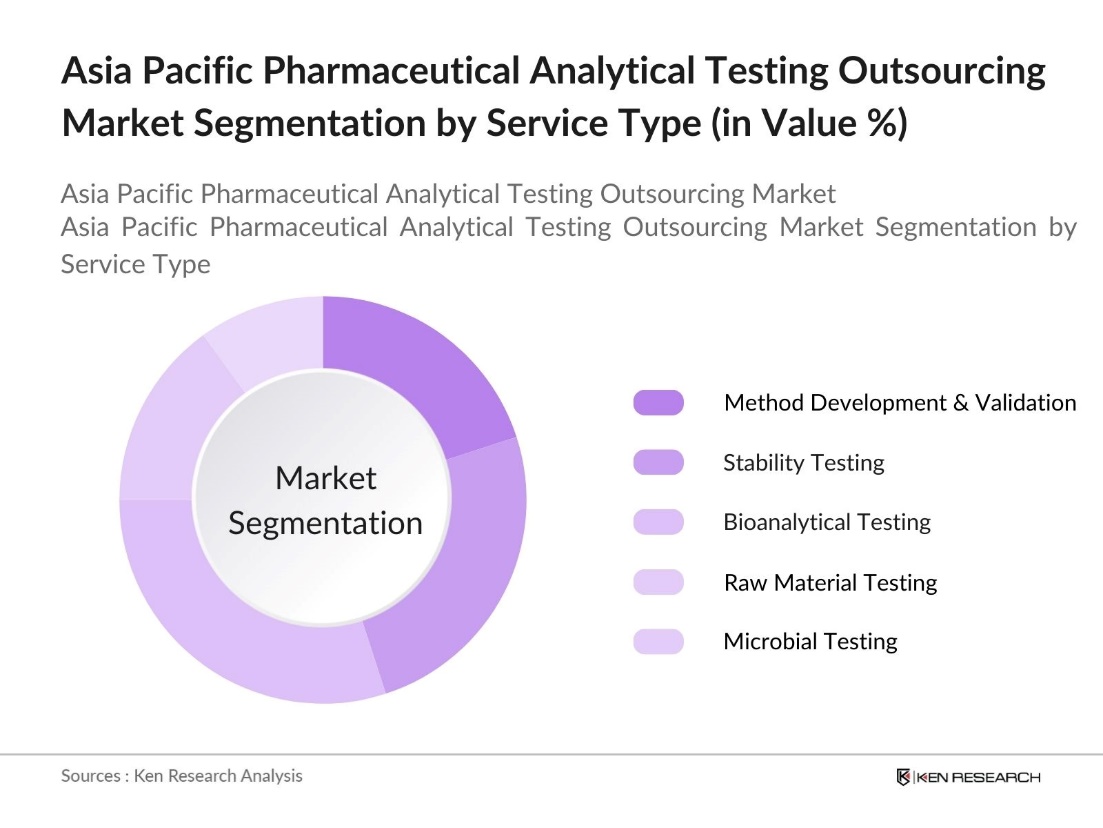
By Service Type: The market is segmented by service type into method development and validation testing, stability testing, bioanalytical testing, raw material testing, and microbial testing. Bioanalytical testing holds a dominant market share due to its critical role in ensuring the safety and efficacy of drugs, especially for biologics and biosimilars. The growing demand for complex drugs and biopharmaceuticals necessitates extensive bioanalytical testing, making it a preferred outsourcing option for pharmaceutical firms aiming to maintain regulatory compliance.
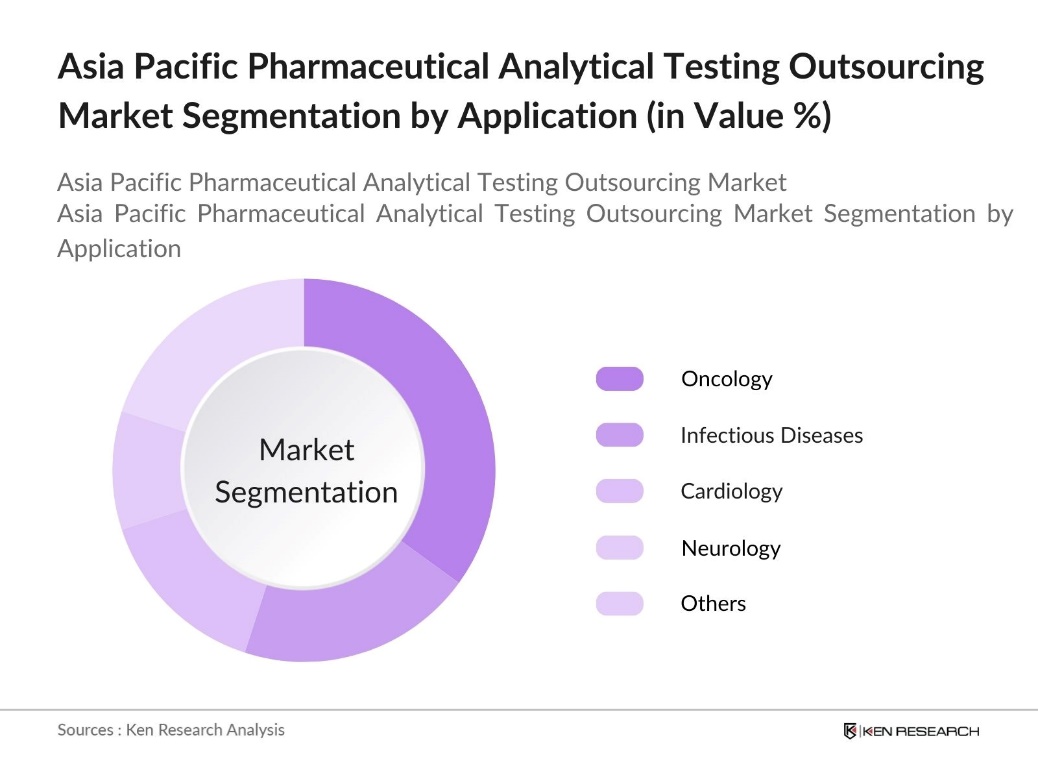
By Application: This market is segmented by application into oncology, infectious diseases, cardiology, neurology, and others. Oncology has a significant share due to the high R&D investment in cancer research and the complexity of cancer treatment drugs. Rigorous testing is required to develop effective oncology drugs, and outsourcing allows companies to expedite testing procedures while reducing costs. This segment continues to grow as the prevalence of cancer rises across the Asia Pacific region.
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Competitive Landscape
The Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market is led by several key players, including multinational corporations with a strong presence in the region. This concentration of large, well-established firms indicates a market dominated by high-quality, trusted players with advanced technology and skilled personnel, ensuring compliance with strict regulatory standards.
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Industry Analysis
Growth Drivers
- Rising R&D Expenditure by Pharmaceutical Companies: Pharmaceutical companies in the Asia-Pacific region have significantly increased their research and development (R&D) investments to foster innovation and develop new drugs. For instance, China's pharmaceutical R&D spending reached approximately $29 billion in 2022, positioning it as the second-largest spender globally after the United States. This substantial investment underscores the region's commitment to advancing pharmaceutical research and development.
- Increasing Complexity of Drug Development: The drug development process has become more intricate, necessitating advanced analytical testing to ensure drug safety and efficacy. In 2023, the global pharmaceutical industry had over 16,000 drugs in various stages of development, with a significant portion originating from the Asia-Pacific region. This complexity drives the demand for specialized analytical testing services to support the development of these novel therapies.
- Regulatory Requirements: Pharmaceutical companies in the Asia-Pacific region must comply with strict regional and global regulatory standards. Initiatives like those from the Asia-Pacific Economic Cooperation (APEC) promote regulatory alignment, especially around Good Manufacturing Practices (GMP). This convergence helps streamline market entry and ensures consistent drug quality and safety across member economies, highlighting the critical need for adherence to evolving regulatory requirements in the pharmaceutical industry.
Market Challenges
- Quality Control and Reliability Issues: Maintaining consistent quality and reliability in outsourced analytical testing presents a notable challenge for pharmaceutical companies. Variability in testing accuracy can compromise drug quality, potentially leading to regulatory non-compliance and even product recalls. This underscores the critical need for stringent oversight and robust quality control in outsourced testing services to uphold safety and efficacy standards.
- Limited Skilled Workforce for Specialized Analytical Testing: The Asia-Pacific region faces a shortage of skilled professionals trained in specialized analytical testing techniques. This talent gap poses challenges for companies striving to uphold high standards in testing quality. The growing demand for skilled personnel in this expanding industry emphasizes the need for workforce development to support advanced pharmaceutical testing requirements.
Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Future Outlook
Over the coming years, the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market is projected to experience robust growth, driven by advancements in pharmaceutical R&D, increasing regulatory pressures, and a growing need for cost-effective solutions. The rising adoption of digital technologies, such as AI-driven data analysis and automation in analytical testing, is anticipated to further streamline processes and enhance testing accuracy.
Market Opportunities
- Demand for Biologics and Biosimilars: The Asia-Pacific region is experiencing growing demand for biologics and biosimilars, which require specialized analytical testing support. This trend opens up opportunities for testing service providers to address the unique analytical needs associated with biologics development, including rigorous quality and safety assessments.
- Integration of Advanced Technologies: Advanced technologies like artificial intelligence (AI) and automation are revolutionizing analytical testing processes. This technological shift enhances testing accuracy and efficiency, enabling service providers to offer innovative and efficient solutions that cater to the evolving needs of pharmaceutical companies.
Scope of the Report
|
Service Type |
Method Development and Validation Testing |
|
Application |
Oncology |
|
End-User |
Pharmaceutical & Biotechnology Companies |
|
Technology |
Chromatography |
|
Country |
China |
Products
Key Target Audience
Pharmaceutical and Biotechnology Companies
Contract Research Organizations (CROs)
Healthcare and Life Sciences Companies
Drug Development Firms
Government and Regulatory Bodies (e.g., FDA, CFDA, PMDA)
Investors and Venture Capitalist Firms
Banks and Financial Institutions
Companies
Players Mentioned in the Report
Charles River Laboratories
Eurofins Scientific
SGS SA
WuXi AppTec
Toxikon Corporation
Intertek Group plc
Covance Inc.
PPD, Inc.
Pace Analytical Services, LLC
Almac Group
Table of Contents
1. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Size (In USD Mn)
2.1. Historical Market Size
2.2. Year-on-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Dynamics
3.1. Growth Drivers
3.1.1. Rising R&D Expenditure by Pharmaceutical Companies
3.1.2. Increasing Complexity of Drug Development (Complexity of Analytical Tests)
3.1.3. Regulatory Requirements (Compliance with Regional and Global Standards)
3.1.4. Cost-Saving Needs through Outsourcing
3.2. Market Challenges
3.2.1. Quality Control and Reliability Issues (Variability in Testing Accuracy)
3.2.2. Limited Skilled Workforce for Specialized Analytical Testing
3.2.3. Stringent Regulatory Barriers
3.3. Opportunities
3.3.1. Demand for Biologics and Biosimilars (Analytical Support for Biologics)
3.3.2. Integration of Advanced Technologies (AI and Automation in Testing)
3.3.3. Rising Demand for Specialized Testing Services
3.4. Trends
3.4.1. Shift Towards Outsourcing Across Asia-Pacific (Shift in Cost-Efficiency Models)
3.4.2. Growth of Small and Mid-Scale CROs (Emergence of New Market Players)
3.4.3. Adoption of Digitalized Quality Management Systems
3.5. Regulatory Environment
3.5.1. APAC Regulatory Standards (ICH Guidelines, National Health Standards)
3.5.2. Good Laboratory Practices (GLP) and ISO Certification
3.5.3. Import and Export Regulations for Analytical Samples
3.5.4. Compliance for Biosafety and Biosecurity
3.6. SWOT Analysis
3.7. Value Chain Analysis
3.8. Porters Five Forces Analysis
3.9. Competitive Landscape
4. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Segmentation
4.1. By Service Type (In Value %)
4.1.1. Method Development and Validation Testing
4.1.2. Stability Testing
4.1.3. Bioanalytical Testing
4.1.4. Raw Material Testing
4.1.5. Microbial Testing
4.2. By Application (In Value %)
4.2.1. Oncology
4.2.2. Infectious Diseases
4.2.3. Cardiology
4.2.4. Neurology
4.2.5. Others
4.3. By End-User (In Value %)
4.3.1. Pharmaceutical & Biotechnology Companies
4.3.2. Contract Research Organizations (CROs)
4.3.3. Academic & Research Institutes
4.3.4. Others
4.4. By Technology (In Value %)
4.4.1. Chromatography
4.4.2. Spectroscopy
4.4.3. Polymerase Chain Reaction (PCR)
4.4.4. Cell-based Assays
4.4.5. Others
4.5. By Country (In Value %)
4.5.1. China
4.5.2. Japan
4.5.3. India
4.5.4. Australia
4.5.5. South Korea
4.5.6. Southeast Asia
5. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Competitive Analysis
5.1. Detailed Profiles of Major Competitors
5.1.1. Charles River Laboratories
5.1.2. Eurofins Scientific
5.1.3. SGS SA
5.1.4. WuXi AppTec
5.1.5. Toxikon Corporation
5.1.6. Intertek Group plc
5.1.7. Covance Inc.
5.1.8. PPD, Inc.
5.1.9. Pace Analytical Services, LLC
5.1.10. Almac Group
5.1.11. Syneos Health
5.1.12. ICON plc
5.1.13. Frontage Laboratories
5.1.14. Pharmaceutical Product Development, LLC
5.1.15. Bioreliance Corporation
5.2. Cross Comparison Parameters (Revenue, Market Presence in APAC, Analytical Service Capabilities, Employee Strength, Regulatory Certifications, R&D Investments, Client Base, Regional Offices)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers and Acquisitions
5.6. Investment Analysis
5.7. Partnerships and Alliances
6. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Regulatory Framework
6.1. Regulatory Requirements for Testing Services
6.2. Good Manufacturing Practice (GMP) Standards
6.3. ISO and Quality Certifications
6.4. Compliance Standards for Biosafety Testing
6.5. Ethical Compliance and Confidentiality Regulations
7. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Future Market Size (In USD Mn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Future Market Segmentation
8.1. By Service Type (In Value %)
8.2. By Application (In Value %)
8.3. By End-User (In Value %)
8.4. By Technology (In Value %)
8.5. By Country (In Value %)
9. Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market Analysts Recommendations
9.1. Total Addressable Market (TAM) and Serviceable Available Market (SAM) Analysis
9.2. Customer Segmentation Strategy
9.3. Target Market Opportunities
9.4. Key Marketing Strategies and Brand Positioning
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
In the initial stage, we mapped the ecosystem of the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market by identifying key stakeholders and analyzing market-specific trends. This process involved comprehensive desk research utilizing proprietary and publicly accessible databases to gather industry-level insights.
Step 2: Market Analysis and Construction
We compiled and examined historical data on market growth, penetration, and the impact of regulatory standards. By assessing market data from both supply and demand perspectives, we established a foundational understanding of revenue streams and the influence of emerging technology on testing accuracy.
Step 3: Hypothesis Validation and Expert Consultation
After formulating preliminary market hypotheses, we conducted interviews with industry experts from various pharmaceutical companies. These consultations, conducted through CATIs, provided crucial insights into operational and financial trends, aiding in the validation of our market estimations.
Step 4: Research Synthesis and Final Output
The final stage involved synthesizing data from direct interactions with multiple analytical testing firms. These engagements confirmed data accuracy and allowed us to provide a well-rounded, evidence-based analysis of the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market.
Frequently Asked Questions
01 How big is the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market?
The Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market is valued at USD 1.37 billion, driven by factors such as increasing drug complexity and stringent quality regulations.
02 What are the primary challenges in the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market?
Key challenges in Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market include high regulatory compliance requirements, the need for skilled analysts, and ensuring quality and reliability across different testing providers.
03 Who are the major players in the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market?
Major players in Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market include Charles River Laboratories, Eurofins Scientific, SGS SA, WuXi AppTec, and Intertek Group, known for their extensive service capabilities and regulatory compliance.
04 What are the growth drivers of the Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market?
The Asia Pacific Pharmaceutical Analytical Testing Outsourcing Market growth drivers include increased R&D expenditure, a demand for high-quality drugs, and a preference for cost-efficient outsourced testing services to reduce time-to-market for pharmaceuticals.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.