
Asia Pacific Pharmaceutical Grade Rice Starch Market Outlook to 2030
Region:Asia
Author(s):Paribhasha Tiwari
Product Code:KROD7169
December 2024
89
About the Report
Asia Pacific Pharmaceutical Grade Rice Starch Market Overview
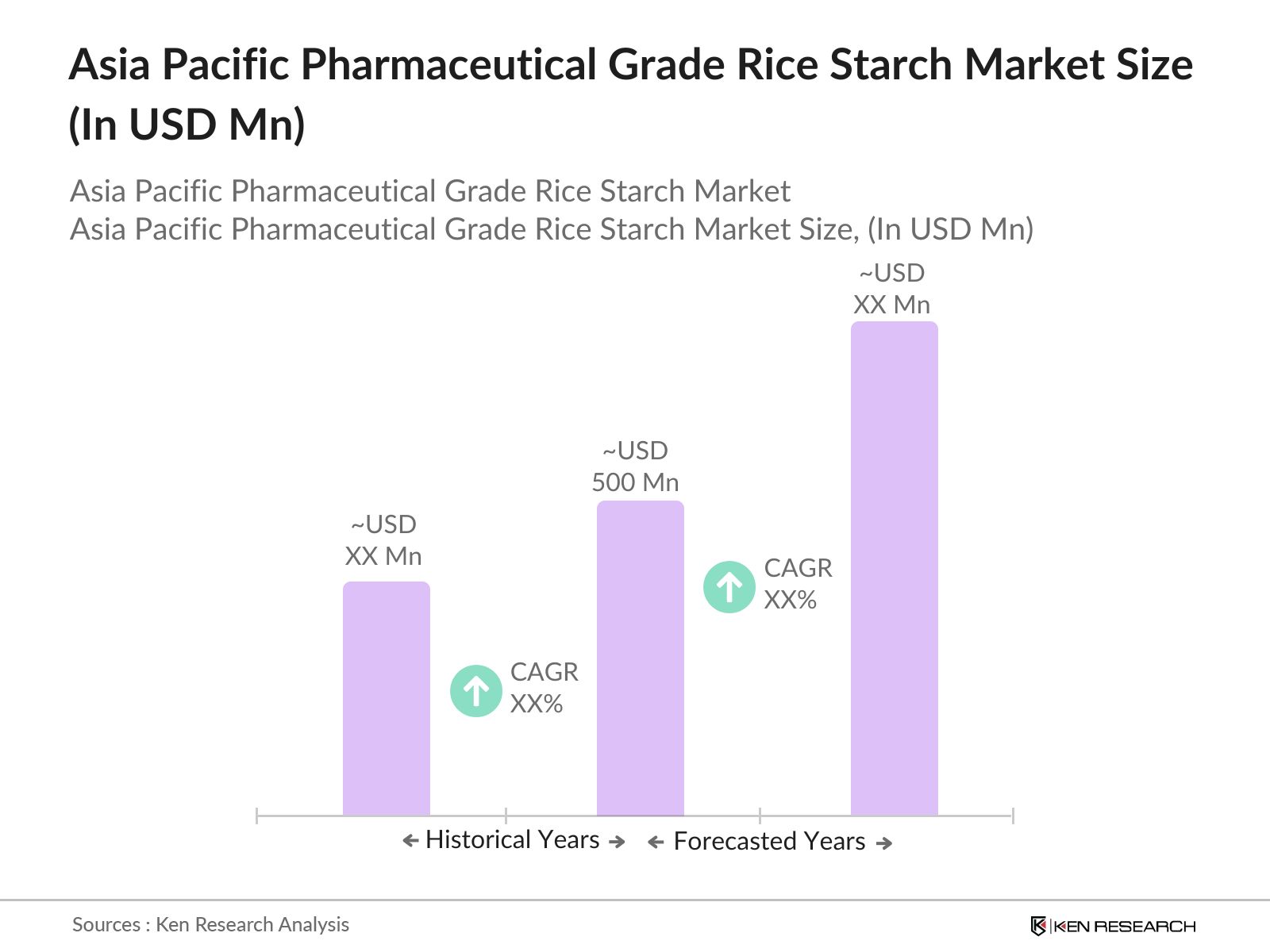
- The Asia Pacific pharmaceutical grade rice starch market is valued at USD 500 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for rice starch as an excipient in pharmaceutical formulations, owing to its hypoallergenic properties and superior binding capabilities. Additionally, the expansion of the pharmaceutical industry in emerging economies within the region has significantly contributed to this market's development.
- China and India are the dominant countries in the Asia Pacific pharmaceutical grade rice starch market. This dominance is attributed to their large-scale rice production, which ensures a steady supply of raw materials, and the rapid growth of their pharmaceutical sectors. Furthermore, favorable government policies supporting pharmaceutical manufacturing and exports have bolstered their positions in the market.
- Governments in India, China, and Brazil have initiated production-linked incentive schemes to boost local manufacturing of pharmaceuticals, including excipients. In India, these incentives contributed to a 1,100 million-unit increase in domestic production capacity in 2023. These measures support starch-based excipient manufacturers, as local suppliers are prioritized over international imports.
Asia Pacific Pharmaceutical Grade Rice Starch Market Segmentation
By Product Type: The Asia Pacific pharmaceutical grade rice starch market is segmented by product type into native rice starch and modified rice starch. Native rice starch holds a dominant market share due to its natural composition, making it highly suitable for pharmaceutical applications that require minimal chemical modification. Its inherent properties, such as excellent binding and disintegration capabilities, make it a preferred choice among pharmaceutical manufacturers.
By Application: The market is further segmented by application into tablet binders, disintegrants, coating agents, capsule fillers, and others. Tablet binders represent the largest segment, driven by the extensive use of rice starch in tablet formulations to enhance cohesion and ensure uniformity. Its compatibility with various active pharmaceutical ingredients and ability to improve tablet hardness without compromising disintegration time contribute to its widespread adoption in this application.
Asia Pacific Pharmaceutical Grade Rice Starch Market Competitive Landscape
The Asia Pacific pharmaceutical grade rice starch market is characterized by the presence of several key players who contribute significantly to its growth and development. These companies are engaged in strategic initiatives such as product innovation, expansion of production capacities, and collaborations to strengthen their market positions.
Asia Pacific Pharmaceutical Grade Rice Starch Market Analysis
Growth Drivers
- Increasing Demand in Pharmaceutical Formulations: The pharmaceutical sector has seen a surge in demand for starch-based excipients, driven by the necessity to produce safe and effective formulations for a growing population. The global demand for pharmaceutical formulations rose to over 7,500 billion units in 2023, indicating the scale of requirements for excipients. The World Health Organization reports that access to essential medicines is expanding, requiring an increase in starch-based components, especially in geographies with aging populations, such as Europe and North America, which is predicted to exceed 950 million by 2025.
- Rising Health Awareness: Health awareness has increased dramatically, with surveys in 2024 showing that over 85% of individuals in urban centers are actively seeking supplements and medications that support healthy lifestyles. This shift is contributing to the demand for starch-based excipients in products such as tablets and capsules. Additionally, the prevalence of chronic diseases worldwide reached over 1.5 billion cases in 2023, further driving the need for safe, sustainable, and plant-based excipients to enhance drug effectiveness.
- Expansion of Pharmaceutical Manufacturing in Asia Pacific: Pharmaceutical manufacturing in the Asia Pacific region is rapidly expanding, with India and China producing approximately 60% of the world's generic drugs. This growth necessitates higher volumes of starch-based materials as excipients. By the end of 2024, it is anticipated that Asia's share in pharmaceutical exports will cross 3,800 million units, with governments supporting domestic production through incentives and subsidies.
Market Challenges
- Availability of Alternative Starches: With the rise of alternative starches, the market faces pressure from materials such as cellulose and synthetic polymers, which are often used as binders and fillers. In 2023, cellulose-based excipients were used in 40% of all drug formulations, presenting significant competition for traditional starch-based options. Alternative sources such as seaweed-derived excipients have also gained ground in applications requiring higher stability.
- High Production Costs: The cost of starch production has been increasing due to raw material costs, labor shortages, and energy expenses. In 2023, the average production cost for pharmaceutical-grade starch rose by over 1,200 units globally due to these factors. This increase can impact smaller manufacturers, who may struggle to maintain competitive pricing without compromising quality.
Asia Pacific Pharmaceutical Grade Rice Starch Market Future Outlook
Over the next five years, the Asia Pacific pharmaceutical grade rice starch market is expected to experience substantial growth. This expansion will be driven by the continuous development of the pharmaceutical industry, increasing demand for natural and hypoallergenic excipients, and advancements in starch processing technologies. Additionally, the rising prevalence of chronic diseases and the subsequent increase in medication consumption are anticipated to further propel market growth.
Market Opportunities
- Emerging Markets in Developing Countries: Developing markets, especially in Africa and Southeast Asia, present a vast potential for growth, as access to healthcare and pharmaceuticals expands. In 2023, Southeast Asias healthcare expenditure crossed 4,200 billion units, reflecting the growing demand for medications. Governments in these regions are promoting local pharmaceutical production, opening doors for starch-based excipient suppliers to establish partnerships and distribution channels.
- Innovations in Drug Delivery Systems: Starch-based excipients are gaining popularity for their adaptability in novel drug delivery systems, such as targeted release and bioavailability-enhancing formulations. By 2023, over 900 million units of such formulations used starch-based materials due to their efficiency in controlling drug release rates. This opportunity is expected to further grow with advancements in nanotechnology and personalized medicine, where starch excipients play an essential role.
Scope of the Report
|
By Product Type |
Native Rice Starch |
|
By Application |
Tablet Binders |
|
By Form |
Powder |
|
By End-User |
Pharmaceutical Manufacturers |
|
By Country |
China |
Products
Key Target Audience
Pharmaceutical Manufacturers
Contract Manufacturing Organizations (CMOs)
Research Laboratories
Raw Material Suppliers
Government and Regulatory Bodies (e.g., Food and Drug Administration)
Investors and Venture Capitalist Firms
Pharmaceutical Equipment Manufacturers
Healthcare Institutions
Companies
Players Mentioned in the Report:
Ingredion Incorporated
BENEO GmbH
AGRANA Beteiligungs-AG
Bangkok Starch Industrial Co., Ltd.
Thai Flour Industry Co., Ltd.
Jiangxi Golden Agriculture Biotech Co., Ltd.
WFM Wholesome Foods
Anhui Lianhe
Anhui Le Huan Tian Biotechnology
Herba Ingredients BV
Table of Contents
1. Asia Pacific Pharmaceutical Grade Rice Starch Market Overview
1.1 Definition and Scope
1.2 Market Taxonomy
1.3 Market Growth Rate
1.4 Market Segmentation Overview
2. Asia Pacific Pharmaceutical Grade Rice Starch Market Size (USD Billion)
2.1 Historical Market Size
2.2 Year-On-Year Growth Analysis
2.3 Key Market Developments and Milestones
3. Asia Pacific Pharmaceutical Grade Rice Starch Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand in Pharmaceutical Formulations
3.1.2 Rising Health Awareness
3.1.3 Expansion of Pharmaceutical Manufacturing in Asia Pacific
3.1.4 Technological Advancements in Starch Processing
3.2 Market Challenges
3.2.1 Availability of Alternative Starches
3.2.2 High Production Costs
3.2.3 Regulatory Compliance Issues
3.3 Opportunities
3.3.1 Emerging Markets in Developing Countries
3.3.2 Innovations in Drug Delivery Systems
3.3.3 Growing Preference for Plant-Based Excipients
3.4 Trends
3.4.1 Shift Towards Clean Label Ingredients
3.4.2 Adoption of Sustainable Production Practices
3.4.3 Integration of Rice Starch in Biopharmaceuticals
3.5 Regulatory Framework
3.5.1 Regional Pharmaceutical Standards
3.5.2 Import and Export Regulations
3.5.3 Quality Assurance and Certification Processes
3.6 SWOT Analysis
3.7 Stakeholder Ecosystem
3.8 Porters Five Forces Analysis
3.9 Competitive Landscape
4. Asia Pacific Pharmaceutical Grade Rice Starch Market Segmentation
4.1 By Product Type (Value %)
4.1.1 Native Rice Starch
4.1.2 Modified Rice Starch
4.2 By Application (Value %)
4.2.1 Tablet Binders
4.2.2 Disintegrants
4.2.3 Coating Agents
4.2.4 Capsule Fillers
4.2.5 Others
4.3 By Form (Value %)
4.3.1 Powder
4.3.2 Granules
4.4 By End-User (Value %)
4.4.1 Pharmaceutical Manufacturers
4.4.2 Contract Manufacturing Organizations (CMOs)
4.4.3 Research Laboratories
4.5 By Country (Value %)
4.5.1 China
4.5.2 India
4.5.3 Japan
4.5.4 South Korea
4.5.5 Australia
4.5.6 Rest of Asia Pacific
5. Asia Pacific Pharmaceutical Grade Rice Starch Competitive Analysis
5.1 Detailed Profiles of Major Companies
5.1.1 Ingredion Incorporated
5.1.2 BENEO GmbH
5.1.3 AGRANA Beteiligungs-AG
5.1.4 Bangkok Starch Industrial Co., Ltd.
5.1.5 Thai Flour Industry Co., Ltd.
5.1.6 Jiangxi Golden Agriculture Biotech Co., Ltd.
5.1.7 WFM Wholesome Foods
5.1.8 Anhui Lianhe
5.1.9 Anhui Le Huan Tian Biotechnology
5.1.10 Herba Ingredients BV
5.1.11 Pruthvi's Foods Private Limited
5.1.12 Starch Asia
5.1.13 Ettlinger Corporation
5.1.14 A&B Ingredients, Inc.
5.1.15 Sanstar Biopolymers Ltd.
5.2 Cross Comparison Parameters (Revenue, Market Share, Product Portfolio, Regional Presence, R&D Investments, Strategic Initiatives, Production Capacity, Certifications)
5.3 Market Share Analysis
5.4 Strategic Initiatives
5.5 Mergers and Acquisitions
5.6 Investment Analysis
5.7 Venture Capital Funding
5.8 Government Grants
5.9 Private Equity Investments
6. Asia Pacific Pharmaceutical Grade Rice Starch Market Regulatory Framework
6.1 Pharmaceutical Standards and Guidelines
6.2 Compliance Requirements
6.3 Certification Processes
7. Asia Pacific Pharmaceutical Grade Rice Starch Future Market Size (USD Billion)
7.1 Future Market Size Projections
7.2 Key Factors Driving Future Market Growth
8. Asia Pacific Pharmaceutical Grade Rice Starch Future Market Segmentation
8.1 By Product Type (Value %)
8.2 By Application (Value %)
8.3 By Form (Value %)
8.4 By End-User (Value %)
8.5 By Country (Value %)
9. Asia Pacific Pharmaceutical Grade Rice Starch Market Analysts Recommendations
9.1 Total Addressable Market (TAM), Serviceable Available Market (SAM), Serviceable Obtainable Market (SOM) Analysis
9.2 Customer Cohort Analysis
9.3 Marketing Initiatives
9.4 White Space Opportunity Analysis
Disclaimer Contact UsResearch Methodology
Step 1: Identification of Key Variables
The initial phase involves constructing an ecosystem map encompassing all major stakeholders within the Asia Pacific Pharmaceutical Grade Rice Starch Market. This step is underpinned by extensive desk research, utilizing a combination of secondary and proprietary databases to gather comprehensive industry-level information. The primary objective is to identify and define the critical variables that influence market dynamics.
Step 2: Market Analysis and Construction
In this phase, we compile and analyze historical data pertaining to the Asia Pacific Pharmaceutical Grade Rice Starch Market. This includes assessing market penetration, the ratio of marketplaces to service providers, and the resultant revenue generation. Furthermore, an evaluation of service quality statistics is conducted to ensure the reliability and accuracy of the revenue estimates.
Step 3: Hypothesis Validation and Expert Consultation
Market hypotheses are developed and subsequently validated through computer-assisted telephone interviews (CATIs) with industry experts representing a diverse array of companies. These consultations provide valuable operational and financial insights directly from industry practitioners, which are instrumental in refining and corroborating the market data.
Step 4: Research Synthesis and Final Output
The final phase involves direct engagement with multiple pharmaceutical manufacturers to acquire detailed insights into product segments, sales performance, consumer preferences, and other pertinent factors. This interaction serves to verify and complement the statistics derived from the bottom-up approach, thereby ensuring a comprehensive, accurate, and validated analysis of the Asia Pacific Pharmaceutical Grade Rice Starch market.
Frequently Asked Questions
01. How big is the Asia Pacific Pharmaceutical Grade Rice Starch Market?
The Asia Pacific pharmaceutical grade rice starch market is valued at USD 500 Million, based on a five-year historical analysis. This growth is primarily driven by increasing demand for rice starch as an excipient in pharmaceutical applications due to its hypoallergenic properties and widespread usage in tablet formulations.
02. What factors are driving the Asia Pacific Pharmaceutical Grade Rice Starch Market?
The primary drivers of the Asia Pacific pharmaceutical grade rice starch market include a rise in the demand for hypoallergenic and plant-based excipients, the expansion of the pharmaceutical manufacturing sector in countries like China and India, and advancements in starch modification technology. These factors support the adoption of rice starch in various pharmaceutical applications.
03. Who are the key players in the Asia Pacific Pharmaceutical Grade Rice Starch Market?
Prominent companies in the Asia Pacific pharmaceutical grade rice starch market include Ingredion Incorporated, BENEO GmbH, AGRANA Beteiligungs-AG, Bangkok Starch Industrial Co., Ltd., and Thai Flour Industry Co., Ltd. These companies have established significant market presence through extensive distribution networks, advanced production technologies, and strong R&D capabilities.
04. What are the challenges faced by the Asia Pacific Pharmaceutical Grade Rice Starch Market?
The Asia Pacific pharmaceutical grade rice starch market faces challenges from the availability of alternative starches, higher production costs, and stringent regulatory requirements. These issues can affect production efficiency and compliance for manufacturers looking to expand in the pharmaceutical sector.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















