About the Report
Base Year 2024Oman Tuberculosis Diagnostics Market Overview
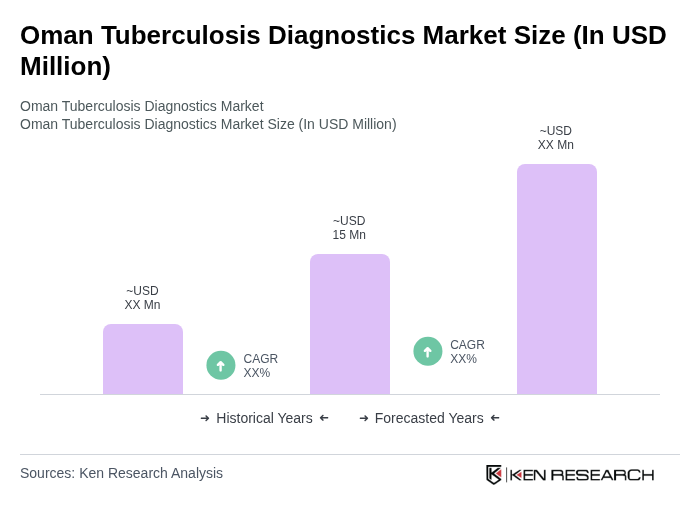
- The Oman Tuberculosis Diagnostics Market is valued at USD 15 million, based on a five-year historical analysis. This market size reflects Oman's share within the broader infectious disease diagnostics landscape and is supported by the rising burden of tuberculosis cases, estimated at around 1,200 annually. Growth is primarily driven by increasing awareness of tuberculosis (TB), the need for early diagnosis and treatment, and the adoption of advanced diagnostic solutions. The rising incidence of TB cases in Oman, coupled with government initiatives to enhance healthcare infrastructure, has significantly contributed to the market's expansion .
- Muscat and Dhofar are the dominant regions in the Oman Tuberculosis Diagnostics Market due to their advanced healthcare facilities and higher population density. The concentration of diagnostic laboratories and hospitals in these areas facilitates better access to TB diagnostic services, thereby driving market growth. Additionally, government health programs in these regions focus on TB control and prevention, further enhancing market dynamics .
- The National Tuberculosis Control Program, implemented by the Ministry of Health, Oman, under the “National Tuberculosis Control and Prevention Guidelines, 2023,” mandates free diagnostic services and treatment for TB patients. This regulation covers mandatory screening, case notification, and standardized laboratory protocols, significantly improving early detection and management of the disease across the country .
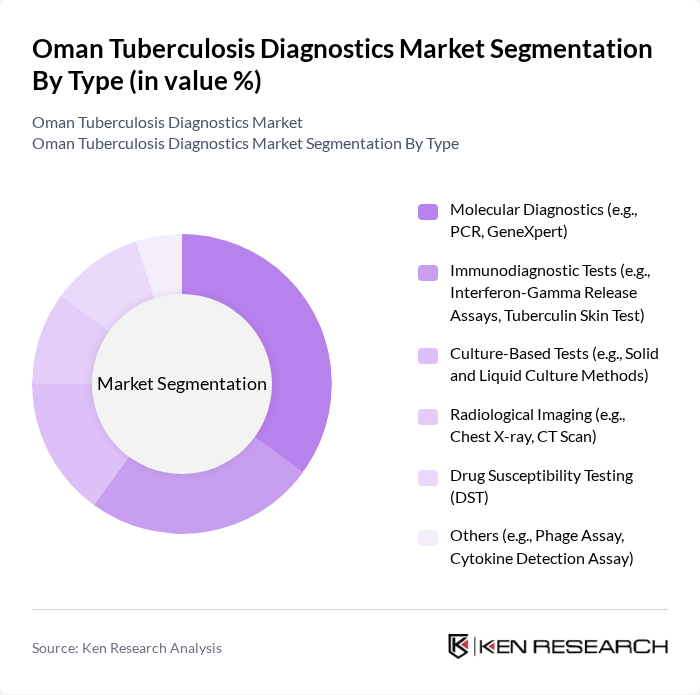
Oman Tuberculosis Diagnostics Market Segmentation
By Type:The market can be segmented into various diagnostic types, including Molecular Diagnostics, Immunodiagnostic Tests, Culture-Based Tests, Radiological Imaging, Drug Susceptibility Testing, and Others. Each of these subsegments plays a crucial role in the diagnosis and management of tuberculosis, with varying levels of adoption based on technological advancements and healthcare infrastructure. Molecular diagnostics and immunodiagnostic tests are increasingly favored due to their rapid turnaround and higher sensitivity, while culture-based methods remain the gold standard for definitive diagnosis. Radiological imaging supports clinical assessment, and drug susceptibility testing is critical for managing drug-resistant TB cases .
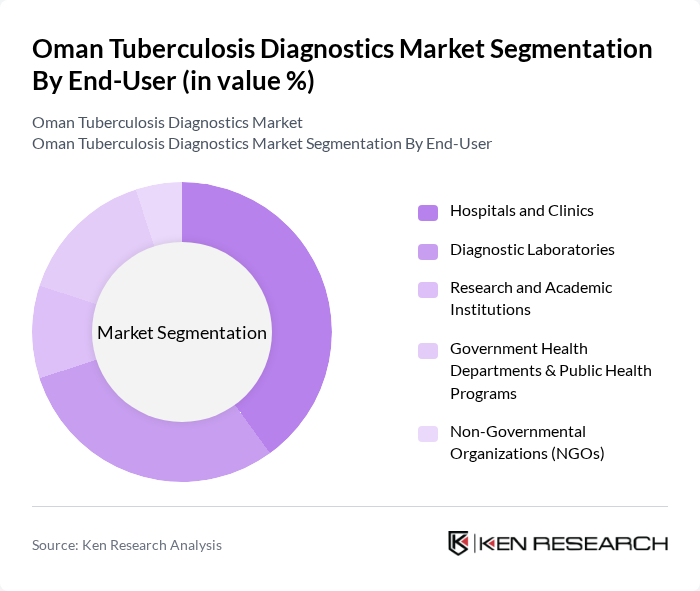
By End-User:The end-user segmentation includes Hospitals and Clinics, Diagnostic Laboratories, Research and Academic Institutions, Government Health Departments & Public Health Programs, Non-Governmental Organizations (NGOs), and Others. Each end-user category has distinct needs and contributes differently to the overall market dynamics. Diagnostic laboratories and hospitals/clinics are the primary users, reflecting their central role in TB detection and management, while government programs and NGOs support outreach and public health initiatives .
Oman Tuberculosis Diagnostics Market Competitive Landscape
The Oman Tuberculosis Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Cepheid, Roche Diagnostics, Abbott Laboratories, Hologic, Inc., Becton, Dickinson and Company, QIAGEN, Thermo Fisher Scientific, bioMérieux, Siemens Healthineers, SD Biosensor, Inc., Eiken Chemical Co., Ltd., Fujifilm Holdings Corporation, Bio-Rad Laboratories, Inc., Danaher Corporation (Beckman Coulter), Foundation for Innovative New Diagnostics (FIND) contribute to innovation, geographic expansion, and service delivery in this space.
Oman Tuberculosis Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Tuberculosis:The incidence of tuberculosis (TB) in Oman has been rising, with approximately 300 new cases reported in future, reflecting a significant public health concern. The World Health Organization (WHO) reported that Oman had a TB incidence rate of 6 per 100,000 population in recent years. This growing prevalence necessitates enhanced diagnostic capabilities, driving demand for advanced TB diagnostic tools and technologies to effectively manage and control the disease.
- Government Initiatives for TB Control:The Omani government has implemented various initiatives to combat TB, including the National Tuberculosis Control Program, which allocated OMR 1.5 million in future for TB prevention and treatment. These initiatives aim to improve screening, diagnosis, and treatment accessibility, thereby fostering a supportive environment for the growth of the tuberculosis diagnostics market. Enhanced funding and resources are expected to bolster the healthcare infrastructure, facilitating better diagnostic services.
- Advancements in Diagnostic Technologies:The introduction of innovative diagnostic technologies, such as GeneXpert and rapid molecular tests, has revolutionized TB detection in Oman. In future, the Ministry of Health reported a 30% increase in the adoption of these technologies, significantly reducing the time to diagnosis. These advancements not only improve accuracy but also enhance the overall efficiency of TB management, driving market growth as healthcare providers seek to implement the latest diagnostic solutions.
Market Challenges
- Limited Healthcare Infrastructure:Oman faces challenges related to its healthcare infrastructure, particularly in rural areas where access to diagnostic facilities is limited. According to the World Bank, only 60% of healthcare facilities are equipped with modern diagnostic tools as of future. This disparity hampers timely TB diagnosis and treatment, posing a significant challenge to the effective management of tuberculosis and limiting market growth potential in underserved regions.
- High Costs of Advanced Diagnostic Tools:The financial burden associated with advanced TB diagnostic technologies remains a significant challenge. For instance, the cost of molecular diagnostic tests can exceed OMR 50 per test, which is prohibitive for many healthcare facilities, especially in the public sector. This high cost limits the widespread adoption of these technologies, creating barriers to effective TB diagnosis and treatment, and ultimately affecting market growth.
Oman Tuberculosis Diagnostics Market Future Outlook
The future of the Oman tuberculosis diagnostics market appears promising, driven by ongoing government initiatives and technological advancements. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the rise of telemedicine will facilitate remote diagnostics, particularly in rural areas. As public-private partnerships expand, the market is likely to witness increased investment in innovative diagnostic solutions, ultimately improving TB management and control across the nation.
Market Opportunities
- Expansion of Public-Private Partnerships:Collaborations between public and private sectors can enhance resource allocation for TB diagnostics. In future, the government initiated partnerships with private firms, aiming to improve diagnostic accessibility and affordability. This approach is expected to foster innovation and increase the availability of advanced diagnostic tools, ultimately benefiting TB management in Oman.
- Development of Cost-Effective Diagnostic Solutions:There is a growing demand for affordable diagnostic solutions in Oman, particularly in low-resource settings. The development of cost-effective tests, such as rapid antigen detection kits, can significantly improve TB diagnosis. In future, initiatives to create these solutions are anticipated to gain momentum, addressing the financial barriers currently faced by healthcare providers and enhancing TB control efforts.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Molecular Diagnostics (e.g., PCR, GeneXpert) Immunodiagnostic Tests (e.g., Interferon-Gamma Release Assays, Tuberculin Skin Test) Culture-Based Tests (e.g., Solid and Liquid Culture Methods) Radiological Imaging (e.g., Chest X-ray, CT Scan) Drug Susceptibility Testing (DST) Others (e.g., Phage Assay, Cytokine Detection Assay) |
| By End-User | Hospitals and Clinics Diagnostic Laboratories Research and Academic Institutions Government Health Departments & Public Health Programs Non-Governmental Organizations (NGOs) Others |
| By Application | Screening (Active and Latent TB) Diagnosis (Pulmonary and Extrapulmonary TB) Monitoring Treatment Response Detection of Drug Resistance Others |
| By Distribution Channel | Direct Sales (Manufacturers to End-Users) Distributors/Wholesalers Online Sales Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Others |
| By Technology | PCR Technology ELISA Technology X-ray Technology GeneXpert Technology Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman)
Manufacturers and Producers of Diagnostic Equipment
Distributors and Retailers of Medical Supplies
Healthcare Providers and Hospitals
Non-Governmental Organizations (NGOs) focused on Health
Pharmaceutical Companies involved in TB Treatment
Public Health Agencies (e.g., World Health Organization - WHO)
Players Mentioned in the Report:
Cepheid
Roche Diagnostics
Abbott Laboratories
Hologic, Inc.
Becton, Dickinson and Company
QIAGEN
Thermo Fisher Scientific
bioMerieux
Siemens Healthineers
SD Biosensor, Inc.
Eiken Chemical Co., Ltd.
Fujifilm Holdings Corporation
Bio-Rad Laboratories, Inc.
Danaher Corporation (Beckman Coulter)
Foundation for Innovative New Diagnostics (FIND)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Tuberculosis Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Tuberculosis Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Tuberculosis Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of tuberculosis in Oman
3.1.2 Government initiatives for TB control
3.1.3 Advancements in diagnostic technologies
3.1.4 Rising awareness and screening programs
3.2 Market Challenges
3.2.1 Limited healthcare infrastructure
3.2.2 High costs of advanced diagnostic tools
3.2.3 Shortage of trained healthcare professionals
3.2.4 Stigma associated with tuberculosis
3.3 Market Opportunities
3.3.1 Expansion of public-private partnerships
3.3.2 Development of cost-effective diagnostic solutions
3.3.3 Increased funding from international organizations
3.3.4 Growth in telemedicine and remote diagnostics
3.4 Market Trends
3.4.1 Shift towards molecular diagnostics
3.4.2 Integration of AI in diagnostic processes
3.4.3 Focus on personalized medicine
3.4.4 Rise in mobile health applications
3.5 Government Regulation
3.5.1 National TB control programs
3.5.2 Regulatory approvals for new diagnostic tests
3.5.3 Guidelines for TB screening and treatment
3.5.4 Incentives for research and development in diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Tuberculosis Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Tuberculosis Diagnostics Market Segmentation
8.1 By Type
8.1.1 Molecular Diagnostics (e.g., PCR, GeneXpert)
8.1.2 Immunodiagnostic Tests (e.g., Interferon-Gamma Release Assays, Tuberculin Skin Test)
8.1.3 Culture-Based Tests (e.g., Solid and Liquid Culture Methods)
8.1.4 Radiological Imaging (e.g., Chest X-ray, CT Scan)
8.1.5 Drug Susceptibility Testing (DST)
8.1.6 Others (e.g., Phage Assay, Cytokine Detection Assay)
8.2 By End-User
8.2.1 Hospitals and Clinics
8.2.2 Diagnostic Laboratories
8.2.3 Research and Academic Institutions
8.2.4 Government Health Departments & Public Health Programs
8.2.5 Non-Governmental Organizations (NGOs)
8.2.6 Others
8.3 By Application
8.3.1 Screening (Active and Latent TB)
8.3.2 Diagnosis (Pulmonary and Extrapulmonary TB)
8.3.3 Monitoring Treatment Response
8.3.4 Detection of Drug Resistance
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales (Manufacturers to End-Users)
8.4.2 Distributors/Wholesalers
8.4.3 Online Sales
8.4.4 Others
8.5 By Region
8.5.1 Muscat
8.5.2 Dhofar
8.5.3 Al Batinah
8.5.4 Al Dakhiliyah
8.5.5 Others
8.6 By Technology
8.6.1 PCR Technology
8.6.2 ELISA Technology
8.6.3 X-ray Technology
8.6.4 GeneXpert Technology
8.6.5 Others
8.7 By Policy Support
8.7.1 Government Subsidies
8.7.2 Tax Incentives
8.7.3 Research Grants
8.7.4 Others
9. Oman Tuberculosis Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue in Oman (USD Million)
9.2.4 Market Share in Oman (%)
9.2.5 Product Portfolio Breadth (Number of TB Diagnostic Products)
9.2.6 Regulatory Approvals (MOH/WHO Prequalification)
9.2.7 Distribution Network Coverage (Number of Cities/Regions Served)
9.2.8 Local Partnerships/Presence (e.g., with Omani distributors or health authorities)
9.2.9 R&D Investment in TB Diagnostics (% of Revenue)
9.2.10 Time-to-Result (Average Diagnostic Turnaround Time)
9.2.11 Customer Satisfaction Score
9.2.12 After-Sales Service Availability
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Cepheid
9.5.2 Roche Diagnostics
9.5.3 Abbott Laboratories
9.5.4 Hologic, Inc.
9.5.5 Becton, Dickinson and Company
9.5.6 QIAGEN
9.5.7 Thermo Fisher Scientific
9.5.8 bioMérieux
9.5.9 Siemens Healthineers
9.5.10 SD Biosensor, Inc.
9.5.11 Eiken Chemical Co., Ltd.
9.5.12 Fujifilm Holdings Corporation
9.5.13 Bio-Rad Laboratories, Inc.
9.5.14 Danaher Corporation (Beckman Coulter)
9.5.15 Foundation for Innovative New Diagnostics (FIND)
10. Oman Tuberculosis Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Social Development
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Diagnostic Equipment Procurement
10.2.3 Training and Development Expenditure
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Labs
10.3.3 Government Health Departments
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Efficiency Gains
10.5.3 Patient Outcomes Improvement
10.5.4 Others
11. Oman Tuberculosis Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of health ministry reports and tuberculosis prevalence data in Oman
- Review of published studies and articles on tuberculosis diagnostics technologies
- Examination of market reports from international health organizations and NGOs
Primary Research
- Interviews with healthcare professionals specializing in infectious diseases
- Surveys with laboratory managers and diagnostic equipment suppliers
- Field interviews with public health officials and policy makers
Validation & Triangulation
- Cross-validation of findings through multiple data sources including government and private sector
- Triangulation of market insights from healthcare professionals and industry reports
- Sanity checks through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Oman related to tuberculosis
- Breakdown of diagnostics market by technology type (e.g., molecular, imaging)
- Incorporation of government health initiatives and funding for tuberculosis control
Bottom-up Modeling
- Volume estimates based on the number of tuberculosis cases reported annually
- Cost analysis of diagnostic tests and equipment used in healthcare facilities
- Calculation of market size based on unit sales and pricing strategies
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth and healthcare access
- Scenario modeling based on potential changes in healthcare policy and funding
- Baseline, optimistic, and pessimistic projections for the market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Sector Insights | 100 | Public Health Officials, Epidemiologists |
| Diagnostic Laboratories | 60 | Laboratory Managers, Technicians |
| Healthcare Providers | 80 | Doctors, Nurses, Healthcare Administrators |
| Government Health Policy | 50 | Health Policy Makers, Program Coordinators |
| NGO and International Organizations | 40 | NGO Representatives, Health Program Managers |
Frequently Asked Questions
What is the current value of the Oman Tuberculosis Diagnostics Market?
The Oman Tuberculosis Diagnostics Market is valued at approximately USD 15 million, reflecting the country's share in the broader infectious disease diagnostics landscape, driven by the rising incidence of tuberculosis cases and government healthcare initiatives.