About the Report
Base Year 2024Philippines Spinal Fusion Device Market Overview
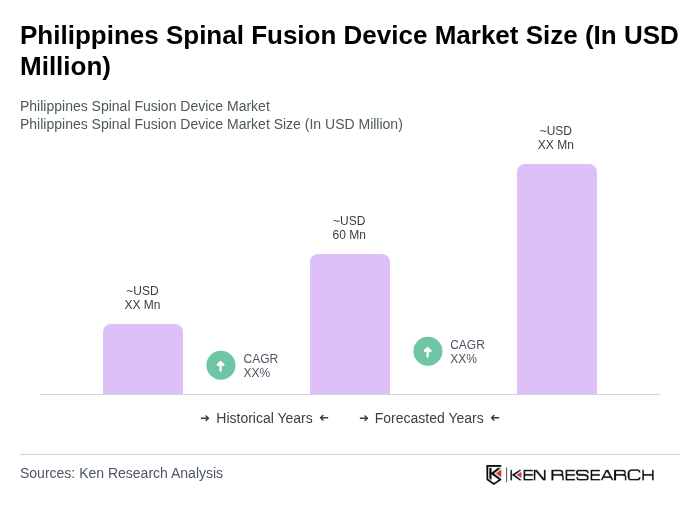
- The Philippines Spinal Fusion Device Market is valued at USD 60 million, based on a five-year historical analysis. This market is experiencing growth driven by the increasing prevalence of spinal disorders, rapid adoption of minimally invasive surgical techniques, and advancements in implant technology such as 3D-printed interbody cages and biologically active implants. The aging population and rising healthcare expenditure further support market expansion, with hospitals and specialized clinics increasingly performing spinal fusion procedures to address degenerative disc diseases and spinal instability.
- Metro Manila, Cebu, and Davao remain the dominant regions in the Philippines Spinal Fusion Device Market. Metro Manila, as the capital, has the highest concentration of tertiary care hospitals, advanced medical infrastructure, and specialized spinal surgeons. Cebu and Davao serve as major healthcare hubs for the Visayas and Mindanao regions, respectively, benefiting from investments in hospital modernization and the establishment of specialty clinics focused on orthopedics and neurosurgery.
- The Universal Health Care Act (Republic Act No. 11223), issued by the Department of Health in 2019 and implemented in subsequent years, mandates comprehensive healthcare coverage for all Filipino citizens. This regulation includes operational provisions for spinal health, ensuring that medically necessary spinal fusion procedures are covered under the Philippine Health Insurance Corporation (PhilHealth) benefits package. Hospitals must comply with standardized reimbursement protocols, and patients gain improved access and affordability for spinal fusion interventions.
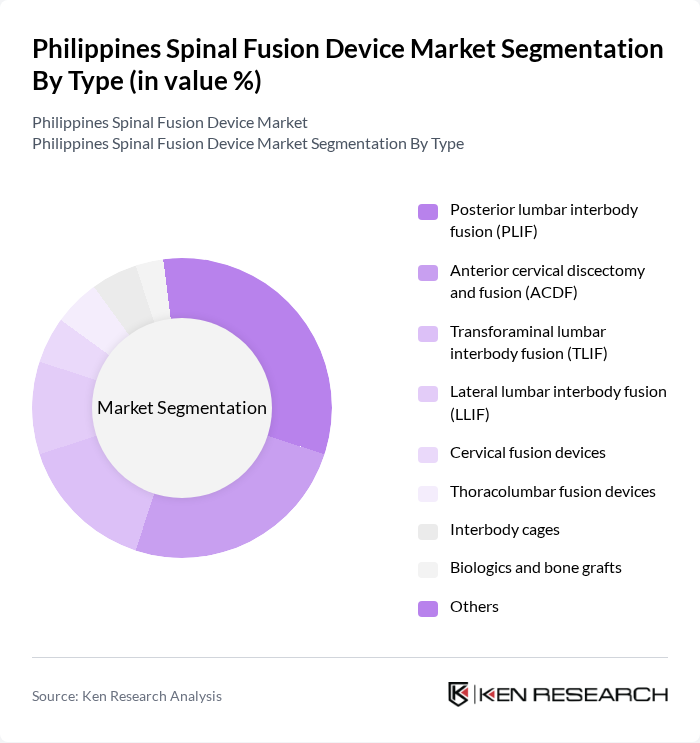
Philippines Spinal Fusion Device Market Segmentation
By Type:The spinal fusion device market in the Philippines is segmented into posterior lumbar interbody fusion (PLIF), anterior cervical discectomy and fusion (ACDF), transforaminal lumbar interbody fusion (TLIF), lateral lumbar interbody fusion (LLIF), cervical fusion devices, thoracolumbar fusion devices, interbody cages, biologics and bone grafts, and others. PLIF and ACDF are the most commonly performed techniques in the country, owing to their proven effectiveness in treating degenerative disc disease and cervical spine disorders. The adoption of interbody cages and biologics is increasing due to their role in enhancing fusion rates and reducing recovery times.
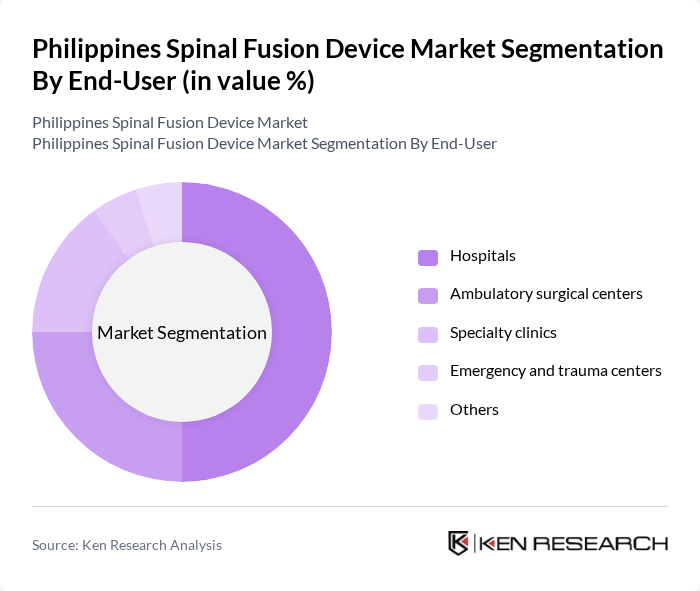
By End-User:The end-user segmentation comprises hospitals, ambulatory surgical centers, specialty clinics, emergency and trauma centers, and others. Hospitals are the leading end-users, attributed to their capacity for complex spinal surgeries and post-operative care. Ambulatory surgical centers are gaining market share due to their efficiency in performing minimally invasive spinal fusion procedures, while specialty clinics and trauma centers provide focused care for spinal injuries and disorders.
Philippines Spinal Fusion Device Market Competitive Landscape
The Philippines Spinal Fusion Device Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, DePuy Synthes (Johnson & Johnson), Stryker Corporation, NuVasive, Inc., Zimmer Biomet Holdings, Inc., Globus Medical, Inc., B. Braun Melsungen AG, Orthofix Medical Inc., K2M Group Holdings, Inc., SpineWave, Inc., RTI Surgical, Inc., Aesculap Implant Systems (B. Braun), Alphatec Holdings, Inc., SeaSpine Holdings Corporation, Xtant Medical Holdings, Inc., Biometrix Medical Devices, Inc., Philippine Medical Supplies, Inc., MedAsia Medical Products Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Philippines Spinal Fusion Device Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Spinal Disorders:The Philippines has seen a significant rise in spinal disorders, with approximately 1.2 million cases reported annually. This increase is attributed to lifestyle changes and an aging population. According to the Department of Health, spinal disorders account for nearly 30% of all musculoskeletal conditions, driving demand for spinal fusion devices. The growing awareness of treatment options further propels market growth, as patients seek advanced surgical interventions to alleviate pain and improve quality of life.
- Advancements in Surgical Techniques:The adoption of innovative surgical techniques, such as minimally invasive surgery, has transformed spinal fusion procedures in the Philippines. In future, it is estimated that over 60% of spinal surgeries will utilize these advanced methods, reducing recovery times and improving patient outcomes. The integration of technology, including imaging and navigation systems, enhances surgical precision, attracting more patients to seek spinal fusion treatments, thus boosting device demand significantly.
- Rising Geriatric Population:The geriatric population in the Philippines is projected to reach 10 million in future, representing a 20% increase from 2020. This demographic shift is crucial, as older adults are more susceptible to spinal disorders due to degenerative changes. The Philippine Statistics Authority indicates that spinal fusion surgeries among seniors are expected to rise, creating a substantial market for spinal fusion devices. Increased healthcare access for this age group further supports market expansion.
Market Challenges
- High Cost of Spinal Fusion Procedures:The average cost of spinal fusion surgery in the Philippines ranges from PHP 200,000 to PHP 500,000, which poses a significant barrier for many patients. With a per capita income of approximately PHP 100,000 in future, affordability remains a critical issue. Many patients rely on government subsidies or health insurance, which may not cover the full costs, limiting access to necessary surgical interventions and hindering market growth.
- Limited Access to Advanced Healthcare Facilities:Access to advanced healthcare facilities is uneven across the Philippines, with urban areas having significantly better resources than rural regions. According to the World Health Organization, only 30% of hospitals in rural areas are equipped for complex spinal surgeries. This disparity restricts patient access to spinal fusion procedures, resulting in delayed treatments and lower overall market penetration for spinal fusion devices in underserved regions.
Philippines Spinal Fusion Device Market Future Outlook
The future of the spinal fusion device market in the Philippines appears promising, driven by technological advancements and increasing healthcare investments. As the government focuses on enhancing healthcare infrastructure, more facilities will be equipped to perform complex spinal surgeries. Additionally, the rising awareness of spinal health among the population is expected to lead to higher demand for surgical interventions, further propelling market growth. The integration of innovative technologies will also play a crucial role in shaping the future landscape of spinal fusion procedures.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Philippine government plans to invest PHP 50 billion in healthcare infrastructure in future, aiming to improve access to advanced medical facilities. This investment will facilitate the establishment of specialized spinal surgery centers, increasing the availability of spinal fusion procedures and devices, ultimately enhancing patient outcomes and market growth.
- Technological Innovations in Spinal Devices:The introduction of next-generation spinal devices, such as bioresorbable implants and smart spinal systems, presents significant market opportunities. With an estimated 15% increase in R&D spending by medical device companies in future, these innovations will likely attract more patients seeking effective and less invasive treatment options, driving demand for advanced spinal fusion devices.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Posterior lumbar interbody fusion (PLIF) Anterior cervical discectomy and fusion (ACDF) Transforaminal lumbar interbody fusion (TLIF) Lateral lumbar interbody fusion (LLIF) Cervical fusion devices Thoracolumbar fusion devices Interbody cages Biologics and bone grafts Others |
| By End-User | Hospitals Ambulatory surgical centers Specialty clinics Emergency and trauma centers Others |
| By Region | Luzon Visayas Mindanao |
| By Technology | Traditional spinal fusion techniques Minimally invasive spinal fusion technologies Robotic-assisted fusion systems Biologics and bone grafts Motion-preservation systems Others |
| By Application | Degenerative disc disease Spinal tumors Spinal deformities (e.g., scoliosis) Trauma-related spinal injuries Others |
| By Investment Source | Private investments Government funding International aid Others |
| By Policy Support | Government subsidies Tax incentives for medical device manufacturers Research grants for spinal health Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration (FDA) Philippines)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations (e.g., Philippine Medical Device Industry Association)
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
DePuy Synthes (Johnson & Johnson)
Stryker Corporation
NuVasive, Inc.
Zimmer Biomet Holdings, Inc.
Globus Medical, Inc.
B. Braun Melsungen AG
Orthofix Medical Inc.
K2M Group Holdings, Inc.
SpineWave, Inc.
RTI Surgical, Inc.
Aesculap Implant Systems (B. Braun)
Alphatec Holdings, Inc.
SeaSpine Holdings Corporation
Xtant Medical Holdings, Inc.
Biometrix Medical Devices, Inc.
Philippine Medical Supplies, Inc.
MedAsia Medical Products Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Philippines Spinal Fusion Device Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Philippines Spinal Fusion Device Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Philippines Spinal Fusion Device Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of spinal disorders
3.1.2 Advancements in surgical techniques
3.1.3 Rising geriatric population
3.1.4 Growing healthcare expenditure
3.2 Market Challenges
3.2.1 High cost of spinal fusion procedures
3.2.2 Limited access to advanced healthcare facilities
3.2.3 Stringent regulatory requirements
3.2.4 Shortage of skilled healthcare professionals
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing awareness about spinal health
3.3.3 Technological innovations in spinal devices
3.3.4 Collaborations with international medical device companies
3.4 Market Trends
3.4.1 Shift towards minimally invasive surgeries
3.4.2 Adoption of robotic-assisted surgical systems
3.4.3 Growth in outpatient spinal surgeries
3.4.4 Rising demand for biologics in spinal fusion
3.5 Government Regulation
3.5.1 Implementation of stricter quality control measures
3.5.2 Introduction of reimbursement policies for spinal surgeries
3.5.3 Regulation of medical device approvals
3.5.4 Monitoring of post-market device performance
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Philippines Spinal Fusion Device Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Philippines Spinal Fusion Device Market Segmentation
8.1 By Type
8.1.1 Posterior lumbar interbody fusion (PLIF)
8.1.2 Anterior cervical discectomy and fusion (ACDF)
8.1.3 Transforaminal lumbar interbody fusion (TLIF)
8.1.4 Lateral lumbar interbody fusion (LLIF)
8.1.5 Cervical fusion devices
8.1.6 Thoracolumbar fusion devices
8.1.7 Interbody cages
8.1.8 Biologics and bone grafts
8.1.9 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory surgical centers
8.2.3 Specialty clinics
8.2.4 Emergency and trauma centers
8.2.5 Others
8.3 By Region
8.3.1 Luzon
8.3.2 Visayas
8.3.3 Mindanao
8.4 By Technology
8.4.1 Traditional spinal fusion techniques
8.4.2 Minimally invasive spinal fusion technologies
8.4.3 Robotic-assisted fusion systems
8.4.4 Biologics and bone grafts
8.4.5 Motion-preservation systems
8.4.6 Others
8.5 By Application
8.5.1 Degenerative disc disease
8.5.2 Spinal tumors
8.5.3 Spinal deformities (e.g., scoliosis)
8.5.4 Trauma-related spinal injuries
8.5.5 Others
8.6 By Investment Source
8.6.1 Private investments
8.6.2 Government funding
8.6.3 International aid
8.6.4 Others
8.7 By Policy Support
8.7.1 Government subsidies
8.7.2 Tax incentives for medical device manufacturers
8.7.3 Research grants for spinal health
8.7.4 Others
9. Philippines Spinal Fusion Device Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Philippines segment)
9.2.4 Market Share (Philippines spinal fusion device market)
9.2.5 Product Portfolio Breadth (number of spinal fusion SKUs)
9.2.6 Regulatory Approval Status (FDA/Philippines FDA)
9.2.7 Distribution Network Coverage (number of hospitals/clinics served)
9.2.8 Local Manufacturing/Import Ratio
9.2.9 Pricing Strategy (premium, value, reimbursement eligibility)
9.2.10 Brand Recognition (among Filipino surgeons/hospitals)
9.2.11 Customer Satisfaction Score (Philippines market)
9.2.12 Innovation Rate (new product launches in Philippines)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 DePuy Synthes (Johnson & Johnson)
9.5.3 Stryker Corporation
9.5.4 NuVasive, Inc.
9.5.5 Zimmer Biomet Holdings, Inc.
9.5.6 Globus Medical, Inc.
9.5.7 B. Braun Melsungen AG
9.5.8 Orthofix Medical Inc.
9.5.9 K2M Group Holdings, Inc.
9.5.10 SpineWave, Inc.
9.5.11 RTI Surgical, Inc.
9.5.12 Aesculap Implant Systems (B. Braun)
9.5.13 Alphatec Holdings, Inc.
9.5.14 SeaSpine Holdings Corporation
9.5.15 Xtant Medical Holdings, Inc.
9.5.16 Biometrix Medical Devices, Inc.
9.5.17 Philippine Medical Supplies, Inc.
9.5.18 MedAsia Medical Products Corporation
10. Philippines Spinal Fusion Device Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government procurement policies
10.1.2 Budget allocation for spinal health
10.1.3 Collaboration with private sectors
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare facilities
10.2.2 Funding for medical equipment
10.2.3 Partnerships with healthcare providers
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to quality healthcare
10.3.2 Affordability of spinal procedures
10.3.3 Availability of skilled surgeons
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of spinal fusion benefits
10.4.2 Training for healthcare professionals
10.4.3 Patient education initiatives
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of surgical outcomes
10.5.2 Long-term patient follow-up
10.5.3 Expansion into new patient demographics
10.5.4 Others
11. Philippines Spinal Fusion Device Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape assessment
1.6 Key partnerships identification
1.7 Risk assessment and mitigation strategies
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Marketing channels selection
2.5 Messaging and communication strategies
2.6 Performance metrics establishment
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales strategies
3.5 Partnership with healthcare providers
4. Channel & Pricing Gaps
4.1 Underserved routes analysis
4.2 Pricing bands evaluation
4.3 Competitor pricing comparison
4.4 Customer willingness to pay assessment
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry barriers assessment
6. Customer Relationship
6.1 Loyalty programs design
6.2 After-sales service strategies
6.3 Customer feedback mechanisms
6.4 Community engagement initiatives
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains development
7.3 Customer-centric product offerings
7.4 Competitive differentiation strategies
8. Key Activities
8.1 Regulatory compliance strategies
8.2 Branding initiatives
8.3 Distribution setup planning
8.4 Training and development programs
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band strategies
9.1.3 Packaging innovations
9.2 Export Entry Strategy
9.2.1 Target countries analysis
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model evaluation
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships analysis
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
14. Potential Partner List
14.1 Distributors identification
14.2 Joint Ventures opportunities
14.3 Acquisition targets analysis
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone planning
15.2.2 Activity tracking mechanisms
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from medical device associations in the Philippines
- Review of government publications on healthcare spending and spinal surgery statistics
- Examination of academic journals and clinical studies related to spinal fusion technologies
Primary Research
- Interviews with orthopedic surgeons specializing in spinal procedures
- Surveys with hospital procurement managers regarding spinal fusion device purchasing
- Focus groups with patients who have undergone spinal fusion surgeries
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews
- Triangulation of data from clinical outcomes and market trends
- Sanity checks through feedback from a panel of healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in the Philippines allocated to spinal surgeries
- Analysis of demographic data to identify target patient populations for spinal fusion
- Incorporation of trends in surgical procedures and device adoption rates
Bottom-up Modeling
- Collection of sales data from leading spinal fusion device manufacturers operating in the Philippines
- Estimation of average selling prices for various spinal fusion devices
- Volume estimates based on surgical procedure rates and device usage per procedure
Forecasting & Scenario Analysis
- Multi-factor analysis considering population aging and increasing incidence of spinal disorders
- Scenario modeling based on potential regulatory changes and healthcare policy impacts
- Baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Orthopedic Surgeons | 45 | Spine Specialists, General Orthopedic Surgeons |
| Hospital Procurement Managers | 38 | Supply Chain Managers, Medical Device Buyers |
| Healthcare Policy Makers | 25 | Health Department Officials, Regulatory Affairs Managers |
| Patients with Spinal Fusion Experience | 32 | Post-operative Patients, Patient Advocacy Group Members |
| Medical Device Distributors | 28 | Sales Representatives, Distribution Managers |
Frequently Asked Questions
What is the current value of the Philippines Spinal Fusion Device Market?
The Philippines Spinal Fusion Device Market is valued at approximately USD 60 million, reflecting a five-year historical analysis. This market is growing due to the rising prevalence of spinal disorders and advancements in surgical techniques and implant technology.