About the Report
Base Year 2024Listen to the audio summary
Saudi Arabia Active Pharmaceutical Ingredients Api Market Overview
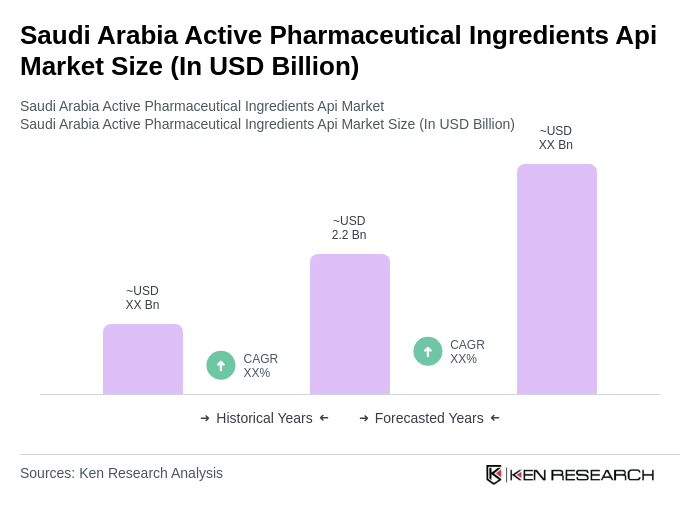
- The Saudi Arabia Active Pharmaceutical Ingredients (API) market is valued at USD 2.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for pharmaceuticals, the rising prevalence of chronic diseases such as diabetes and cardiovascular conditions, and a growing focus on healthcare infrastructure. The need for innovative drug formulations and the adoption of personalized medicine are further propelling the market, making it a critical component of the healthcare sector .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure, presence of major pharmaceutical companies, and strategic government initiatives aimed at enhancing local production capabilities. These cities serve as hubs for research and development, attracting investments and fostering collaborations between public and private sectors .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented new regulations to enhance the quality and safety of APIs produced in the country. This includes stringent compliance requirements for Good Manufacturing Practices (GMP) and regular inspections of manufacturing facilities. The initiative aims to ensure that locally produced APIs meet international standards, thereby boosting the competitiveness of Saudi Arabia's pharmaceutical industry .
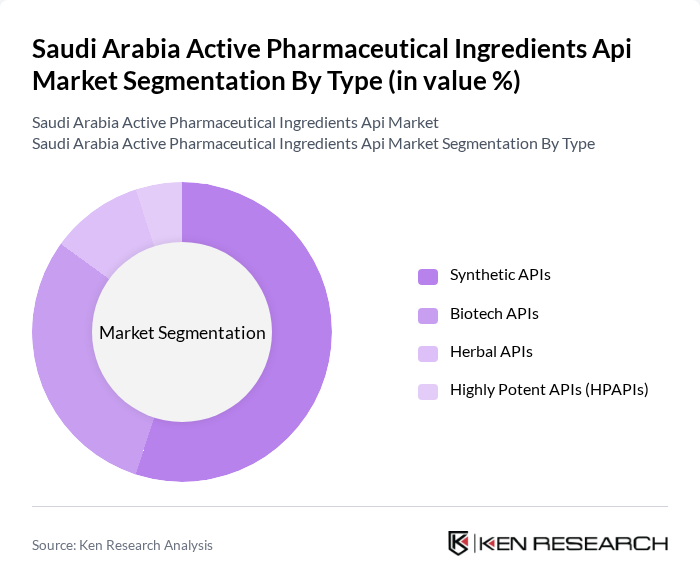
Saudi Arabia Active Pharmaceutical Ingredients Api Market Segmentation
By Type:In the Saudi Arabian market, the segment of synthetic APIs is currently dominating due to their widespread application in various therapeutic areas, including cardiovascular diseases and oncology. The increasing prevalence of chronic diseases has led to a surge in demand for synthetic drugs, which are often more cost-effective and easier to produce than their biotech counterparts. Advancements in synthetic chemistry and process optimization have enabled manufacturers to develop more effective and targeted therapies, further driving the growth of this sub-segment .
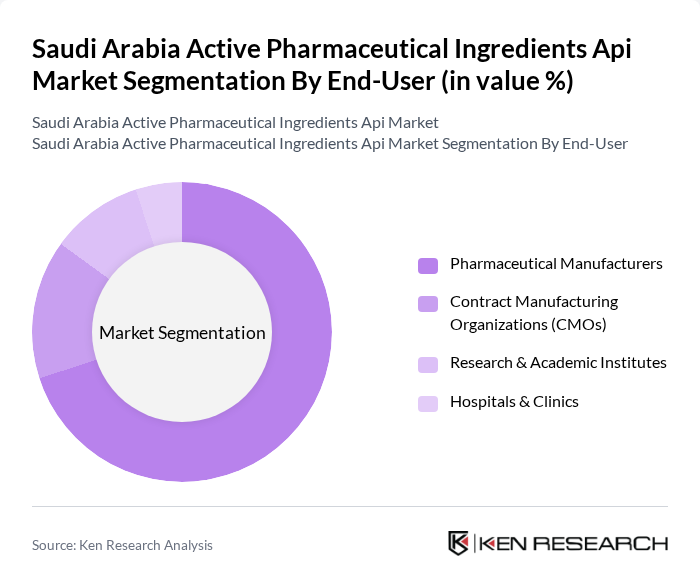
By End-User:Pharmaceutical manufacturers represent the largest end-user segment in the Saudi Arabian API market. This dominance is attributed to the increasing number of local pharmaceutical companies expanding their production capabilities to meet the growing demand for generic and branded medications. The trend towards vertical integration, where manufacturers seek to control their supply chains by producing their own APIs, is further solidifying the position of pharmaceutical manufacturers in the market .
Saudi Arabia Active Pharmaceutical Ingredients Api Market Competitive Landscape
The Saudi Arabia Active Pharmaceutical Ingredients Api Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals Manufacturing Company, Jamjoom Pharma, Riyadh Pharma (Saudi Chemical Company Holding), Sudair Pharma Company, Gulf Pharmaceutical Industries (Julphar), Hikma Pharmaceuticals, AJA Pharma, SPIMACO Addwaieh, Al-Dawaa Pharmacies, Al Nahdi Medical Company, Pfizer Saudi Limited, Novartis Saudi Arabia, Sanofi Saudi Arabia, Bayer Saudi Arabia contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Active Pharmaceutical Ingredients Api Market Industry Analysis
Growth Drivers
- Increasing Demand for Generic Drugs:The Saudi Arabian market is witnessing a significant rise in the demand for generic drugs, driven by a growing population and increasing prevalence of chronic diseases. In future, the generic drug market is projected to reach approximately SAR 5.5 billion, reflecting a 10% increase from the previous year. This surge is attributed to the government's efforts to enhance healthcare accessibility and affordability, encouraging the use of cost-effective alternatives to branded medications.
- Government Initiatives to Boost Local Manufacturing:The Saudi government is actively promoting local manufacturing of active pharmaceutical ingredients (APIs) through initiatives such as the National Industrial Development and Logistics Program. In future, the government aims to increase local API production by 30%, reducing dependency on imports, which currently account for over 70% of the market. This strategic focus is expected to enhance the country's self-sufficiency in pharmaceuticals and create job opportunities.
- Rising Healthcare Expenditure:Saudi Arabia's healthcare expenditure is projected to reach SAR 210 billion in future, reflecting a 5% increase from the previous year. This rise is driven by the government's commitment to improving healthcare services and infrastructure as part of Vision 2030. Increased spending on healthcare is expected to boost the demand for APIs, particularly in therapeutic areas such as oncology and cardiology, where innovative treatments are essential.
Market Challenges
- Stringent Regulatory Compliance:The Saudi pharmaceutical industry faces significant challenges due to stringent regulatory compliance requirements imposed by the Saudi Food and Drug Authority (SFDA). In future, companies must navigate complex approval processes that can take up to 18 months, delaying product launches. This regulatory environment can hinder the agility of local manufacturers, making it difficult to compete with international players who may have more streamlined processes.
- High Production Costs:High production costs remain a critical challenge for the Saudi API market, with average manufacturing costs estimated at SAR 1,300 per kilogram in future. Factors contributing to these costs include limited access to raw materials and advanced manufacturing technologies. As a result, local manufacturers struggle to maintain competitive pricing against imported APIs, which can be produced at lower costs in countries with established supply chains.
Saudi Arabia Active Pharmaceutical Ingredients Api Market Future Outlook
The future of the Saudi Arabia active pharmaceutical ingredients market appears promising, driven by increasing investments in local manufacturing and a growing focus on biopharmaceuticals. As the government continues to implement Vision 2030, initiatives aimed at enhancing healthcare infrastructure and promoting research and development will likely foster innovation. Additionally, the rising interest in personalized medicine and sustainable manufacturing practices will shape the market landscape, creating opportunities for growth and collaboration among industry stakeholders.
Market Opportunities
- Growth in Biopharmaceuticals:The biopharmaceutical sector in Saudi Arabia is expected to expand significantly, with investments projected to reach SAR 3.5 billion in future. This growth presents opportunities for local API manufacturers to develop specialized ingredients for biologics, catering to the increasing demand for advanced therapies in oncology and autoimmune diseases.
- Strategic Partnerships and Collaborations:Collaborations between local manufacturers and international pharmaceutical companies are anticipated to increase, facilitating knowledge transfer and technology sharing. In future, at least five major partnerships are expected to be established, enhancing local capabilities in API production and expanding market reach, particularly in the GCC region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Synthetic APIs Biotech APIs Herbal APIs Highly Potent APIs (HPAPIs) |
| By End-User | Pharmaceutical Manufacturers Contract Manufacturing Organizations (CMOs) Research & Academic Institutes Hospitals & Clinics |
| By Therapeutic Area | Cardiovascular Diseases Oncology (Cancer) Infectious Diseases Diabetes Central Nervous System Disorders Others |
| By Source | Domestic Production Imports |
| By Distribution Channel | Direct Sales Distributors/Wholesalers Online Platforms |
| By Formulation | Solid Liquid Semi-solid |
| By Regulatory Compliance | SFDA-GMP Compliant WHO-GMP/Internationally Compliant Non-GMP Compliant |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Pharmaceutical Companies
Distributors and Wholesalers
Biotechnology Firms
Industry Associations (e.g., Saudi Pharmaceutical Society)
Financial Institutions
Players Mentioned in the Report:
Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO)
Tabuk Pharmaceuticals Manufacturing Company
Jamjoom Pharma
Riyadh Pharma (Saudi Chemical Company Holding)
Sudair Pharma Company
Gulf Pharmaceutical Industries (Julphar)
Hikma Pharmaceuticals
AJA Pharma
SPIMACO Addwaieh
Al-Dawaa Pharmacies
Al Nahdi Medical Company
Pfizer Saudi Limited
Novartis Saudi Arabia
Sanofi Saudi Arabia
Bayer Saudi Arabia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Active Pharmaceutical Ingredients Api Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Active Pharmaceutical Ingredients Api Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Active Pharmaceutical Ingredients Api Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Generic Drugs
3.1.2 Government Initiatives to Boost Local Manufacturing
3.1.3 Rising Healthcare Expenditure
3.1.4 Expansion of Pharmaceutical R&D Activities
3.2 Market Challenges
3.2.1 Stringent Regulatory Compliance
3.2.2 High Production Costs
3.2.3 Limited Access to Raw Materials
3.2.4 Competition from International Players
3.3 Market Opportunities
3.3.1 Growth in Biopharmaceuticals
3.3.2 Strategic Partnerships and Collaborations
3.3.3 Technological Advancements in Manufacturing
3.3.4 Export Potential to GCC Countries
3.4 Market Trends
3.4.1 Shift Towards Sustainable Manufacturing Practices
3.4.2 Increasing Focus on Quality Assurance
3.4.3 Adoption of Digital Technologies in Production
3.4.4 Growing Interest in Personalized Medicine
3.5 Government Regulation
3.5.1 Implementation of Saudi Vision 2030
3.5.2 New Drug Approval Processes
3.5.3 Intellectual Property Rights Enforcement
3.5.4 Pricing and Reimbursement Policies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Active Pharmaceutical Ingredients Api Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Active Pharmaceutical Ingredients Api Market Segmentation
8.1 By Type
8.1.1 Synthetic APIs
8.1.2 Biotech APIs
8.1.3 Herbal APIs
8.1.4 Highly Potent APIs (HPAPIs)
8.2 By End-User
8.2.1 Pharmaceutical Manufacturers
8.2.2 Contract Manufacturing Organizations (CMOs)
8.2.3 Research & Academic Institutes
8.2.4 Hospitals & Clinics
8.3 By Therapeutic Area
8.3.1 Cardiovascular Diseases
8.3.2 Oncology (Cancer)
8.3.3 Infectious Diseases
8.3.4 Diabetes
8.3.5 Central Nervous System Disorders
8.3.6 Others
8.4 By Source
8.4.1 Domestic Production
8.4.2 Imports
8.5 By Distribution Channel
8.5.1 Direct Sales
8.5.2 Distributors/Wholesalers
8.5.3 Online Platforms
8.6 By Formulation
8.6.1 Solid
8.6.2 Liquid
8.6.3 Semi-solid
8.7 By Regulatory Compliance
8.7.1 SFDA-GMP Compliant
8.7.2 WHO-GMP/Internationally Compliant
8.7.3 Non-GMP Compliant
9. Saudi Arabia Active Pharmaceutical Ingredients Api Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD Million, Latest Year)
9.2.4 Revenue Growth Rate (CAGR %)
9.2.5 Market Share (%)
9.2.6 Product Portfolio Breadth (Number of APIs/Drug Classes)
9.2.7 Regulatory Approvals (SFDA, US FDA, EMA, etc.)
9.2.8 R&D Investment as % of Revenue
9.2.9 Local Manufacturing Capacity (Tons/Year)
9.2.10 Supply Chain Reliability (Lead Time, Fill Rate)
9.2.11 Export Share (%)
9.2.12 Strategic Partnerships/Collaborations (Count)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO)
9.5.2 Tabuk Pharmaceuticals Manufacturing Company
9.5.3 Jamjoom Pharma
9.5.4 Riyadh Pharma (Saudi Chemical Company Holding)
9.5.5 Sudair Pharma Company
9.5.6 Gulf Pharmaceutical Industries (Julphar)
9.5.7 Hikma Pharmaceuticals
9.5.8 AJA Pharma
9.5.9 SPIMACO Addwaieh
9.5.10 Al-Dawaa Pharmacies
9.5.11 Al Nahdi Medical Company
9.5.12 Pfizer Saudi Limited
9.5.13 Novartis Saudi Arabia
9.5.14 Sanofi Saudi Arabia
9.5.15 Bayer Saudi Arabia
10. Saudi Arabia Active Pharmaceutical Ingredients Api Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Commerce
10.1.3 Ministry of Investment
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Infrastructure Investments
10.2.2 Energy Consumption Patterns
10.2.3 Budget Allocations for R&D
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Management
10.3.2 Quality Assurance Challenges
10.3.3 Regulatory Compliance Issues
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of New Technologies
10.4.2 Training and Skill Development
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion into New Therapeutic Areas
10.5.3 User Feedback and Adaptation
10.5.4 Others
11. Saudi Arabia Active Pharmaceutical Ingredients Api Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from the Saudi Food and Drug Authority (SFDA)
- Review of market studies published by pharmaceutical associations in Saudi Arabia
- Examination of trade publications and journals focusing on active pharmaceutical ingredients (APIs)
Primary Research
- Interviews with regulatory experts from the SFDA and Ministry of Health
- Surveys with pharmaceutical manufacturers and distributors operating in Saudi Arabia
- Field interviews with R&D heads at local pharmaceutical companies
Validation & Triangulation
- Cross-validation of data through multiple industry sources and expert opinions
- Triangulation of findings from primary interviews and secondary data sources
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size in Saudi Arabia as a baseline
- Segmentation of the API market by therapeutic categories and application areas
- Incorporation of government healthcare spending and initiatives impacting API demand
Bottom-up Modeling
- Collection of sales data from leading API manufacturers in the region
- Estimation of production capacities and output levels of local firms
- Analysis of pricing trends and cost structures for various API segments
Forecasting & Scenario Analysis
- Development of growth projections based on historical data and market trends
- Scenario modeling considering regulatory changes and market entry of new players
- Baseline, optimistic, and pessimistic forecasts through 2030 based on economic indicators
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Production Managers, Quality Assurance Heads |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Distributors and Wholesalers | 60 | Supply Chain Managers, Sales Directors |
| Healthcare Providers | 50 | Pharmacists, Hospital Procurement Officers |
| Research Institutions | 40 | Research Scientists, Academic Professors |
Frequently Asked Questions
What is the current value of the Saudi Arabia Active Pharmaceutical Ingredients (API) market?
The Saudi Arabia Active Pharmaceutical Ingredients market is valued at approximately USD 2.2 billion, driven by increasing pharmaceutical demand, chronic disease prevalence, and healthcare infrastructure improvements.