About the Report
Base Year 2024Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Overview
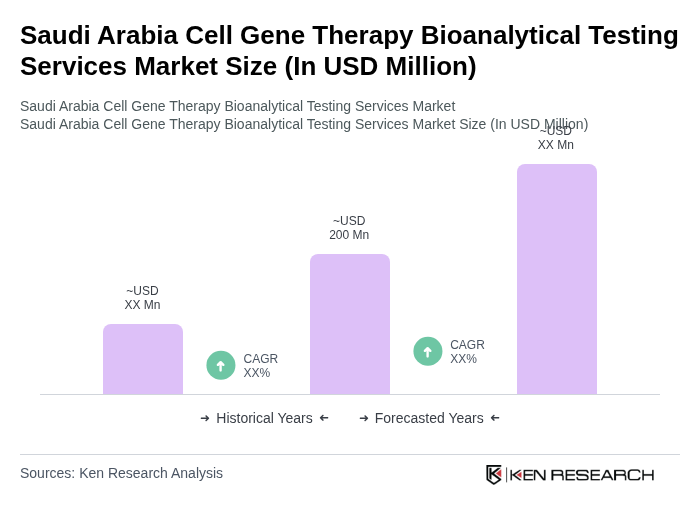
- The Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market is valued at USD 200 million, based on a five-year historical analysis. This growth is primarily driven by advancements in biotechnology, increasing prevalence of genetic disorders, and rising investments in healthcare infrastructure. The market is also supported by a growing demand for personalized medicine and innovative therapies that require specialized bioanalytical testing services. The Kingdom's Vision 2030 initiative emphasizes healthcare innovation and biotechnology advancement, positioning Saudi Arabia as a regional biotech leader with localized manufacturing capabilities and infrastructure expansion.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure, presence of leading hospitals, and research institutions. Riyadh, being the capital, is a hub for medical research and biotechnology firms, with the Northern and Central regions serving as the primary centers for advanced healthcare institutions and clinical trials. Jeddah and Dammam are significant for their strategic locations and access to international markets, facilitating collaborations and innovations in cell and gene therapy.
- The Saudi Food and Drug Authority (SFDA) has established comprehensive regulatory frameworks to govern gene therapy products, including streamlined approval processes that enhance patient access to innovative therapies while ensuring safety and efficacy. The SFDA's guidelines focus on rigorous testing and compliance standards, fostering a conducive environment for the growth of the bioanalytical testing services market. These regulatory measures support the Kingdom's commitment to becoming a global biotech hub with accelerated trial activation and patient access capabilities.
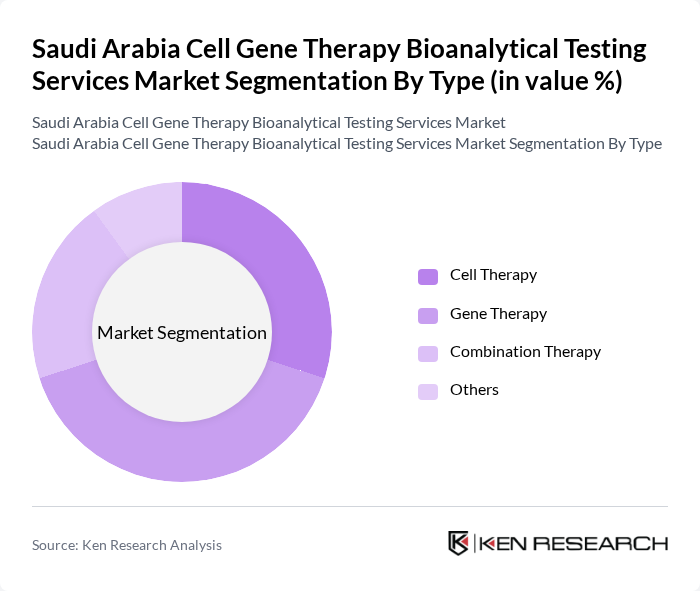
Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Segmentation
By Type:The market is segmented into various types, including Cell Therapy, Gene Therapy, Combination Therapy, and Others. Among these, Gene Therapy is currently the leading sub-segment due to its increasing application in treating genetic disorders and cancers. The rise in research and development activities, coupled with the growing number of clinical trials, has significantly boosted the demand for gene therapy services. Cell Therapy also holds a substantial share, driven by advancements in regenerative medicine and the increasing prevalence of chronic diseases. Gene Augmentation has emerged as the fastest-growing segment, driven by the higher-than-average prevalence of inherited genetic conditions such as sickle cell disease and thalassemia in Saudi Arabia.
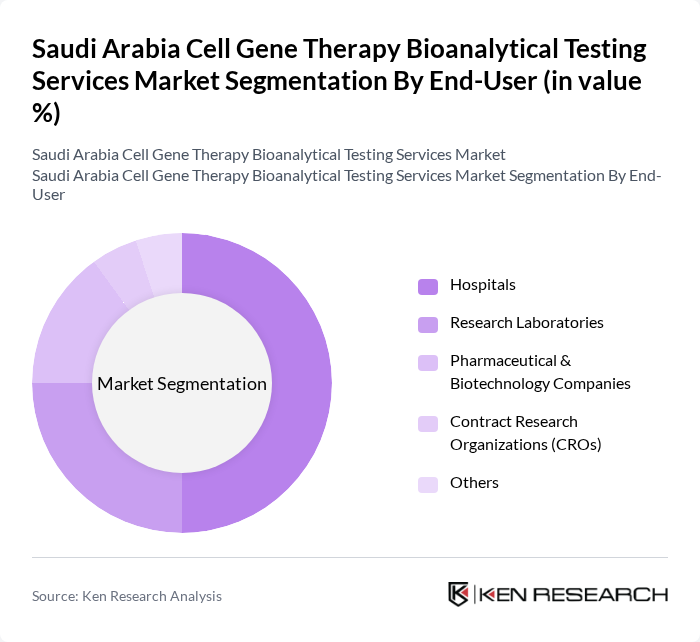
By End-User:The end-user segmentation includes Hospitals, Research Laboratories, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Others. Hospitals are the dominant end-user segment, primarily due to the increasing number of patients requiring advanced therapies and the growing adoption of personalized medicine. Research Laboratories also play a crucial role, as they are involved in extensive research and development activities, driving the demand for bioanalytical testing services. The clinical segment represents the largest revenue-generating category, with commercial applications registering the fastest growth during the forecast period.
Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Competitive Landscape
The Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as King Faisal Specialist Hospital & Research Centre, SaudiVax Ltd., Riyadh Valley Company, Tabuk Pharmaceuticals Manufacturing Company, National Guard Health Affairs (NGHA), Saudi Biotechnology Manufacturing Co. (SBM), Gulf Pharmaceutical Industries (Julphar), Aljomaih and Shell Lubricating Oil Company (JOSLOC) – Life Sciences Division, Saudi Food and Drug Authority (SFDA), Al-Hokair Group – Healthcare Division, BioSaudi, Saudi Biotechnology Company (SBC), Alfaisal University – College of Medicine, King Abdulaziz University – Center of Innovation in Personalized Medicine, Research Products Development Company (RPDC) contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders:The prevalence of genetic disorders in Saudi Arabia is rising, with approximately 1 in 4 children affected by genetic conditions, according to the Saudi Ministry of Health. This alarming statistic drives demand for advanced bioanalytical testing services, as healthcare providers seek effective gene therapies. The increasing number of diagnosed cases, estimated to reach over 1 million in future, necessitates innovative testing solutions to support personalized treatment approaches and improve patient outcomes.
- Rising Investment in Biotechnology Research:Saudi Arabia's biotechnology sector is experiencing significant growth, with investments projected to exceed SAR 1.5 billion (approximately USD 400 million) in future. This surge in funding is primarily directed towards research and development in gene therapy and bioanalytical testing services. The government’s Vision 2030 initiative emphasizes biotechnology as a key area for economic diversification, fostering an environment conducive to innovation and attracting global partnerships to enhance local capabilities.
- Government Support for Healthcare Innovation:The Saudi government is actively promoting healthcare innovation through various initiatives, including the establishment of the Saudi Health Investment Strategy, which allocates SAR 2 billion (around USD 533 million) for healthcare advancements. This support includes funding for bioanalytical testing services, aimed at enhancing the country's capabilities in gene therapy. Such initiatives are expected to create a robust ecosystem that encourages research, development, and commercialization of innovative healthcare solutions.
Market Challenges
- High Cost of Bioanalytical Testing Services:The cost of bioanalytical testing services in Saudi Arabia can be prohibitively high, with average testing prices ranging from SAR 10,000 to SAR 50,000 (USD 2,667 to USD 13,333) per patient. This financial barrier limits access to essential testing for many patients, particularly in underserved regions. As healthcare providers strive to offer comprehensive gene therapy solutions, addressing these cost challenges is crucial for expanding service accessibility and improving patient outcomes.
- Limited Skilled Workforce in Advanced Therapies:The shortage of skilled professionals in the field of gene therapy and bioanalytical testing poses a significant challenge for the market. Currently, there are only about 1,500 qualified specialists in Saudi Arabia, which is insufficient to meet the growing demand for advanced therapies. This skills gap hampers the development and implementation of innovative testing services, necessitating targeted training programs and partnerships with educational institutions to cultivate a competent workforce.
Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Future Outlook
The future of the Saudi Arabia cell gene therapy bioanalytical testing services market appears promising, driven by increasing investments in biotechnology and supportive government policies. As the prevalence of genetic disorders continues to rise, the demand for innovative testing solutions will likely expand. Furthermore, advancements in automation and artificial intelligence are expected to enhance testing efficiency and accuracy, paving the way for more personalized medicine approaches. Collaborative efforts between academia and industry will also play a crucial role in fostering innovation and addressing workforce challenges.
Market Opportunities
- Expansion of Clinical Trials in Gene Therapy:The number of clinical trials for gene therapies in Saudi Arabia is projected to increase significantly, with over 50 new trials expected in future. This expansion presents opportunities for bioanalytical testing service providers to engage in partnerships with research institutions and pharmaceutical companies, enhancing their service offerings and contributing to the development of groundbreaking therapies.
- Collaborations with International Biotech Firms:Collaborations with international biotech firms are on the rise, with at least 10 new partnerships anticipated in future. These collaborations can facilitate knowledge transfer, access to advanced technologies, and shared resources, ultimately strengthening the local bioanalytical testing landscape and enhancing the capabilities of Saudi firms in gene therapy development.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Cell Therapy Gene Therapy Combination Therapy Others |
| By End-User | Hospitals Research Laboratories Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
| By Application | Oncology Rare Genetic Disorders Cardiovascular Diseases Immunological Disorders Others |
| By Testing Method | PCR-based Testing Sequencing-based Testing (NGS, Sanger, etc.) ELISA & Immunoassays Flow Cytometry Mass Spectrometry Chromatography Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Service Type | Preclinical Testing Clinical Testing Post-Market Surveillance Regulatory Compliance Testing Others |
| By Technology | Flow Cytometry Mass Spectrometry Chromatography Digital PCR Next-Generation Sequencing (NGS) Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Biotechnology and Pharmaceutical Companies
Contract Research Organizations (CROs)
Healthcare Providers and Hospitals
Biomanufacturers and Cell Therapy Developers
Clinical Research Institutions
Insurance Companies and Payers
Players Mentioned in the Report:
King Faisal Specialist Hospital & Research Centre
SaudiVax Ltd.
Riyadh Valley Company
Tabuk Pharmaceuticals Manufacturing Company
National Guard Health Affairs (NGHA)
Saudi Biotechnology Manufacturing Co. (SBM)
Gulf Pharmaceutical Industries (Julphar)
Aljomaih and Shell Lubricating Oil Company (JOSLOC) Life Sciences Division
Saudi Food and Drug Authority (SFDA)
Al-Hokair Group Healthcare Division
BioSaudi
Saudi Biotechnology Company (SBC)
Alfaisal University College of Medicine
King Abdulaziz University Center of Innovation in Personalized Medicine
Research Products Development Company (RPDC)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of genetic disorders
3.1.2 Rising investment in biotechnology research
3.1.3 Government support for healthcare innovation
3.1.4 Growing demand for personalized medicine
3.2 Market Challenges
3.2.1 High cost of bioanalytical testing services
3.2.2 Limited skilled workforce in advanced therapies
3.2.3 Regulatory hurdles and compliance issues
3.2.4 Market competition from established players
3.3 Market Opportunities
3.3.1 Expansion of clinical trials in gene therapy
3.3.2 Collaborations with international biotech firms
3.3.3 Development of innovative testing technologies
3.3.4 Increasing awareness and education on gene therapies
3.4 Market Trends
3.4.1 Shift towards automation in bioanalytical testing
3.4.2 Integration of AI and machine learning in analysis
3.4.3 Focus on patient-centric testing approaches
3.4.4 Rise in partnerships between academia and industry
3.5 Government Regulation
3.5.1 Implementation of stringent quality control standards
3.5.2 Establishment of ethical guidelines for gene therapy
3.5.3 Supportive policies for biotech startups
3.5.4 Regulatory frameworks for clinical trial approvals
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Segmentation
8.1 By Type
8.1.1 Cell Therapy
8.1.2 Gene Therapy
8.1.3 Combination Therapy
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Laboratories
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Contract Research Organizations (CROs)
8.2.5 Others
8.3 By Application
8.3.1 Oncology
8.3.2 Rare Genetic Disorders
8.3.3 Cardiovascular Diseases
8.3.4 Immunological Disorders
8.3.5 Others
8.4 By Testing Method
8.4.1 PCR-based Testing
8.4.2 Sequencing-based Testing (NGS, Sanger, etc.)
8.4.3 ELISA & Immunoassays
8.4.4 Flow Cytometry
8.4.5 Mass Spectrometry
8.4.6 Chromatography
8.4.7 Others
8.5 By Region
8.5.1 Central Region
8.5.2 Eastern Region
8.5.3 Western Region
8.5.4 Southern Region
8.6 By Service Type
8.6.1 Preclinical Testing
8.6.2 Clinical Testing
8.6.3 Post-Market Surveillance
8.6.4 Regulatory Compliance Testing
8.6.5 Others
8.7 By Technology
8.7.1 Flow Cytometry
8.7.2 Mass Spectrometry
8.7.3 Chromatography
8.7.4 Digital PCR
8.7.5 Next-Generation Sequencing (NGS)
8.7.6 Others
9. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Saudi Arabia Cell & Gene Therapy Bioanalytical Testing Services)
9.2.4 Market Penetration Rate (Share of Saudi Arabia market by segment)
9.2.5 Number of Bioanalytical Tests Conducted Annually
9.2.6 Turnaround Time for Testing (Average TAT in days)
9.2.7 Service Portfolio Breadth (Number of distinct test types offered)
9.2.8 R&D Investment as % of Revenue
9.2.9 Regulatory Compliance Track Record (e.g., SFDA/ISO accreditations)
9.2.10 Customer Satisfaction Score (Net Promoter Score or equivalent)
9.2.11 Strategic Partnerships (Number of active collaborations with pharma/biotech/academia)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 King Faisal Specialist Hospital & Research Centre
9.5.2 SaudiVax Ltd.
9.5.3 Riyadh Valley Company
9.5.4 Tabuk Pharmaceuticals Manufacturing Company
9.5.5 National Guard Health Affairs (NGHA)
9.5.6 Saudi Biotechnology Manufacturing Co. (SBM)
9.5.7 Gulf Pharmaceutical Industries (Julphar)
9.5.8 Aljomaih and Shell Lubricating Oil Company (JOSLOC) – Life Sciences Division
9.5.9 Saudi Food and Drug Authority (SFDA)
9.5.10 Al-Hokair Group – Healthcare Division
9.5.11 BioSaudi
9.5.12 Saudi Biotechnology Company (SBC)
9.5.13 Alfaisal University – College of Medicine
9.5.14 King Abdulaziz University – Center of Innovation in Personalized Medicine
9.5.15 Research Products Development Company (RPDC)
10. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Industry and Mineral Resources
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Infrastructure
10.2.2 Funding for Research and Development
10.2.3 Expenditure on Advanced Medical Equipment
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Gene Therapy Benefits
10.4.2 Training and Education Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Expansion into New Therapeutic Areas
10.5.4 Others
11. Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the Saudi Food and Drug Authority (SFDA)
- Review of scientific publications and clinical trial registries related to cell gene therapy
- Examination of market trends and forecasts from healthcare market research firms
Primary Research
- Interviews with key opinion leaders in the field of gene therapy and biotechnology
- Surveys with biopharmaceutical companies involved in cell gene therapy development
- Focus groups with healthcare professionals and researchers in Saudi Arabia
Validation & Triangulation
- Cross-validation of findings through multiple data sources including academic journals and industry reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and investment in biotechnology
- Segmentation of the market by therapeutic areas such as oncology, genetic disorders, and rare diseases
- Incorporation of government initiatives and funding for gene therapy research and development
Bottom-up Modeling
- Collection of data on the number of ongoing clinical trials and their respective phases
- Estimation of service pricing based on cost structures of bioanalytical testing services
- Volume estimates derived from the number of patients eligible for cell gene therapy treatments
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as technological advancements and regulatory changes
- Scenario modeling based on varying levels of market adoption and healthcare policy impacts
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Gene Therapy Services | 100 | Oncologists, Clinical Researchers |
| Genetic Disorder Treatment Providers | 70 | Pediatricians, Genetic Counselors |
| Regulatory Affairs in Gene Therapy | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Biotechnology Research Institutions | 60 | Research Scientists, Lab Managers |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
Frequently Asked Questions
What is the current value of the Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market?
The Saudi Arabia Cell Gene Therapy Bioanalytical Testing Services Market is valued at approximately USD 200 million, reflecting significant growth driven by advancements in biotechnology and increasing healthcare investments.