About the Report
Base Year 2024United States Toxicity Testing Outsourcing Market Overview
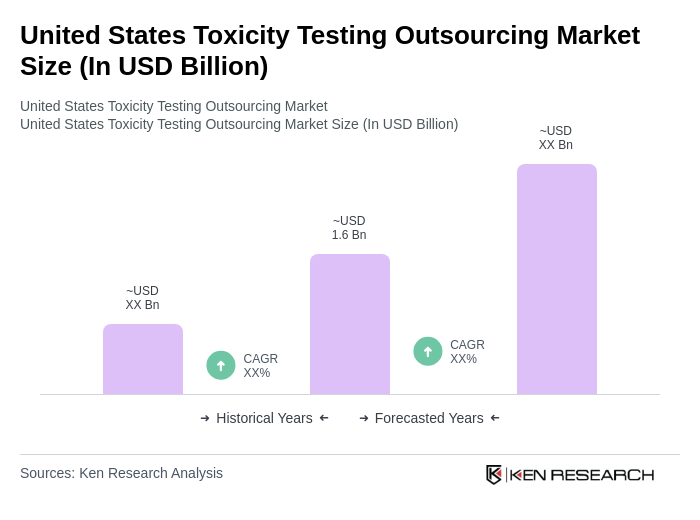
- The United States Toxicity Testing Outsourcing Market is valued at USD 1.6 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for safety assessments in pharmaceuticals, chemicals, and consumer products, alongside stringent regulatory requirements for toxicological evaluations. The rise in awareness regarding environmental safety and the need for compliance with health regulations have further propelled the market's expansion.
- Key players in this market are concentrated in major cities such as New York, San Francisco, and Boston. These locations are hubs for pharmaceutical and biotechnology firms, which are the primary clients for toxicity testing services. The presence of leading research institutions and a robust network of service providers in these cities enhances their dominance in the market. The U.S. toxicity testing outsourcing market represents approximately 43% of the global market, reflecting North America's dominant position in toxicity testing services.
- The Toxic Substances Control Act (TSCA), originally enacted in 1976 and significantly reformed through the Frank R. Lautenberg Chemical Safety for the 21st Century Act of 2016, mandates comprehensive risk assessments for new and existing chemicals. This regulation emphasizes the need for rigorous toxicity testing, thereby increasing the demand for outsourcing these services to specialized firms. Additionally, in December 2024, the FDA announced proposed standard testing methods for cosmetic products containing talc to identify the presence of carcinogenic asbestos, requiring manufacturers to analyze samples using advanced analytical techniques such as polarized light and transmission electron microscopy, further driving demand for specialized toxicity testing services.
United States Toxicity Testing Outsourcing Market Segmentation
By Type:The market is segmented into various types of toxicity testing services, including In Vivo Testing, In Vitro Testing, Computational Toxicology, Ecotoxicology Testing, Inhalation Toxicology Testing, and Others. Each of these sub-segments plays a crucial role in assessing the safety and efficacy of products across different industries.
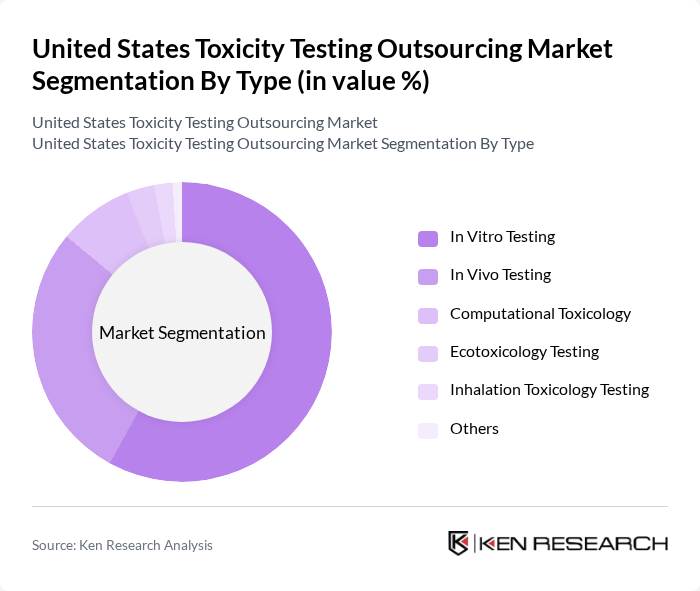
In Vitro Testing currently dominates the market due to its cost-effectiveness, rapid turnaround time, and alignment with regulatory preferences for reducing animal testing. This method enables organizations to conduct preliminary hazard assessments and screen high-risk compounds efficiently. The method is increasingly preferred for initial safety screening in the pharmaceutical and cosmetic industries. In addition, computational toxicology and in silico predictive tools, such as the EPA's CompTox Chemicals Dashboard, are accelerating the transition to alternative testing models by enabling early identification of high-risk compounds and reducing reliance on traditional animal testing methods.
By End-User:The market is segmented based on end-users, including Pharmaceutical Companies, Biotechnology Firms, Chemical Manufacturers, Academic & Research Institutes, Medical Device Companies, and Others. Each of these segments has unique requirements and contributes to the overall demand for toxicity testing services.
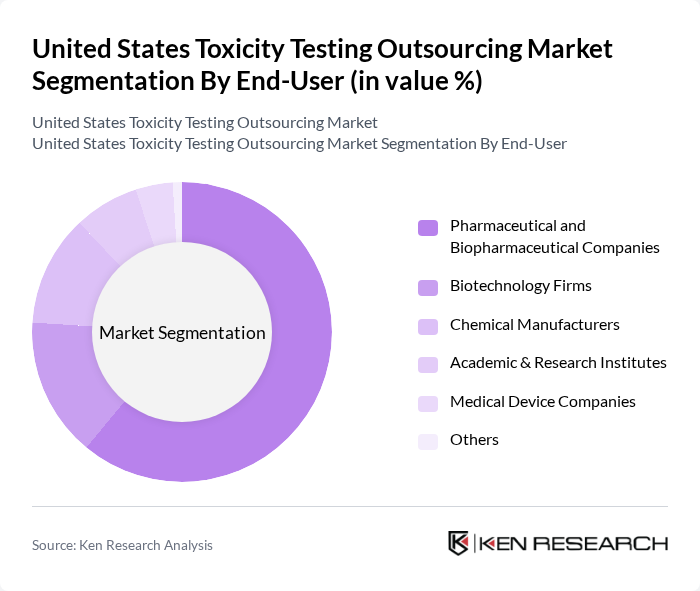
Pharmaceutical and biopharmaceutical companies represent the largest end-user segment in the toxicity testing outsourcing market. This dominance is attributed to rigorous testing requirements mandated by regulatory authorities for drug approval processes and the increasing complexity of therapeutics including biologics, biosimilars, genetically targeted therapies, and stem cell therapies. The U.S. Food and Drug Administration approved 53 new drug applications and biologics license applications in 2024, representing a 15% increase from the previous year, driving heightened demand for comprehensive toxicity testing services. The increasing investment in drug development and the need for sophisticated analytical testing to ensure safety, purity, and efficacy further drive the demand for outsourced toxicity testing services among pharmaceutical firms.
United States Toxicity Testing Outsourcing Market Competitive Landscape
The United States Toxicity Testing Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Charles River Laboratories International, Inc., Labcorp Drug Development (formerly Covance Inc.), Eurofins Scientific SE, SGS S.A., Intertek Group plc, WuXi AppTec Co., Ltd., Medpace Holdings, Inc., Envigo RMS LLC, Toxikon Corporation (now part of Labcorp), BioReliance (a Merck KGaA company), Q2 Solutions (an IQVIA & Quest Diagnostics joint venture), ICON plc, Thermo Fisher Scientific Inc., Accelera S.r.l., Catalent, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
United States Toxicity Testing Outsourcing Market Industry Analysis
Growth Drivers
- Increasing Regulatory Requirements:The United States has seen a significant rise in regulatory requirements for toxicity testing, with the Environmental Protection Agency (EPA) enforcing over 1,000 new chemical assessments annually. This surge is driven by the need for enhanced safety protocols, particularly in pharmaceuticals and consumer products. The FDA's recent guidelines mandate comprehensive toxicity evaluations, leading to an estimated increase in testing demand by approximately $600 million in future, reflecting the industry's commitment to consumer safety.
- Rising Demand for Safety Assessments:The demand for safety assessments in the United States is projected to reach $3 billion in future, driven by heightened public awareness and regulatory scrutiny. Industries such as cosmetics and food additives are particularly affected, with companies investing heavily in third-party testing services to ensure compliance. This trend is further supported by the increasing number of product recalls, which have risen by 20% over the past year, emphasizing the need for rigorous safety evaluations.
- Advancements in Testing Technologies:Technological advancements in toxicity testing, such as high-throughput screening and in vitro methods, are revolutionizing the industry. In future, the market for innovative testing technologies is expected to exceed $1.5 billion, driven by the adoption of AI and machine learning. These technologies not only enhance the accuracy of assessments but also reduce the time and cost associated with traditional testing methods, making outsourcing more attractive to companies seeking efficiency.
Market Challenges
- High Competition Among Service Providers:The toxicity testing outsourcing market is characterized by intense competition, with over 250 service providers operating in the United States. This saturation leads to price wars, which can undermine profit margins. In future, the average revenue per service provider is projected to decline by 15%, as companies struggle to differentiate their offerings in a crowded marketplace, impacting overall market stability.
- Variability in Regulatory Standards:The inconsistency in regulatory standards across states poses a significant challenge for toxicity testing providers. For instance, California's Proposition 65 requires stricter testing protocols compared to federal guidelines, creating confusion and compliance issues. This variability can lead to increased operational costs, with companies potentially facing fines exceeding $1.5 million for non-compliance, thereby complicating the outsourcing landscape and deterring investment.
United States Toxicity Testing Outsourcing Market Future Outlook
The future of the toxicity testing outsourcing market in the United States appears promising, driven by ongoing advancements in technology and increasing regulatory scrutiny. As companies prioritize safety and compliance, the integration of AI and machine learning into testing processes is expected to enhance efficiency and accuracy. Furthermore, the growing emphasis on environmental sustainability will likely lead to the development of greener testing methods, positioning the industry for significant growth and innovation in the coming years.
Market Opportunities
- Expansion into Emerging Markets:The potential for expansion into emerging markets is substantial, with countries like India and Brazil increasing their regulatory frameworks. This shift presents an opportunity for U.S. firms to leverage their expertise in toxicity testing, potentially increasing revenue streams by an estimated $400 million in future as these markets seek reliable testing services.
- Development of Innovative Testing Methods:The development of innovative testing methods, particularly those that reduce animal testing, is gaining traction. With the global market for alternative testing methods projected to reach $1.5 billion in future, U.S. companies that invest in these technologies can capture a significant share of this growing segment, enhancing their competitive edge and market presence.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | In Vivo Testing In Vitro Testing Computational Toxicology Ecotoxicology Testing Inhalation Toxicology Testing Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Chemical Manufacturers Academic & Research Institutes Medical Device Companies Others |
| By Application | Drug Development Environmental Safety Product Safety Testing Regulatory Compliance Food & Beverage Safety Cosmetics & Personal Care Testing Others |
| By Service Type | Full-Service Testing Specialized Testing Services Consulting Services Data Management & Reporting Others |
| By Region | Northeast Midwest South West Others |
| By Pricing Model | Fixed Pricing Hourly Rates Subscription-Based Outcome-Based Pricing Others |
| By Client Type | Large Enterprises SMEs Government Agencies Nonprofit Organizations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Environmental Protection Agency, Food and Drug Administration)
Pharmaceutical Companies
Chemical Manufacturers
Biotechnology Firms
Contract Research Organizations
Health and Safety Regulatory Agencies
Insurance Companies
Players Mentioned in the Report:
Charles River Laboratories International, Inc.
Labcorp Drug Development (formerly Covance Inc.)
Eurofins Scientific SE
SGS S.A.
Intertek Group plc
WuXi AppTec Co., Ltd.
Medpace Holdings, Inc.
Envigo RMS LLC
Toxikon Corporation (now part of Labcorp)
BioReliance (a Merck KGaA company)
Q2 Solutions (an IQVIA & Quest Diagnostics joint venture)
ICON plc
Thermo Fisher Scientific Inc.
Accelera S.r.l.
Catalent, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. United States Toxicity Testing Outsourcing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 United States Toxicity Testing Outsourcing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. United States Toxicity Testing Outsourcing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Regulatory Requirements
3.1.2 Rising Demand for Safety Assessments
3.1.3 Advancements in Testing Technologies
3.1.4 Cost-Effectiveness of Outsourcing
3.2 Market Challenges
3.2.1 High Competition Among Service Providers
3.2.2 Variability in Regulatory Standards
3.2.3 Limited Awareness Among Small Enterprises
3.2.4 Data Privacy Concerns
3.3 Market Opportunities
3.3.1 Expansion into Emerging Markets
3.3.2 Development of Innovative Testing Methods
3.3.3 Strategic Partnerships with Biotech Firms
3.3.4 Increased Investment in R&D
3.4 Market Trends
3.4.1 Shift Towards In Vitro Testing
3.4.2 Integration of AI in Toxicity Testing
3.4.3 Growing Focus on Environmental Sustainability
3.4.4 Rise of Personalized Medicine
3.5 Government Regulation
3.5.1 FDA Guidelines on Toxicity Testing
3.5.2 EPA Regulations on Chemical Safety
3.5.3 TSCA Compliance Requirements
3.5.4 State-Level Toxicity Testing Mandates
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. United States Toxicity Testing Outsourcing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. United States Toxicity Testing Outsourcing Market Segmentation
8.1 By Type
8.1.1 In Vivo Testing
8.1.2 In Vitro Testing
8.1.3 Computational Toxicology
8.1.4 Ecotoxicology Testing
8.1.5 Inhalation Toxicology Testing
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Chemical Manufacturers
8.2.4 Academic & Research Institutes
8.2.5 Medical Device Companies
8.2.6 Others
8.3 By Application
8.3.1 Drug Development
8.3.2 Environmental Safety
8.3.3 Product Safety Testing
8.3.4 Regulatory Compliance
8.3.5 Food & Beverage Safety
8.3.6 Cosmetics & Personal Care Testing
8.3.7 Others
8.4 By Service Type
8.4.1 Full-Service Testing
8.4.2 Specialized Testing Services
8.4.3 Consulting Services
8.4.4 Data Management & Reporting
8.4.5 Others
8.5 By Region
8.5.1 Northeast
8.5.2 Midwest
8.5.3 South
8.5.4 West
8.5.5 Others
8.6 By Pricing Model
8.6.1 Fixed Pricing
8.6.2 Hourly Rates
8.6.3 Subscription-Based
8.6.4 Outcome-Based Pricing
8.6.5 Others
8.7 By Client Type
8.7.1 Large Enterprises
8.7.2 SMEs
8.7.3 Government Agencies
8.7.4 Nonprofit Organizations
8.7.5 Others
9. United States Toxicity Testing Outsourcing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 U.S. Toxicity Testing Outsourcing Revenue
9.2.4 Revenue Growth Rate (U.S. Market)
9.2.5 Number of U.S. Toxicology Studies Conducted Annually
9.2.6 Market Penetration Rate (U.S. Share)
9.2.7 Service Portfolio Breadth (Number of Service Categories)
9.2.8 Average Study Turnaround Time
9.2.9 Client Retention Rate
9.2.10 Regulatory Compliance Track Record (GLP/FDA/EPA)
9.2.11 Innovation Index (Patents, New Methods Introduced)
9.2.12 Client Satisfaction Score (U.S. Market)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Charles River Laboratories International, Inc.
9.5.2 Labcorp Drug Development (formerly Covance Inc.)
9.5.3 Eurofins Scientific SE
9.5.4 SGS S.A.
9.5.5 Intertek Group plc
9.5.6 WuXi AppTec Co., Ltd.
9.5.7 Medpace Holdings, Inc.
9.5.8 Envigo RMS LLC
9.5.9 Toxikon Corporation (now part of Labcorp)
9.5.10 BioReliance (a Merck KGaA company)
9.5.11 Q2 Solutions (an IQVIA & Quest Diagnostics joint venture)
9.5.12 ICON plc
9.5.13 Thermo Fisher Scientific Inc.
9.5.14 Accelera S.r.l.
9.5.15 Catalent, Inc.
10. United States Toxicity Testing Outsourcing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Federal Agencies
10.1.2 State Departments
10.1.3 Local Government Units
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Budget Allocations
10.2.2 Investment Trends
10.2.3 Funding Sources
10.3 Pain Point Analysis by End-User Category
10.3.1 Regulatory Compliance Challenges
10.3.2 Cost Management Issues
10.3.3 Quality Assurance Concerns
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Long-term Benefits
11. United States Toxicity Testing Outsourcing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Timeline
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the EPA and FDA
- Review of scientific literature on toxicity testing methodologies and outsourcing trends
- Examination of market reports from industry associations and consultancy firms
Primary Research
- Interviews with executives from contract research organizations (CROs) specializing in toxicity testing
- Surveys targeting pharmaceutical and chemical companies outsourcing toxicity testing
- Focus groups with regulatory affairs professionals to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks conducted through expert panel reviews to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on U.S. spending on toxicity testing services
- Segmentation of the market by industry verticals such as pharmaceuticals, chemicals, and cosmetics
- Incorporation of trends in regulatory requirements influencing outsourcing decisions
Bottom-up Modeling
- Collection of data on service pricing from leading CROs and testing laboratories
- Estimation of market share based on the number of contracts awarded in the past year
- Volume x cost analysis to derive revenue estimates for different service categories
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as R&D spending and regulatory changes
- Scenario modeling based on potential shifts in outsourcing trends and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Toxicity Testing | 100 | R&D Directors, Regulatory Affairs Managers |
| Chemical Industry Outsourcing | 80 | Procurement Managers, Quality Assurance Officers |
| Cosmetic Product Testing | 70 | Product Development Managers, Compliance Specialists |
| Environmental Toxicology Services | 60 | Environmental Scientists, Risk Assessment Analysts |
| Contract Research Organizations | 90 | Business Development Managers, Operations Directors |
Frequently Asked Questions
What is the current value of the United States Toxicity Testing Outsourcing Market?
The United States Toxicity Testing Outsourcing Market is valued at approximately USD 1.6 billion, reflecting a significant growth driven by increasing demand for safety assessments in pharmaceuticals, chemicals, and consumer products, along with stringent regulatory requirements.