About the Report
Base Year 2024US Anesthesia Needles Syringes Market Overview
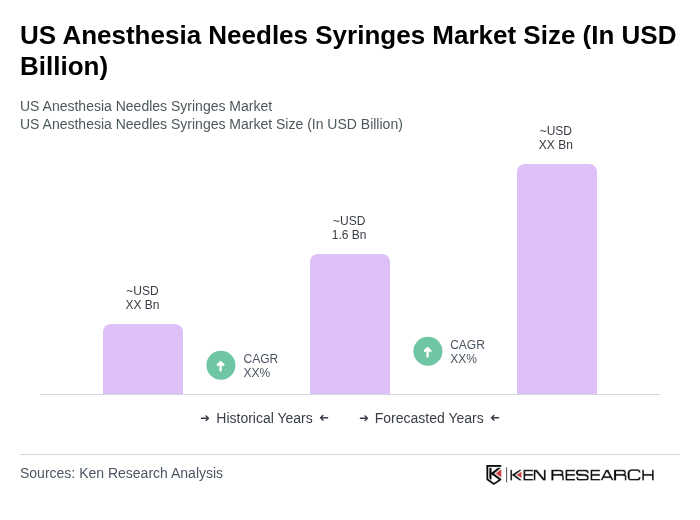
- The US Anesthesia Needles Syringes Market is valued at USD 1.6 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases requiring surgical interventions, the rising number of inpatient and outpatient surgical procedures in the US, and continuous advancements in anesthesia drug delivery and regional anesthesia techniques. The growing focus on patient safety, accurate dosing, and minimally invasive procedures has further supported demand for specialized anesthesia needles and syringes in operating rooms and ambulatory surgical centers.
- Demand for anesthesia needles and syringes is concentrated in major healthcare hubs such as New York, Los Angeles, and Chicago, supported by extensive hospital networks, high surgical case volumes, and a strong presence of academic medical centers and specialty surgery facilities. These metropolitan areas host leading integrated delivery networks and research-focused institutions that are early adopters of advanced anesthesia delivery products and safety-engineered devices.
- US requirements on safety-engineered needles and syringes in healthcare settings are primarily governed by the Needlestick Safety and Prevention Act, 2000, enacted by the United States Congress and implemented through the Occupational Safety and Health Administration (OSHA) Bloodborne Pathogens Standard (29 CFR 1910.1030, revised 2001). This framework obliges employers to use safety-engineered sharps where feasible, maintain an exposure control plan, and involve non?managerial clinical staff in selecting safer medical devices, which has significantly driven adoption of safety syringes and safety needles, including those used for anesthesia applications.
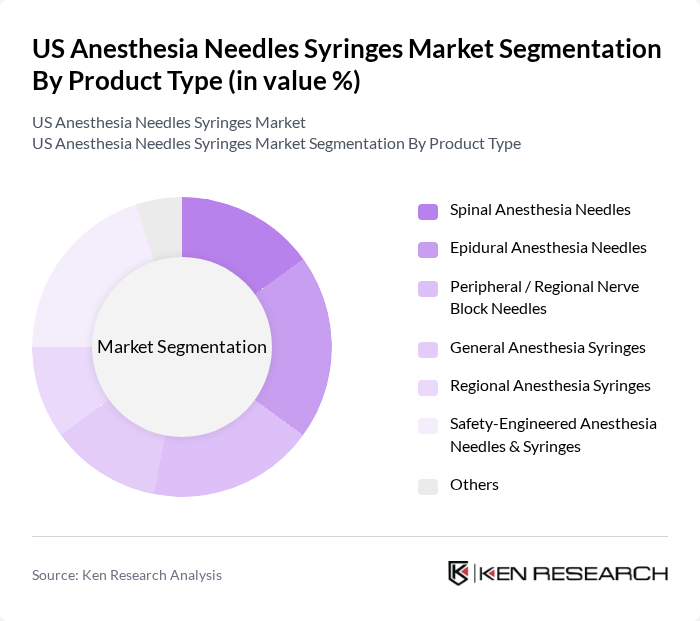
US Anesthesia Needles Syringes Market Segmentation
By Product Type:The product type segmentation includes various categories of anesthesia needles and syringes, each serving specific medical needs. The subsegments are Spinal Anesthesia Needles, Epidural Anesthesia Needles, Peripheral / Regional Nerve Block Needles, General Anesthesia Syringes, Regional Anesthesia Syringes, Safety-Engineered Anesthesia Needles & Syringes, and Others. Spinal and epidural needles are widely used for neuraxial anesthesia in obstetric, orthopedic, and lower abdominal procedures, while peripheral nerve block needles support ultrasound-guided regional techniques for postoperative pain control. Among these, Safety-Engineered Anesthesia Needles & Syringes are gaining traction due to occupational safety requirements, OSHA-driven sharps injury prevention policies, and heightened awareness regarding needlestick injury risk in operating rooms and recovery units.
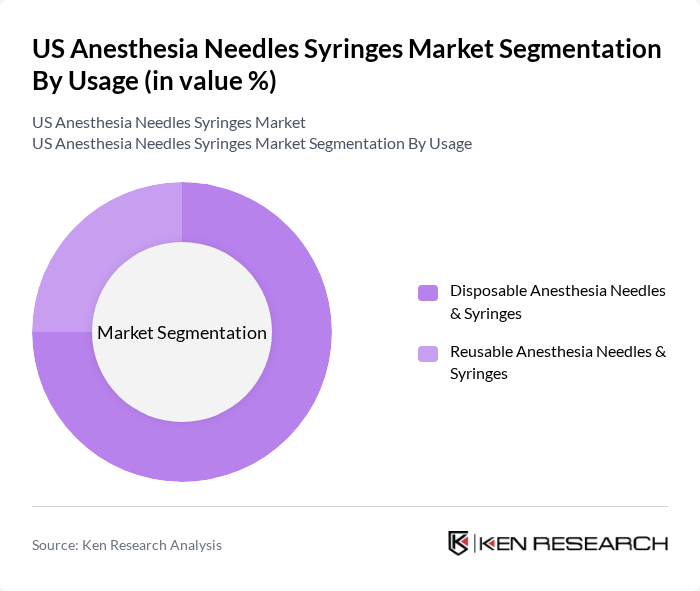
By Usage:The usage segmentation categorizes anesthesia needles and syringes into Disposable and Reusable types. Disposable Anesthesia Needles & Syringes are preferred in most healthcare settings due to their convenience, alignment with infection prevention protocols, and compliance with hospital policies favoring single-use devices in operating rooms and ambulatory surgical centers. Reusable Anesthesia Needles & Syringes are utilized in specific scenarios where cost-effectiveness and controlled reprocessing capabilities are prioritized, often in institutions with robust sterilization infrastructure. The disposable segment is currently leading the market due to the growing emphasis on hygiene, prevention of cross-contamination, and integration of safety-engineered features in single-use anesthesia delivery products.
US Anesthesia Needles Syringes Market Competitive Landscape
The US Anesthesia Needles Syringes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Becton, Dickinson and Company (BD), Medtronic plc, B. Braun Melsungen AG, Smiths Medical (ICU Medical, Inc.), Terumo Corporation, Cardinal Health, Inc., Fresenius Kabi AG, Nipro Corporation, Vygon SA, Cook Medical LLC, Teleflex Incorporated, Boston Scientific Corporation, Baxter International Inc., GE HealthCare Technologies Inc., Philips Healthcare (Koninklijke Philips N.V.) contribute to innovation, geographic expansion, and service delivery in this space.
US Anesthesia Needles Syringes Market Industry Analysis
Growth Drivers
- Increasing Surgical Procedures:The number of surgical procedures in the U.S. is projected to reach approximately 50 million in future, driven by an aging population and advancements in surgical techniques. The American College of Surgeons reported that outpatient surgeries have increased by 20% over the past five years, indicating a growing demand for anesthesia needles and syringes. This surge in surgical activity directly correlates with the need for efficient anesthesia delivery systems, thereby boosting market growth.
- Rising Prevalence of Chronic Diseases:Chronic diseases such as diabetes and cardiovascular conditions are affecting over 133 million Americans, according to the CDC. This rising prevalence necessitates more frequent surgical interventions and pain management procedures, which in turn increases the demand for anesthesia needles and syringes. The healthcare expenditure on chronic disease management is expected to exceed $4 trillion in future, further driving the need for effective anesthesia solutions in clinical settings.
- Technological Advancements in Anesthesia Delivery:Innovations in anesthesia delivery systems, including the development of smart syringes and automated anesthesia machines, are enhancing patient safety and procedural efficiency. The market for smart syringes alone is anticipated to grow to $1.5 billion in future, as these devices reduce the risk of needle-stick injuries and improve dosage accuracy. Such advancements are pivotal in meeting the increasing demands of modern surgical practices, thereby propelling market growth.
Market Challenges
- Stringent Regulatory Requirements:The U.S. FDA imposes rigorous regulations on medical devices, including anesthesia needles and syringes, which can delay product approvals. In future, the FDA is expected to increase scrutiny on safety and efficacy data, potentially extending the average approval timeline to 18 months. This regulatory environment poses a significant challenge for manufacturers aiming to innovate and bring new products to market swiftly, impacting overall market dynamics.
- High Cost of Advanced Anesthesia Equipment:The cost of advanced anesthesia equipment can exceed $100,000, making it a significant investment for healthcare facilities. With budget constraints prevalent in many hospitals, particularly in rural areas, the high upfront costs can limit access to the latest technologies. This financial barrier can hinder the adoption of innovative anesthesia solutions, thereby affecting the overall growth of the anesthesia needles and syringes market.
US Anesthesia Needles Syringes Market Future Outlook
The future of the U.S. anesthesia needles and syringes market appears promising, driven by ongoing advancements in technology and an increasing focus on patient safety. As healthcare facilities continue to adopt minimally invasive procedures, the demand for innovative anesthesia solutions will likely rise. Additionally, the expansion of outpatient surgical centers is expected to create new opportunities for market players, fostering a competitive landscape that prioritizes efficiency and safety in anesthesia delivery.
Market Opportunities
- Expansion of Healthcare Infrastructure:The U.S. is investing over $200 billion in healthcare infrastructure improvements in future, which includes the establishment of new surgical centers. This expansion will create a higher demand for anesthesia needles and syringes, providing manufacturers with opportunities to supply innovative products tailored to modern surgical environments.
- Development of Innovative Anesthesia Products:The market for innovative anesthesia products, such as needle-free delivery systems, is projected to grow significantly. With an estimated investment of $1 billion in R&D by major players, the introduction of safer and more efficient products will cater to the evolving needs of healthcare providers, enhancing patient outcomes and driving market growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Spinal Anesthesia Needles Epidural Anesthesia Needles Peripheral / Regional Nerve Block Needles General Anesthesia Syringes Regional Anesthesia Syringes Safety-Engineered Anesthesia Needles & Syringes Others |
| By Usage | Disposable Anesthesia Needles & Syringes Reusable Anesthesia Needles & Syringes |
| By Application | General Anesthesia Regional Anesthesia (including nerve blocks) Local Anesthesia Pain Management Procedures Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics & Pain Clinics Office-based Anesthesia Practices Others |
| By Material | Stainless Steel Plastic / Polymer Components Glass Components Others |
| By Distribution Channel | Direct Sales / Tender-based Procurement Group Purchasing Organizations (GPOs) Hospital & Retail Pharmacies Online / E-commerce Channels Others |
| By US Region | Northeast Midwest South West |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Centers for Medicare & Medicaid Services)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Wholesalers
Industry Associations (e.g., American Society of Anesthesiologists)
Financial Institutions
Players Mentioned in the Report:
Becton, Dickinson and Company (BD)
Medtronic plc
B. Braun Melsungen AG
Smiths Medical (ICU Medical, Inc.)
Terumo Corporation
Cardinal Health, Inc.
Fresenius Kabi AG
Nipro Corporation
Vygon SA
Cook Medical LLC
Teleflex Incorporated
Boston Scientific Corporation
Baxter International Inc.
GE HealthCare Technologies Inc.
Philips Healthcare (Koninklijke Philips N.V.)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Anesthesia Needles Syringes Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Anesthesia Needles Syringes Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Anesthesia Needles Syringes Market Analysis
3.1 Growth Drivers
3.1.1 Increasing surgical procedures
3.1.2 Rising prevalence of chronic diseases
3.1.3 Technological advancements in anesthesia delivery
3.1.4 Growing demand for outpatient surgeries
3.2 Market Challenges
3.2.1 Stringent regulatory requirements
3.2.2 High cost of advanced anesthesia equipment
3.2.3 Risk of needle-stick injuries
3.2.4 Limited reimbursement policies
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing focus on patient safety
3.3.3 Growth in medical tourism
3.3.4 Development of innovative anesthesia products
3.4 Market Trends
3.4.1 Shift towards minimally invasive procedures
3.4.2 Adoption of smart syringes
3.4.3 Integration of digital technologies in anesthesia
3.4.4 Rising awareness about pain management
3.5 Government Regulation
3.5.1 FDA regulations on medical devices
3.5.2 Compliance with safety standards
3.5.3 Guidelines for anesthesia practices
3.5.4 Reporting requirements for adverse events
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Anesthesia Needles Syringes Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Anesthesia Needles Syringes Market Segmentation
8.1 By Product Type
8.1.1 Spinal Anesthesia Needles
8.1.2 Epidural Anesthesia Needles
8.1.3 Peripheral / Regional Nerve Block Needles
8.1.4 General Anesthesia Syringes
8.1.5 Regional Anesthesia Syringes
8.1.6 Safety-Engineered Anesthesia Needles & Syringes
8.1.7 Others
8.2 By Usage
8.2.1 Disposable Anesthesia Needles & Syringes
8.2.2 Reusable Anesthesia Needles & Syringes
8.3 By Application
8.3.1 General Anesthesia
8.3.2 Regional Anesthesia (including nerve blocks)
8.3.3 Local Anesthesia
8.3.4 Pain Management Procedures
8.3.5 Others
8.4 By End-User
8.4.1 Hospitals
8.4.2 Ambulatory Surgical Centers
8.4.3 Specialty Clinics & Pain Clinics
8.4.4 Office-based Anesthesia Practices
8.4.5 Others
8.5 By Material
8.5.1 Stainless Steel
8.5.2 Plastic / Polymer Components
8.5.3 Glass Components
8.5.4 Others
8.6 By Distribution Channel
8.6.1 Direct Sales / Tender-based Procurement
8.6.2 Group Purchasing Organizations (GPOs)
8.6.3 Hospital & Retail Pharmacies
8.6.4 Online / E-commerce Channels
8.6.5 Others
8.7 By US Region
8.7.1 Northeast
8.7.2 Midwest
8.7.3 South
8.7.4 West
9. US Anesthesia Needles Syringes Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 US Anesthesia Needles & Syringes Revenue (USD Mn)
9.2.4 3-year CAGR in Segment Revenue
9.2.5 US Market Share in Anesthesia Needles & Syringes (%)
9.2.6 Volume Sold (Units / Million Units per Year)
9.2.7 R&D Intensity (% of Revenue)
9.2.8 Product Innovation Index (New SKUs / Patents Filed p.a.)
9.2.9 Average Selling Price Positioning vs Market
9.2.10 EBITDA Margin – Anesthesia Portfolio (%)
9.2.11 Contract Wins with GPOs / IDNs (Number per Year)
9.2.12 Geographic Coverage (No. of States / Regions Served)
9.2.13 Key Hospital & ASC Accounts (Number of Active Accounts)
9.2.14 On-time Delivery / Service Level Performance (%)
9.2.15 Customer Satisfaction / Net Promoter Score (NPS)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Becton, Dickinson and Company (BD)
9.5.2 Medtronic plc
9.5.3 B. Braun Melsungen AG
9.5.4 Smiths Medical (ICU Medical, Inc.)
9.5.5 Terumo Corporation
9.5.6 Cardinal Health, Inc.
9.5.7 Fresenius Kabi AG
9.5.8 Nipro Corporation
9.5.9 Vygon SA
9.5.10 Cook Medical LLC
9.5.11 Teleflex Incorporated
9.5.12 Boston Scientific Corporation
9.5.13 Baxter International Inc.
9.5.14 GE HealthCare Technologies Inc.
9.5.15 Philips Healthcare (Koninklijke Philips N.V.)
10. US Anesthesia Needles Syringes Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Procurement Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Facilities
10.2.2 Spending on Anesthesia Equipment
10.2.3 Budgeting for Safety Measures
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Ambulatory Surgical Centers
10.3.3 Concerns of Clinics
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion Opportunities
10.5.3 User Feedback Mechanisms
10.5.4 Others
11. US Anesthesia Needles Syringes Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare associations and market research firms
- Review of published articles and white papers on anesthesia practices and needle technology
- Examination of regulatory guidelines and standards from the FDA and CDC regarding anesthesia equipment
Primary Research
- Interviews with anesthesiologists and nurse anesthetists to gather insights on product preferences
- Surveys with hospital procurement managers to understand purchasing trends and volume
- Field visits to surgical centers to observe the usage of anesthesia needles and syringes
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and focus groups
- Triangulation of data from sales reports, clinical studies, and market trends
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and surgical procedure volumes
- Segmentation of the market by product type, including disposable and reusable syringes
- Incorporation of growth rates from related medical device markets and anesthesia trends
Bottom-up Modeling
- Collection of sales data from leading manufacturers of anesthesia needles and syringes
- Estimation of market share based on unit sales and pricing strategies
- Volume calculations based on the number of procedures performed across various healthcare settings
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical data and projected growth in surgical procedures
- Scenario analysis based on potential changes in healthcare policies and technological advancements
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Anesthesia Departments | 140 | Anesthesiologists, Nurse Anesthetists |
| Outpatient Surgery Centers | 90 | Surgeons, Surgical Coordinators |
| Medical Device Distributors | 60 | Sales Managers, Product Specialists |
| Healthcare Procurement Offices | 110 | Procurement Officers, Supply Chain Managers |
| Clinical Research Organizations | 50 | Clinical Researchers, Regulatory Affairs Specialists |
Frequently Asked Questions
What is the current value of the US Anesthesia Needles Syringes Market?
The US Anesthesia Needles Syringes Market is valued at approximately USD 1.6 billion, reflecting a significant growth driven by the increasing prevalence of chronic diseases and advancements in anesthesia drug delivery systems.