About the Report
Base Year 2024Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Overview
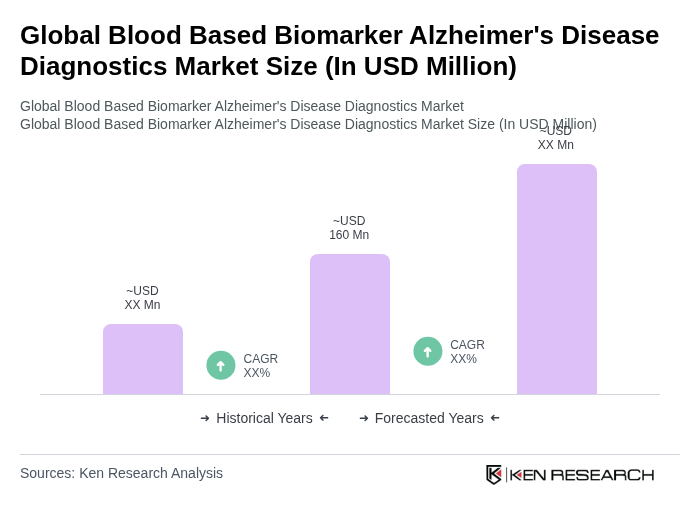
- The Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market is valued at USD 160 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of Alzheimer's disease, advancements in biomarker research, and the rising demand for early and accurate diagnostic tools. The integration of blood-based biomarkers into clinical practice has significantly improved diagnostic accuracy and patient management.
- Key players in this market include the United States, Germany, and Japan, which dominate due to their robust healthcare infrastructure, significant investments in research and development, and a high prevalence of Alzheimer's disease. The presence of leading pharmaceutical and biotechnology companies in these regions further enhances their market position, facilitating innovation and access to advanced diagnostic technologies. North America emerged as the largest revenue-generating region in 2024, driven by early adoption of blood-based diagnostic technologies and strong regulatory support.
- In May 2025, the U.S. Food and Drug Administration cleared the Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio as the first in vitro diagnostic device for blood-based Alzheimer's disease diagnosis. This regulatory milestone represents a significant advancement in streamlining the approval process for blood-based biomarker tests, ensuring that innovative diagnostic tests can reach the market while maintaining high standards for safety and efficacy. The clearance is expected to foster innovation and improve patient outcomes in Alzheimer's disease management.
Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Segmentation
By Type:This segmentation includes various types of biomarkers used in Alzheimer's diagnostics. The subsegments are as follows:
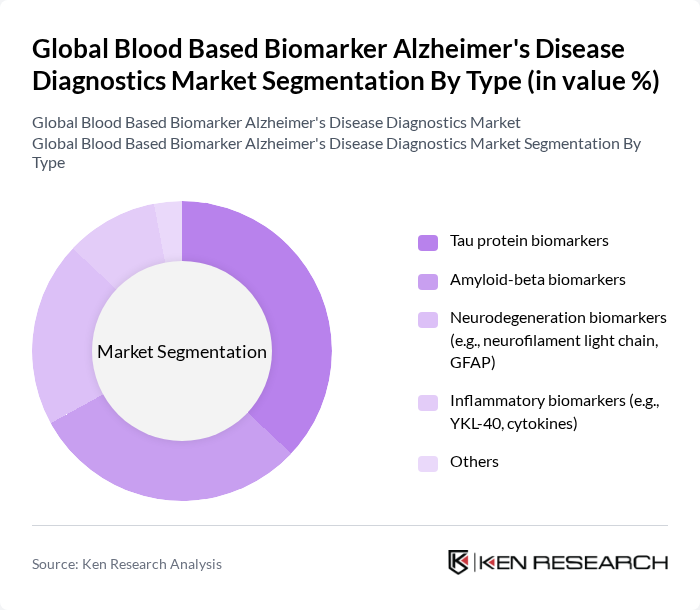
The market for blood-based biomarkers in Alzheimer's diagnostics is increasingly driven by tau-related markers, which demonstrate strong correlation with neurodegeneration and predictive value for cognitive decline and disease progression. Tau markers' proven effectiveness in detecting neurofibrillary tangle pathology and monitoring disease progression directly addresses clinical needs for accurate staging and prognosis in Alzheimer's disease management. Amyloid-beta biomarkers remain crucial for early detection and diagnosis, widely accepted in clinical settings due to their strong correlation with Alzheimer's pathology. The increasing focus on early diagnosis and the growing body of research supporting the use of these biomarkers contribute to their market dominance. Additionally, advancements in technology and the development of more sensitive assays are expected to further enhance their market position.
By End-User:This segmentation focuses on the various end-users of blood-based biomarkers in Alzheimer's diagnostics. The subsegments are as follows:
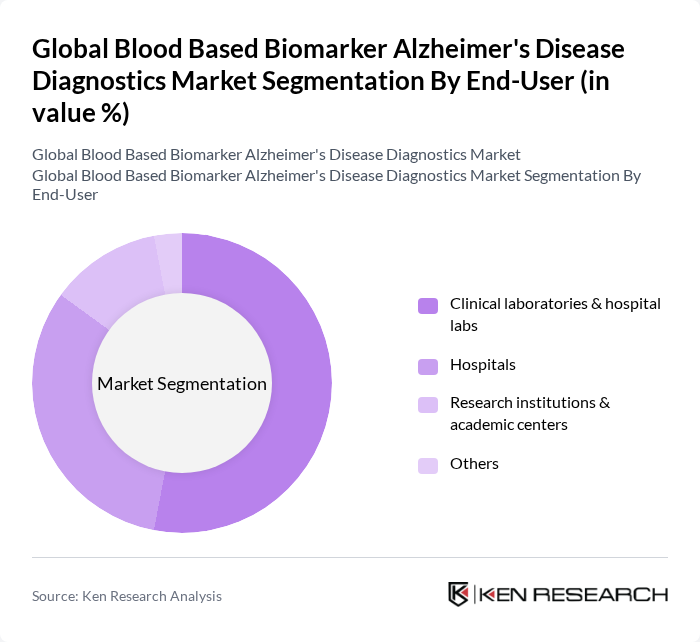
Clinical laboratories and hospital laboratories are the leading end-users of blood-based biomarkers for Alzheimer's diagnostics, accounting for a significant share of the market. This dominance is attributed to the integration of blood-based biomarker testing into routine laboratory menus while providing comprehensive diagnostic services for neurological disorders. Hospital laboratories and commercial diagnostic labs increasingly align with healthcare delivery models that emphasize centralized laboratory testing and standardized diagnostic protocols. The high volume of diagnostic tests conducted in these settings, where timely and accurate diagnosis is critical for patient management, further strengthens this segment. Additionally, these facilities often have access to advanced diagnostic technologies and skilled personnel, facilitating the implementation of innovative biomarker tests. The increasing collaboration between hospitals and diagnostic companies continues to strengthen this segment's position.
Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Competitive Landscape
The Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Eli Lilly and Company, Biogen Inc., Abbott Laboratories, Quest Diagnostics, Siemens Healthineers, C2N Diagnostics, Quanterix Corporation, Fujirebio, Grifols S.A., Avid Radiopharmaceuticals, Simoa (Quanterix Simoa Technology), Alzheon, Inc., Amprion Inc., MagQu Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Alzheimer's Disease:The global prevalence of Alzheimer's disease is projected to reach approximately 152 million cases by 2050, according to the World Health Organization. This rising incidence is driving demand for effective diagnostic tools. In the future, an estimated 6.7 million individuals aged 65 and older in the U.S. are expected to be living with Alzheimer's, highlighting the urgent need for innovative diagnostic solutions that can facilitate early detection and intervention.
- Advancements in Blood Biomarker Technologies:Recent advancements in blood biomarker technologies have led to the identification of over 30 potential biomarkers for Alzheimer's disease, significantly enhancing diagnostic accuracy. The National Institutes of Health reported that blood tests can now detect amyloid-beta and tau proteins, which are critical in Alzheimer's pathology. These innovations are expected to streamline the diagnostic process, making it more accessible and efficient for healthcare providers in the future and beyond.
- Rising Demand for Non-Invasive Diagnostic Methods:The demand for non-invasive diagnostic methods is surging, with a projected market value of $2.5 billion for blood-based diagnostics in neurology in the future. Patients and healthcare providers prefer these methods due to their ease of use and reduced risk compared to traditional invasive procedures. This trend is further supported by a growing emphasis on patient-centered care, which prioritizes comfort and convenience in the diagnostic process.
Market Challenges
- High Costs of Research and Development:The average cost of developing a new diagnostic test can exceed $1 billion, according to industry reports. This financial burden poses a significant challenge for companies in the blood-based biomarker sector, particularly for small to mid-sized enterprises. The high costs associated with R&D can limit innovation and slow the introduction of new diagnostic solutions to the market, impacting overall growth in the future.
- Regulatory Hurdles in Product Approval:Navigating the regulatory landscape for blood-based diagnostics can be complex and time-consuming. The FDA and EMA have stringent requirements that can delay product approvals by several years. For instance, the average time for a diagnostic test to receive FDA approval is approximately 12-18 months, which can hinder timely access to innovative solutions for Alzheimer's diagnostics, affecting market dynamics in the future.
Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Future Outlook
The future of blood-based biomarker diagnostics for Alzheimer's disease appears promising, driven by technological advancements and increasing healthcare investments. As the global population ages, the demand for early and accurate diagnostic tools will intensify. Innovations in artificial intelligence and machine learning are expected to enhance diagnostic accuracy and efficiency. Furthermore, collaborations between biotech firms and research institutions will likely accelerate the development of personalized medicine approaches, ensuring tailored treatment options for patients in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets, particularly in Asia-Pacific and Latin America, present significant growth opportunities. With a combined population of over 4 billion, these regions are witnessing an increase in Alzheimer's cases, creating a demand for accessible diagnostic solutions. Companies that strategically enter these markets can capitalize on the growing need for effective Alzheimer's diagnostics, potentially increasing their market share.
- Collaborations with Research Institutions:Collaborating with academic and research institutions can enhance innovation in blood-based biomarker diagnostics. Such partnerships can facilitate access to cutting-edge research and technology, enabling companies to develop more effective diagnostic tools. By leveraging shared resources and expertise, these collaborations can accelerate the introduction of new products to the market, driving growth in the sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Amyloid-beta biomarkers Tau protein biomarkers Neurodegeneration biomarkers (e.g., neurofilament light chain, GFAP) Inflammatory biomarkers (e.g., YKL-40, cytokines) Others |
| By End-User | Hospitals Clinical laboratories & diagnostic laboratories Research institutions & academic centers Others |
| By Sample Type | Plasma Serum Whole blood Dried blood spot Others |
| By Technology | Immunoassays (e.g., ELISA, Simoa) Mass spectrometry PCR-based methods Multiplex platforms Others |
| By Distribution Channel | Direct sales Online sales Distributors Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Application | Clinical diagnostics Research applications Drug development & clinical trials Screening & risk assessment Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical Companies
Biotechnology Firms
Clinical Laboratories
Healthcare Providers and Institutions
Diagnostic Test Manufacturers
Health Insurance Companies
Players Mentioned in the Report:
Roche Diagnostics
Eli Lilly and Company
Biogen Inc.
Abbott Laboratories
Quest Diagnostics
Siemens Healthineers
C2N Diagnostics
Quanterix Corporation
Fujirebio
Grifols S.A.
Avid Radiopharmaceuticals
Simoa (Quanterix Simoa Technology)
Alzheon, Inc.
Amprion Inc.
MagQu Co., Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Alzheimer's disease
3.1.2 Advancements in blood biomarker technologies
3.1.3 Rising demand for non-invasive diagnostic methods
3.1.4 Growing investment in Alzheimer's research
3.2 Market Challenges
3.2.1 High costs of research and development
3.2.2 Regulatory hurdles in product approval
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with research institutions
3.3.3 Development of personalized medicine approaches
3.3.4 Integration of AI in diagnostic processes
3.4 Market Trends
3.4.1 Increasing focus on preventive healthcare
3.4.2 Growth of telemedicine and remote diagnostics
3.4.3 Rising consumer demand for health monitoring
3.4.4 Advancements in biomarker discovery
3.5 Government Regulation
3.5.1 FDA guidelines for blood-based diagnostics
3.5.2 European Medicines Agency (EMA) regulations
3.5.3 Health Canada requirements for diagnostics
3.5.4 Compliance with CLIA regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Segmentation
8.1 By Type
8.1.1 Amyloid-beta biomarkers
8.1.2 Tau protein biomarkers
8.1.3 Neurodegeneration biomarkers (e.g., neurofilament light chain, GFAP)
8.1.4 Inflammatory biomarkers (e.g., YKL-40, cytokines)
8.1.5 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Clinical laboratories & diagnostic laboratories
8.2.3 Research institutions & academic centers
8.2.4 Others
8.3 By Sample Type
8.3.1 Plasma
8.3.2 Serum
8.3.3 Whole blood
8.3.4 Dried blood spot
8.3.5 Others
8.4 By Technology
8.4.1 Immunoassays (e.g., ELISA, Simoa)
8.4.2 Mass spectrometry
8.4.3 PCR-based methods
8.4.4 Multiplex platforms
8.4.5 Others
8.5 By Distribution Channel
8.5.1 Direct sales
8.5.2 Online sales
8.5.3 Distributors
8.5.4 Others
8.6 By Region
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa
8.7 By Application
8.7.1 Clinical diagnostics
8.7.2 Research applications
8.7.3 Drug development & clinical trials
8.7.4 Screening & risk assessment
8.7.5 Others
9. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue from Alzheimer's Blood Biomarker Diagnostics
9.2.4 R&D Investment as % of Revenue
9.2.5 Number of Regulatory Approvals (e.g., FDA, CE Mark)
9.2.6 Market Penetration Rate (Installed base or test volume)
9.2.7 Sensitivity/Specificity of Lead Assay
9.2.8 Time-to-Result (Turnaround Time)
9.2.9 Geographic Coverage (Regions/Countries Served)
9.2.10 Strategic Partnerships/Collaborations
9.2.11 Pipeline Products (Blood-based AD diagnostics)
9.2.12 Pricing Strategy
9.2.13 Customer Satisfaction Score
9.2.14 Brand Awareness Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Eli Lilly and Company
9.5.3 Biogen Inc.
9.5.4 Abbott Laboratories
9.5.5 Quest Diagnostics
9.5.6 Siemens Healthineers
9.5.7 C2N Diagnostics
9.5.8 Quanterix Corporation
9.5.9 Fujirebio
9.5.10 Grifols S.A.
9.5.11 Avid Radiopharmaceuticals
9.5.12 Simoa (Quanterix Simoa Technology)
9.5.13 Alzheon, Inc.
9.5.14 Amprion Inc.
9.5.15 MagQu Co., Ltd.
10. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for Alzheimer's research
10.1.2 Collaboration with healthcare providers
10.1.3 Adoption of innovative diagnostic solutions
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in diagnostic facilities
10.2.2 Funding for research initiatives
10.2.3 Partnerships with technology firms
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to advanced diagnostic tools
10.3.2 Cost of diagnostic procedures
10.3.3 Training for healthcare professionals
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of blood-based diagnostics
10.4.2 Willingness to invest in new technologies
10.4.3 Integration with existing systems
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of diagnostic effectiveness
10.5.2 Long-term cost savings
10.5.3 Expansion into new applications
10.5.4 Others
11. Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps and opportunities
1.2 Business model components
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on blood-based biomarkers for Alzheimer's diagnostics
- Review of scientific literature and clinical studies from medical journals focusing on Alzheimer's disease
- Examination of regulatory guidelines and approvals from health authorities regarding diagnostic tests
Primary Research
- Interviews with neurologists and geriatric specialists involved in Alzheimer's diagnostics
- Surveys with laboratory managers and technicians working with blood-based biomarker tests
- Focus groups with patients and caregivers to understand perceptions and needs regarding diagnostics
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on the prevalence of Alzheimer's disease globally
- Segmentation of the market by geographical regions and types of blood-based biomarkers
- Incorporation of trends in healthcare spending and diagnostic advancements
Bottom-up Modeling
- Collection of sales data from leading diagnostic companies specializing in Alzheimer's biomarkers
- Estimation of market share based on product offerings and pricing strategies
- Volume estimates derived from the number of tests conducted annually across various healthcare settings
Forecasting & Scenario Analysis
- Multi-variable forecasting using factors such as aging population, healthcare policies, and technological advancements
- Scenario analysis based on potential regulatory changes and market entry of new diagnostic technologies
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurology Clinics | 100 | Neurologists, Clinic Administrators |
| Diagnostic Laboratories | 80 | Laboratory Managers, Biomedical Scientists |
| Healthcare Providers | 70 | General Practitioners, Geriatricians |
| Patient Advocacy Groups | 50 | Patient Advocates, Caregivers |
| Regulatory Bodies | 30 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market?
The Global Blood Based Biomarker Alzheimer's Disease Diagnostics Market is valued at approximately USD 160 million, reflecting a significant growth driven by the increasing prevalence of Alzheimer's disease and advancements in biomarker research.