About the Report
Base Year 2024Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Overview
- The Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market is valued at USD 6.2 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of Alzheimer's disease, advancements in biomarker research, and the rising demand for early and accurate diagnostic tools. The integration of blood-based biomarkers into clinical practice has significantly improved diagnostic accuracy, leading to better patient outcomes. Key growth drivers include the adoption of minimally invasive diagnostic methods, expansion of healthcare infrastructure in the Gulf region, and the introduction of next-generation biomarker assays targeting amyloid and phosphorylated tau. The deployment of AI-powered analytics and automated immunoassay platforms is further accelerating market adoption and enhancing diagnostic precision .
- Key players in this market include countries such as the United States, Germany, and the United Kingdom, which dominate due to their robust healthcare infrastructure, significant investments in research and development, and a high prevalence of Alzheimer's disease. These regions are also home to leading diagnostic companies that are at the forefront of innovation in blood-based biomarker technologies. In the MEA region, countries like Saudi Arabia, the United Arab Emirates, and South Africa are emerging as growth hubs, supported by government-led healthcare reforms, local clinical trials, and international collaborations that facilitate access to advanced diagnostic solutions .
- In 2023, the European Medicines Agency (EMA) implemented Regulation (EU) 2017/746 on in vitro diagnostic medical devices (IVDR), which established a harmonized framework for the evaluation and approval of blood-based biomarkers used in Alzheimer's diagnostics. This regulation, issued by the European Parliament and the Council, mandates rigorous clinical validation, performance evaluation, and post-market surveillance for diagnostic assays, thereby expediting the approval process and encouraging the development of innovative diagnostic tools while ensuring patient safety and timely market access .
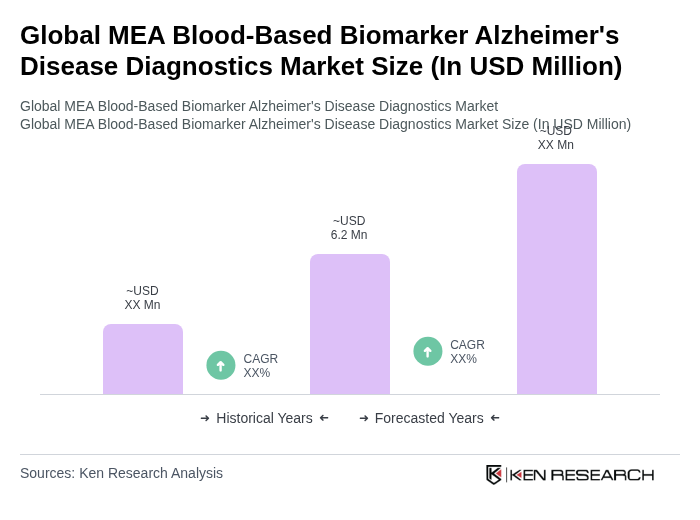
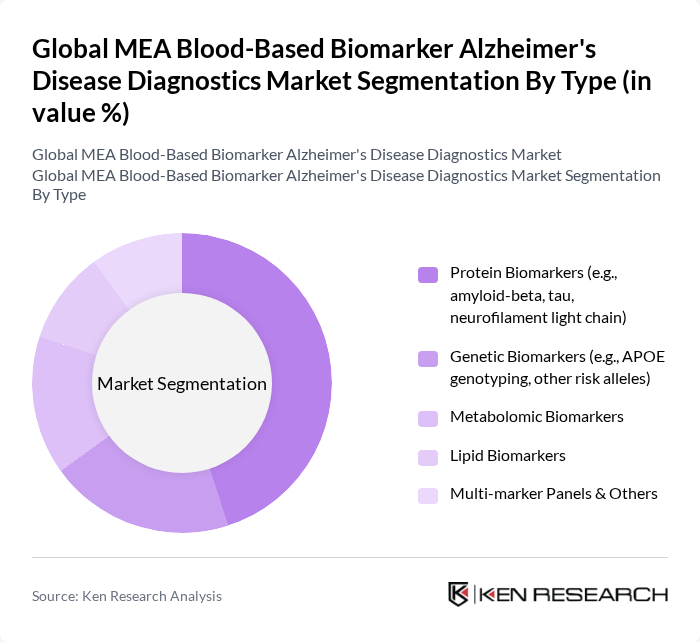
Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Segmentation
By Type:The market is segmented into various types of biomarkers, including Protein Biomarkers, Genetic Biomarkers, Metabolomic Biomarkers, Lipid Biomarkers, and Multi-marker Panels & Others. Among these,Protein Biomarkers, such as amyloid-beta and tau, are leading the market due to their established role in Alzheimer's diagnostics and their ability to provide critical insights into disease pathology. The increasing focus on precision medicine and personalized treatment approaches further drives the demand for these biomarkers. Next-generation assays targeting phosphorylated tau and neurofilament light chain are gaining traction for their enhanced sensitivity and early detection capabilities .
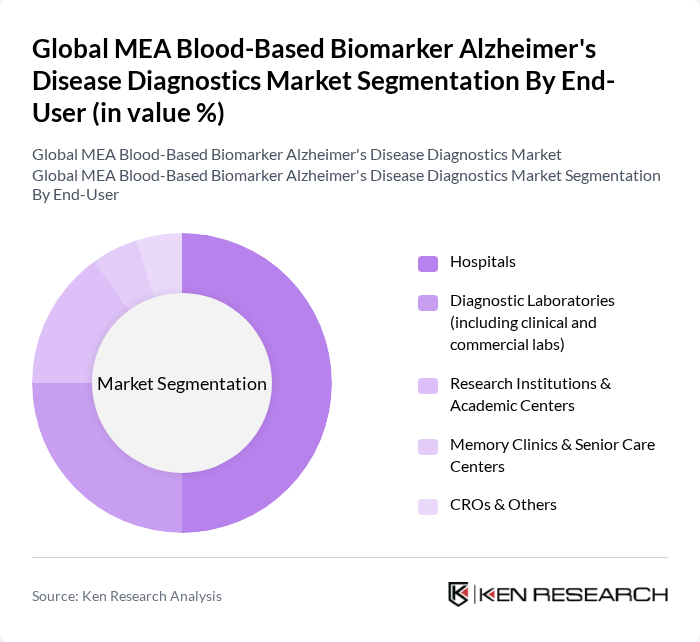
By End-User:The market is segmented by end-users, including Hospitals, Diagnostic Laboratories, Research Institutions & Academic Centers, Memory Clinics & Senior Care Centers, and CROs & Others.Hospitalsare the leading end-user segment, driven by the increasing adoption of advanced diagnostic technologies and the need for accurate and timely diagnosis of Alzheimer's disease. The growing number of patients seeking diagnosis and treatment in hospital settings further supports this trend. Diagnostic laboratories, including clinical and commercial labs, also account for a significant share due to the centralization of laboratory testing and the integration of blood-based biomarker assays into routine diagnostic workflows .
Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Competitive Landscape
The Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Eli Lilly and Company, Biogen Inc., Quest Diagnostics, Siemens Healthineers, C2N Diagnostics, Quanterix Corporation, Fujirebio, Alzheon Inc., Genentech, Avid Radiopharmaceuticals, MagQu Co., Ltd., ADx NeuroSciences, Araclon Biotech contribute to innovation, geographic expansion, and service delivery in this space.
Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Alzheimer's Disease:The World Health Organization reported that approximately 55 million people worldwide are living with dementia, with Alzheimer's disease accounting for 60-70% of these cases. In the Middle East and Africa, the prevalence is projected to rise by 25% in the near future, driven by an aging population and lifestyle factors. This growing patient base is fueling demand for effective diagnostic solutions, particularly blood-based biomarkers, which are seen as a less invasive and more accessible option for early detection.
- Advancements in Blood-Based Biomarker Technologies:Recent innovations in biomarker technologies have led to the identification of over 40 potential blood-based biomarkers for Alzheimer's disease. For instance, the development of assays that can detect amyloid-beta and tau proteins in blood samples has shown promise. The global investment in biomarker research is expected to exceed $6 billion in the near future, enhancing the capabilities of diagnostics and improving accuracy, thus driving market growth in the MEA region.
- Rising Demand for Early Diagnosis and Personalized Medicine:The shift towards personalized medicine is evident, with a growing emphasis on early diagnosis of Alzheimer's disease. According to the Alzheimer's Association, early intervention can delay the onset of severe symptoms by up to 3 years. In future, the demand for early diagnostic tools is expected to increase, with healthcare systems in the MEA region allocating approximately $1.5 billion towards innovative diagnostic solutions, including blood-based biomarkers, to enhance patient outcomes.
Market Challenges
- High Costs Associated with Biomarker Testing:The cost of blood-based biomarker testing can be prohibitive, with prices ranging from $600 to $1,800 per test. This financial barrier limits accessibility, particularly in low- and middle-income countries within the MEA region. As healthcare budgets tighten, the allocation for advanced diagnostic tools may be deprioritized, hindering market growth and adoption of these innovative solutions in clinical settings.
- Regulatory Hurdles and Approval Processes:The regulatory landscape for biomarker testing is complex, with stringent guidelines imposed by health authorities. In the MEA region, the average time for regulatory approval can exceed 24 months, delaying market entry for new diagnostic tests. This prolonged approval process can stifle innovation and deter investment in research and development, posing a significant challenge for companies looking to introduce new blood-based biomarkers.
Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Future Outlook
The future of blood-based biomarker diagnostics for Alzheimer's disease in the MEA region appears promising, driven by technological advancements and increasing healthcare investments. As the prevalence of Alzheimer's continues to rise, the demand for innovative diagnostic solutions will likely grow. Furthermore, the integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, paving the way for more personalized treatment approaches. Collaborative efforts between private and public sectors will be crucial in overcoming existing challenges and fostering market growth.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets in the MEA region present significant opportunities for blood-based biomarker diagnostics. With a growing middle class and increasing healthcare expenditure, the demand for advanced diagnostic solutions is expected to rise. Companies can leverage this trend by establishing partnerships with local healthcare providers to enhance market penetration and accessibility.
- Development of Novel Biomarkers:The ongoing research into novel biomarkers offers substantial opportunities for innovation. By focusing on the discovery of unique biomarkers that can improve diagnostic accuracy, companies can differentiate themselves in a competitive market. Collaborations with academic institutions and research organizations can facilitate the development of these novel solutions, driving growth in the sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Protein Biomarkers (e.g., amyloid-beta, tau, neurofilament light chain) Genetic Biomarkers (e.g., APOE genotyping, other risk alleles) Metabolomic Biomarkers Lipid Biomarkers Multi-marker Panels & Others |
| By End-User | Hospitals Diagnostic Laboratories (including clinical and commercial labs) Research Institutions & Academic Centers Memory Clinics & Senior Care Centers CROs & Others |
| By Sample Type | Whole Blood Samples Plasma Samples Serum Samples Dried Blood Spot & Others |
| By Biomarker Class | Amyloid Biomarkers (e.g., A?42/40 ratio) Tau Biomarkers (e.g., p-tau181, p-tau217, total tau) Neurodegeneration Biomarkers (e.g., NfL, GFAP) Inflammatory & Other Biomarkers |
| By Distribution Channel | Direct Sales (to hospitals, labs, clinics) Online Sales (including digital health platforms) Distributors & Local Partners Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa (with focus on Saudi Arabia, UAE, South Africa, Egypt) |
| By Research Type | Clinical Research Preclinical Research Real-World Evidence & Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, European Medicines Agency)
Biotechnology Companies
Pharmaceutical Companies
Clinical Laboratories
Healthcare Providers and Institutions
Diagnostic Test Manufacturers
Health Insurance Companies
Players Mentioned in the Report:
Roche Diagnostics
Abbott Laboratories
Eli Lilly and Company
Biogen Inc.
Quest Diagnostics
Siemens Healthineers
C2N Diagnostics
Quanterix Corporation
Fujirebio
Alzheon Inc.
Genentech
Avid Radiopharmaceuticals
MagQu Co., Ltd.
ADx NeuroSciences
Araclon Biotech
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of Alzheimer's disease
3.1.2 Advancements in blood-based biomarker technologies
3.1.3 Rising demand for early diagnosis and personalized medicine
3.1.4 Growing investments in Alzheimer's research and development
3.2 Market Challenges
3.2.1 High costs associated with biomarker testing
3.2.2 Regulatory hurdles and approval processes
3.2.3 Limited awareness among healthcare professionals
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Development of novel biomarkers
3.3.3 Collaborations with research institutions
3.3.4 Integration of AI and machine learning in diagnostics
3.4 Market Trends
3.4.1 Increasing focus on preventive healthcare
3.4.2 Shift towards home-based testing solutions
3.4.3 Rising consumer demand for transparency in testing
3.4.4 Growth of telemedicine and remote diagnostics
3.5 Government Regulation
3.5.1 Stricter guidelines for biomarker validation
3.5.2 Incentives for research and development
3.5.3 Policies promoting early diagnosis
3.5.4 Regulations on data privacy and patient consent
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Segmentation
8.1 By Type
8.1.1 Protein Biomarkers (e.g., amyloid-beta, tau, neurofilament light chain)
8.1.2 Genetic Biomarkers (e.g., APOE genotyping, other risk alleles)
8.1.3 Metabolomic Biomarkers
8.1.4 Lipid Biomarkers
8.1.5 Multi-marker Panels & Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories (including clinical and commercial labs)
8.2.3 Research Institutions & Academic Centers
8.2.4 Memory Clinics & Senior Care Centers
8.2.5 CROs & Others
8.3 By Sample Type
8.3.1 Whole Blood Samples
8.3.2 Plasma Samples
8.3.3 Serum Samples
8.3.4 Dried Blood Spot & Others
8.4 By Biomarker Class
8.4.1 Amyloid Biomarkers (e.g., A?42/40 ratio)
8.4.2 Tau Biomarkers (e.g., p-tau181, p-tau217, total tau)
8.4.3 Neurodegeneration Biomarkers (e.g., NfL, GFAP)
8.4.4 Inflammatory & Other Biomarkers
8.5 By Distribution Channel
8.5.1 Direct Sales (to hospitals, labs, clinics)
8.5.2 Online Sales (including digital health platforms)
8.5.3 Distributors & Local Partners
8.5.4 Others
8.6 By Region
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa (with focus on Saudi Arabia, UAE, South Africa, Egypt)
8.7 By Research Type
8.7.1 Clinical Research
8.7.2 Preclinical Research
8.7.3 Real-World Evidence & Others
9. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (specific to blood-based AD diagnostics segment)
9.2.4 Market Penetration Rate (MEA and global, % of target population reached)
9.2.5 Regulatory Approvals (number and type of regional/international clearances)
9.2.6 Clinical Validation Metrics (sensitivity, specificity, NPV, PPV of assays)
9.2.7 Product Portfolio Breadth (number of assays, biomarker classes covered)
9.2.8 Strategic Partnerships (number and type: pharma, CROs, academic, local distributors)
9.2.9 R&D Intensity (R&D spend as % of revenue, pipeline assets)
9.2.10 Commercialization Timeline (average time from development to launch in MEA)
9.2.11 Pricing Strategy (premium, value-based, bundled, etc.)
9.2.12 Customer Satisfaction Score (NPS, survey-based metrics)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Roche Diagnostics
9.5.2 Abbott Laboratories
9.5.3 Eli Lilly and Company
9.5.4 Biogen Inc.
9.5.5 Quest Diagnostics
9.5.6 Siemens Healthineers
9.5.7 C2N Diagnostics
9.5.8 Quanterix Corporation
9.5.9 Fujirebio
9.5.10 Alzheon Inc.
9.5.11 Genentech
9.5.12 Avid Radiopharmaceuticals
9.5.13 MagQu Co., Ltd.
9.5.14 ADx NeuroSciences
9.5.15 Araclon Biotech
10. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Alzheimer's Research
10.1.2 Collaboration with Private Sector
10.1.3 Policy Support for Diagnostic Innovations
10.1.4 Training Programs for Healthcare Professionals
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Diagnostic Facilities
10.2.2 Funding for Research Initiatives
10.2.3 Partnerships with Technology Providers
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Accessibility of Diagnostic Tests
10.3.2 Affordability of Biomarker Testing
10.3.3 Awareness of Available Solutions
10.3.4 Integration of New Technologies
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Infrastructure Readiness
10.4.3 Acceptance of New Diagnostic Methods
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Diagnostic Accuracy
10.5.2 Long-term Cost Savings
10.5.3 Expansion into New Markets
10.5.4 Others
11. Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on blood-based biomarkers for Alzheimer's diagnostics
- Review of scientific literature and clinical studies related to Alzheimer's disease biomarkers
- Examination of regulatory guidelines and approvals from health authorities regarding diagnostic tests
Primary Research
- Interviews with leading researchers and clinicians specializing in Alzheimer's diagnostics
- Surveys with laboratory managers and diagnostic test developers in the MEA region
- Field interviews with healthcare professionals involved in Alzheimer's patient care and diagnostics
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including market reports and expert opinions
- Triangulation of quantitative data from surveys with qualitative insights from expert interviews
- Sanity checks through peer reviews and feedback from industry stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on healthcare expenditure in the MEA region
- Segmentation of the market by diagnostic test types and patient demographics
- Incorporation of trends in Alzheimer's disease prevalence and awareness initiatives
Bottom-up Modeling
- Collection of sales data from key diagnostic companies operating in the MEA market
- Estimation of market share based on product offerings and regional presence
- Volume and pricing analysis of blood-based biomarker tests for Alzheimer's diagnostics
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as aging population and healthcare policy changes
- Scenario modeling based on potential advancements in biomarker research and technology
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 100 | Laboratory Directors, Diagnostic Test Managers |
| Healthcare Providers | 60 | Neurologists, Geriatricians |
| Biotechnology Firms | 50 | Product Development Managers, R&D Scientists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Patient Advocacy Groups | 45 | Advocacy Leaders, Community Outreach Coordinators |
Frequently Asked Questions
What is the current value of the Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market?
The Global MEA Blood-Based Biomarker Alzheimer's Disease Diagnostics Market is valued at approximately USD 6.2 million, reflecting a significant growth driven by the increasing prevalence of Alzheimer's disease and advancements in biomarker research.