About the Report
Base Year 2024Global Liquid Biopsy Early Cancer Detection Monitoring Market Overview
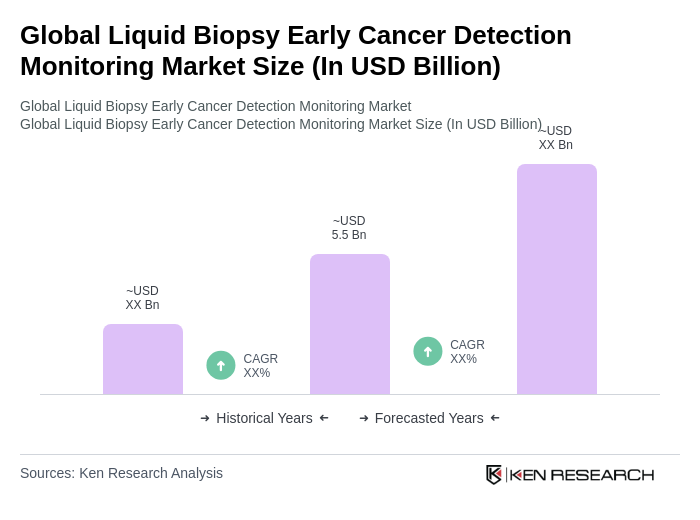
- The Global Liquid Biopsy Early Cancer Detection Monitoring Market is valued at USD 5.5 billion, based on a five-year historical analysis. This growth is primarily driven by continuous technological advancements in cancer diagnostics, the increasing global prevalence of cancer, and the rising demand for non-invasive diagnostic methods. The market is experiencing robust research and development activity focused on improving the precision, sensitivity, and clinical utility of liquid biopsy tests, particularly in early cancer detection and personalized medicine applications .
- Key players in this market include the United States, Germany, and Japan, which lead due to their advanced healthcare infrastructure, substantial investments in research and development, and high adoption rates of innovative diagnostic technologies. The presence of major biotechnology companies and a strong emphasis on precision oncology further reinforce their market dominance .
- The U.S. Food and Drug Administration (FDA) issued the “Breakthrough Devices Program Guidance” in 2022, streamlining the approval process for innovative liquid biopsy tests. This regulation, published by the Center for Devices and Radiological Health (CDRH), facilitates expedited review for devices that offer more effective diagnosis or treatment of life-threatening diseases, thereby enhancing patient access to advanced diagnostic tools while maintaining rigorous safety and efficacy standards .
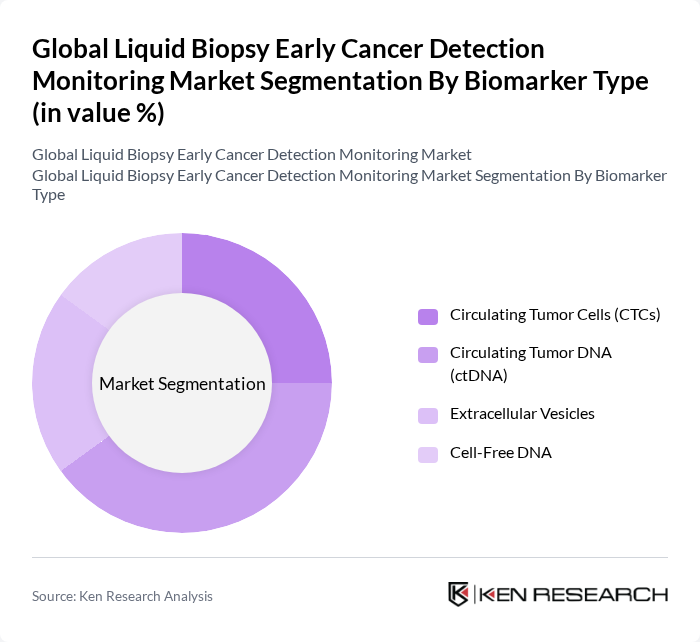
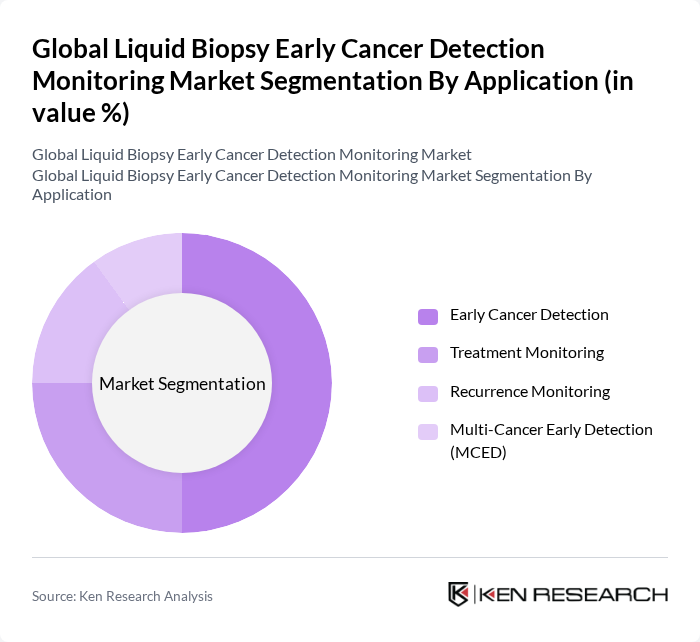
Global Liquid Biopsy Early Cancer Detection Monitoring Market Segmentation
By Biomarker Type:The biomarker type segmentation includes Circulating Tumor Cells (CTCs), Circulating Tumor DNA (ctDNA), Extracellular Vesicles, and Cell-Free DNA. Among these, Circulating Tumor DNA (ctDNA) is currently the leading sub-segment due to its high sensitivity and specificity for early-stage cancer detection. The growing preference for non-invasive testing and the expanding role of personalized medicine are driving demand for ctDNA-based assays. Recent advancements in next-generation sequencing (NGS) and digital PCR technologies have further improved the analytical performance and clinical adoption of ctDNA analysis, making it the preferred choice for clinicians managing early cancer detection and monitoring .
By Application:The application segmentation encompasses Early Cancer Detection, Treatment Monitoring, Recurrence Monitoring, and Multi-Cancer Early Detection (MCED). Early Cancer Detection is the dominant application segment, underpinned by the increasing global focus on preventive healthcare and the critical importance of early diagnosis in improving cancer outcomes. The rising incidence of cancer and the proliferation of multi-cancer early detection (MCED) technologies have further accelerated demand for liquid biopsy solutions in this segment. Additionally, technological progress in multiplexed biomarker detection and integration with artificial intelligence is enabling the simultaneous detection of multiple cancer types, reinforcing the central role of early detection applications .
Global Liquid Biopsy Early Cancer Detection Monitoring Market Competitive Landscape
The Global Liquid Biopsy Early Cancer Detection Monitoring Market is characterized by a dynamic mix of regional and international players. Leading participants such as Guardant Health, Inc., Foundation Medicine, Inc., Exact Sciences Corporation, Natera, Inc., Roche Diagnostics, Illumina, Inc., GRAIL, Inc., Thermo Fisher Scientific, Bio-Rad Laboratories, NeoGenomics Laboratories, Freenome Holdings, Inc., Myriad Genetics, Inc., QIAGEN N.V., Sysmex Corporation, Abbott Laboratories contribute to innovation, geographic expansion, and service delivery in this space.
Global Liquid Biopsy Early Cancer Detection Monitoring Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer:The World Health Organization reported that cancer cases are projected to rise to 30 million by future, up from 19.3 million in 2020. This alarming trend is driving the demand for innovative diagnostic solutions, including liquid biopsies. As healthcare systems strive to manage this growing burden, the need for early detection methods becomes critical, propelling the liquid biopsy market forward. The increasing incidence of lung, breast, and colorectal cancers particularly fuels this demand.
- Advancements in Liquid Biopsy Technologies:Significant technological advancements have been made in liquid biopsy methodologies, enhancing their sensitivity and specificity. For instance, the development of next-generation sequencing (NGS) has improved the detection of circulating tumor DNA (ctDNA). According to a report by MarketsandMarkets, the global liquid biopsy market is expected to reach $5 billion by future, driven by these innovations. Such advancements are crucial for improving diagnostic accuracy and patient outcomes, further stimulating market growth.
- Rising Demand for Non-Invasive Diagnostic Methods:The shift towards non-invasive diagnostic techniques is gaining momentum, as patients and healthcare providers seek alternatives to traditional biopsies. A study published in the Journal of Clinical Oncology indicated that 75% of patients prefer non-invasive testing methods. This preference is supported by the increasing availability of liquid biopsy tests, which offer a safer and more comfortable option for cancer detection. The growing acceptance of these methods is a key driver for market expansion.
Market Challenges
- High Costs Associated with Liquid Biopsy Tests:The cost of liquid biopsy tests can be a significant barrier to widespread adoption. For example, comprehensive liquid biopsy panels can range from $1,200 to $3,500 per test, making them less accessible for many patients. This high cost is compounded by the limited insurance coverage for such tests, which can deter healthcare providers from recommending them. As a result, the financial burden remains a challenge for market growth.
- Regulatory Hurdles and Approval Processes:The regulatory landscape for liquid biopsy tests is complex and can delay market entry. The U.S. FDA has stringent guidelines for the approval of diagnostic tests, which can take several years. For instance, the approval process for a new liquid biopsy test can involve extensive clinical trials and data submissions, often leading to increased costs and time-to-market. These regulatory challenges can hinder innovation and slow the adoption of new technologies in the market.
Global Liquid Biopsy Early Cancer Detection Monitoring Market Future Outlook
The future of the liquid biopsy market appears promising, driven by ongoing technological advancements and a growing emphasis on preventive healthcare. As healthcare systems increasingly prioritize early cancer detection, liquid biopsies are likely to become integral to routine diagnostics. Furthermore, the integration of artificial intelligence in data analysis is expected to enhance diagnostic accuracy and efficiency. These trends indicate a robust growth trajectory for the market, with significant potential for innovation and improved patient outcomes in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets present significant growth opportunities for liquid biopsy companies. With rising healthcare expenditures and increasing cancer awareness, countries like India and Brazil are investing in advanced diagnostic technologies. The World Bank projects that healthcare spending in these regions will grow by 12% annually, creating a favorable environment for liquid biopsy adoption and market penetration.
- Development of Personalized Medicine Approaches:The trend towards personalized medicine is creating new avenues for liquid biopsy applications. By enabling tailored treatment plans based on individual genetic profiles, liquid biopsies can enhance therapeutic efficacy. The global personalized medicine market is expected to reach $3 trillion by future, indicating a substantial opportunity for liquid biopsy technologies to play a pivotal role in this evolving landscape.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Biomarker Type | Circulating Tumor Cells (CTCs) Circulating Tumor DNA (ctDNA) Extracellular Vesicles Cell-Free DNA |
| By Application | Early Cancer Detection Treatment Monitoring Recurrence Monitoring Multi-Cancer Early Detection (MCED) |
| By End-User | Hospitals Cancer Institutes Diagnostic Laboratories Academic Research Institutes |
| By Product Type | Kits and Reagents Assay Panels Instruments Services |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology | Next-Generation Sequencing (NGS) Digital PCR Microfluidics Immunoassays |
| By Sample Type | Blood Samples Urine Samples Saliva Samples Other Bodily Fluids |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Liquid Biopsy Technologies
Healthcare Providers and Hospitals
Biotechnology and Pharmaceutical Companies
Clinical Laboratories and Diagnostic Centers
Health Insurance Companies
Industry Associations and Advocacy Groups
Players Mentioned in the Report:
Guardant Health, Inc.
Foundation Medicine, Inc.
Exact Sciences Corporation
Natera, Inc.
Roche Diagnostics
Illumina, Inc.
GRAIL, Inc.
Thermo Fisher Scientific
Bio-Rad Laboratories
NeoGenomics Laboratories
Freenome Holdings, Inc.
Myriad Genetics, Inc.
QIAGEN N.V.
Sysmex Corporation
Abbott Laboratories
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Liquid Biopsy Early Cancer Detection Monitoring Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Liquid Biopsy Early Cancer Detection Monitoring Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Liquid Biopsy Early Cancer Detection Monitoring Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cancer
3.1.2 Advancements in liquid biopsy technologies
3.1.3 Rising demand for non-invasive diagnostic methods
3.1.4 Growing awareness and acceptance of early cancer detection
3.2 Market Challenges
3.2.1 High costs associated with liquid biopsy tests
3.2.2 Regulatory hurdles and approval processes
3.2.3 Limited reimbursement policies
3.2.4 Competition from traditional biopsy methods
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Development of personalized medicine approaches
3.3.3 Collaborations with research institutions
3.3.4 Integration of AI and machine learning in diagnostics
3.4 Market Trends
3.4.1 Increasing investment in R&D for liquid biopsy
3.4.2 Shift towards preventive healthcare
3.4.3 Growing partnerships between biotech firms and healthcare providers
3.4.4 Rise of telemedicine and remote monitoring solutions
3.5 Government Regulation
3.5.1 FDA guidelines for liquid biopsy tests
3.5.2 CLIA regulations for laboratory testing
3.5.3 Reimbursement policies for liquid biopsy diagnostics
3.5.4 International standards for quality control in diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Liquid Biopsy Early Cancer Detection Monitoring Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Liquid Biopsy Early Cancer Detection Monitoring Market Segmentation
8.1 By Biomarker Type
8.1.1 Circulating Tumor Cells (CTCs)
8.1.2 Circulating Tumor DNA (ctDNA)
8.1.3 Extracellular Vesicles
8.1.4 Cell-Free DNA
8.2 By Application
8.2.1 Early Cancer Detection
8.2.2 Treatment Monitoring
8.2.3 Recurrence Monitoring
8.2.4 Multi-Cancer Early Detection (MCED)
8.3 By End-User
8.3.1 Hospitals
8.3.2 Cancer Institutes
8.3.3 Diagnostic Laboratories
8.3.4 Academic Research Institutes
8.4 By Product Type
8.4.1 Kits and Reagents
8.4.2 Assay Panels
8.4.3 Instruments
8.4.4 Services
8.5 By Region
8.5.1 North America
8.5.2 Europe
8.5.3 Asia-Pacific
8.5.4 Latin America
8.5.5 Middle East & Africa
8.6 By Technology
8.6.1 Next-Generation Sequencing (NGS)
8.6.2 Digital PCR
8.6.3 Microfluidics
8.6.4 Immunoassays
8.7 By Sample Type
8.7.1 Blood Samples
8.7.2 Urine Samples
8.7.3 Saliva Samples
8.7.4 Other Bodily Fluids
9. Global Liquid Biopsy Early Cancer Detection Monitoring Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Capitalization and Firm Size Classification
9.2.3 Year-over-Year Revenue Growth Rate (%)
9.2.4 Geographic Market Penetration Index
9.2.5 Clinical Test Volume and Customer Retention Rate
9.2.6 Average Test Pricing and Reimbursement Rate
9.2.7 FDA Approvals and Regulatory Clearances Count
9.2.8 Test Development and Commercialization Time-to-Market
9.2.9 Research Partnerships and Collaborations Index
9.2.10 Clinical Sensitivity and Specificity Metrics
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Guardant Health, Inc.
9.5.2 Foundation Medicine, Inc.
9.5.3 Exact Sciences Corporation
9.5.4 Natera, Inc.
9.5.5 Roche Diagnostics
9.5.6 Illumina, Inc.
9.5.7 GRAIL, Inc.
9.5.8 Thermo Fisher Scientific
9.5.9 Bio-Rad Laboratories
9.5.10 NeoGenomics Laboratories
9.5.11 Freenome Holdings, Inc.
9.5.12 Myriad Genetics, Inc.
9.5.13 QIAGEN N.V.
9.5.14 Sysmex Corporation
9.5.15 Abbott Laboratories
10. Global Liquid Biopsy Early Cancer Detection Monitoring Market End-User Analysis
10.1 Procurement Behavior of Key Institutions
10.1.1 Government funding and reimbursement models for cancer diagnostics
10.1.2 Collaboration between healthcare institutions and diagnostic providers
10.1.3 Adoption drivers for innovative liquid biopsy solutions
10.2 Healthcare System Spending on Diagnostic Infrastructure
10.2.1 Investment in liquid biopsy testing platforms and equipment
10.2.2 Budget allocation for cancer early detection programs
10.2.3 Strategic partnerships with molecular diagnostics firms
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to comprehensive, validated diagnostic assays
10.3.2 Cost barriers and reimbursement uncertainty for liquid biopsy tests
10.3.3 Need for rapid turnaround times and actionable clinical insights
10.4 User Readiness for Adoption
10.4.1 Clinical awareness and acceptance of liquid biopsy benefits
10.4.2 Training and certification of healthcare professionals in test interpretation
10.4.3 Laboratory infrastructure readiness and regulatory compliance capabilities
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Clinical and economic outcomes from early cancer detection programs
10.5.2 Expansion of testing capabilities across multiple cancer types
10.5.3 Integration of liquid biopsy into standard treatment protocols
11. Global Liquid Biopsy Early Cancer Detection Monitoring Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Key Partnerships
1.5 Cost Structure Analysis
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Hospital and Cancer Institute Direct Sales
3.2 Diagnostic Laboratory Partnerships
3.3 Online Test Ordering and Direct-to-Consumer Channels
3.4 Partnerships with Healthcare Providers
4. Channel & Pricing Gaps
4.1 Underserved Geographic and Clinical Segments
4.2 Pricing Band Analysis for Liquid Biopsy Tests
4.3 Competitor Pricing Strategies
4.4 Healthcare System and Patient Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Clinical Application Gaps
5.2 Healthcare Provider Segments Analysis
5.2.1 Oncology Centers
5.2.2 Primary Care Facilities
5.2.3 Emerging Market Healthcare Systems
5.3 Emerging Trends in Personalized Cancer Diagnostics
6. Customer Relationship
6.1 Physician Education and Clinical Support Programs
6.2 After-Test Support and Result Interpretation Services
6.3 Customer Feedback and Clinical Evidence Building Mechanisms
7. Value Proposition
7.1 Clinical Accuracy and Sensitivity/Specificity Advantages
7.2 Integrated Care Pathways and Real-Time Monitoring
7.3 Personalized and Precision Oncology Solutions
8. Key Activities
8.1 Regulatory Compliance and FDA Clearance Management
8.2 Clinical Evidence Generation and Publication Strategy
8.3 Distribution Network and Laboratory Partnership Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Test Portfolio Development for High-Incidence Cancer Types
9.1.2 Reimbursement and Pricing Band Strategy
9.1.3 Marketing and Clinical Education Initiatives
9.2 Export Entry Strategy
9.2.1 Priority Markets Identification (High-Growth Asia-Pacific, Europe)
9.2.2 Regulatory Pathway Development for Major Markets
10. Entry Mode Assessment
10.1 Joint Ventures with Established Diagnostic Providers
10.2 Greenfield Investments in Core Markets
10.3 Mergers & Acquisitions of Complementary Technologies
10.4 Laboratory and Distribution Partner Network Model
11. Capital and Timeline Estimation
11.1
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on liquid biopsy technologies
- Review of scientific journals and articles focusing on early cancer detection methodologies
- Examination of regulatory frameworks and guidelines from health authorities regarding liquid biopsy applications
Primary Research
- Interviews with oncologists and pathologists specializing in cancer diagnostics
- Surveys with laboratory managers and technicians involved in liquid biopsy testing
- Focus groups with patients and healthcare providers to understand perceptions and acceptance of liquid biopsy
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from clinical studies, market trends, and expert opinions
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure on cancer diagnostics
- Segmentation of the market by geographical regions and cancer types
- Incorporation of trends in personalized medicine and non-invasive testing methods
Bottom-up Modeling
- Collection of data on the number of liquid biopsy tests performed annually across various regions
- Estimation of average pricing for liquid biopsy tests based on laboratory pricing structures
- Calculation of market size using volume of tests multiplied by average test price
Forecasting & Scenario Analysis
- Utilization of historical growth rates to project future market trends through 2030
- Scenario analysis based on advancements in technology and changes in regulatory landscapes
- Development of best-case, worst-case, and most-likely market scenarios
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinics | 100 | Oncologists, Nurse Practitioners |
| Diagnostic Laboratories | 80 | Laboratory Directors, Laboratory Technicians |
| Healthcare Providers | 60 | General Practitioners, Family Physicians |
| Patient Advocacy Groups | 40 | Patient Advocates, Community Health Workers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Global Liquid Biopsy Early Cancer Detection Monitoring Market?
The Global Liquid Biopsy Early Cancer Detection Monitoring Market is valued at approximately USD 5.5 billion, driven by advancements in cancer diagnostics and the increasing demand for non-invasive testing methods.