About the Report
Base Year 2024Global Ventral Hernia Mesh Devices Market Overview
- The Global Ventral Hernia Mesh Devices Market is valued at USD 1 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of ventral and incisional hernias associated with obesity, aging populations, prior abdominal surgeries, and lifestyle risk factors. Advancements in surgical techniques, particularly laparoscopic and robotic-assisted ventral hernia repair, and the rising adoption of minimally invasive procedures are accelerating mesh utilization. The demand for innovative mesh materials and designs, including lightweight, macroporous, composite, and partially absorbable meshes, has also contributed significantly to market expansion as healthcare providers seek to improve patient outcomes, lower recurrence rates, and reduce recovery times.
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany being among the most dominant countries in the hernia mesh devices landscape. The U.S. benefits from a well-established healthcare infrastructure, high healthcare expenditure, high hernia repair volumes, and a strong focus on research and development, which supports rapid adoption of advanced mesh technologies and minimally invasive techniques. Germany’s prominence is attributed to its advanced medical technology sector, strong hospital network, high procedure rates for hernia repair, and a robust regulatory framework under the European Union Medical Device Regulation (EU MDR) that supports innovation and post?market surveillance for medical devices, including hernia mesh.
- In 2023, the U.S. Food and Drug Administration (FDA) continued to regulate ventral hernia mesh devices as Class II medical devices requiring premarket notification through the 510(k) process, with certain higher?risk or novel designs subject to more stringent pre?market review. Under the Federal Food, Drug, and Cosmetic Act and implementing regulations such as 21 CFR Part 807 and 21 CFR Part 820, manufacturers must provide appropriate bench, animal, and where applicable clinical data to demonstrate substantial equivalence, safety, and effectiveness, and must comply with Quality System Regulation, post?market reporting, and adverse event surveillance requirements. These measures reflect a growing emphasis on patient safety, product quality, and long?term performance of mesh implants in the medical device industry.
Global Ventral Hernia Mesh Devices Market Segmentation
By Product Type:The product type segmentation includes various mesh materials designed for hernia repair. The dominant subsegment is Synthetic Mesh, which is widely used due to its durability, favorable long?term outcomes, and effectiveness in preventing hernia recurrence across a broad range of ventral hernia indications. Biologic Mesh is gaining traction, particularly in contaminated fields and complex abdominal wall reconstructions, as it offers better integration with human tissue and reduced risk of chronic infection in selected patients. Composite Mesh combines the benefits of synthetic strength with barrier or anti?adhesion layers designed for intraperitoneal placement, making it important in laparoscopic ventral hernia repair. Bioabsorbable/Partially Absorbable Mesh is also emerging, appealing to patients and surgeons seeking less permanent foreign material load while maintaining initial mechanical support, and is increasingly evaluated in minimally invasive procedures and bridging techniques. The increasing preference for minimally invasive surgeries and complex abdominal wall reconstruction procedures is driving the demand for these differentiated mesh products.

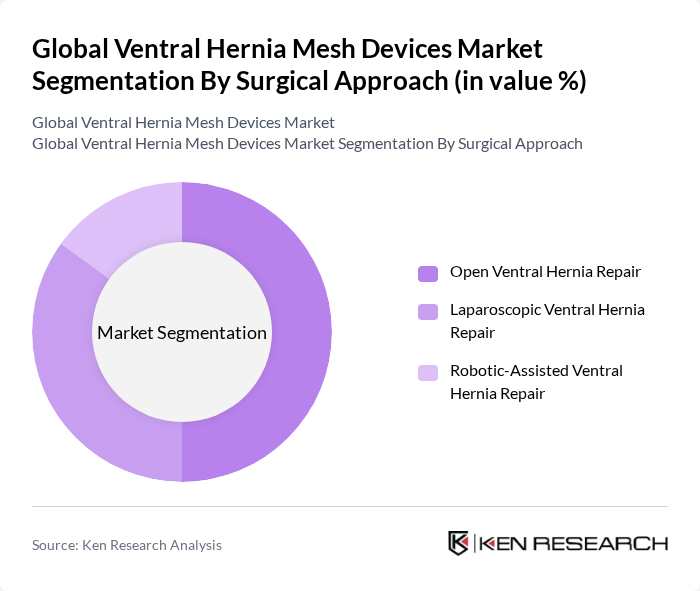
By Surgical Approach:This segmentation highlights the different surgical techniques employed in ventral hernia repair. The Open Ventral Hernia Repair approach remains widely used, particularly for large, complex, or recurrent ventral hernias, and in settings where advanced minimally invasive infrastructure or expertise is limited. However, Laparoscopic Ventral Hernia Repair is rapidly gaining popularity due to its minimally invasive nature, smaller incisions, shorter recovery times, reduced postoperative pain, and lower wound complication rates for appropriately selected defects. Robotic-Assisted Ventral Hernia Repair is also emerging, offering enhanced precision, three?dimensional visualization, and improved suturing and mesh placement capabilities, which is driving its uptake in complex abdominal wall reconstruction and in high?volume centers focused on advanced minimally invasive surgery.
Global Ventral Hernia Mesh Devices Market Competitive Landscape
The Global Ventral Hernia Mesh Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Ethicon Inc. (Johnson & Johnson MedTech), Becton, Dickinson and Company (BD) – C.R. Bard, W. L. Gore & Associates, Inc., B. Braun Melsungen AG, Cook Medical LLC, Stryker Corporation, Integra LifeSciences Holdings Corporation, Medline Industries, LP, HerniaMesh S.r.l., FEG Textiltechnik mbH, Cousin Surgery, Atrium Medical Corporation (Getinge AB), LifeCell (Allergan Aesthetics, an AbbVie company), TELA Bio, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Global Ventral Hernia Mesh Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Ventral Hernias:The global incidence of ventral hernias is estimated to be around 3.5 million cases annually, with a significant rise in cases attributed to lifestyle changes and obesity. According to the World Health Organization, obesity rates have increased by 27% since 2000, leading to a higher demand for surgical interventions. This growing prevalence directly drives the need for ventral hernia mesh devices, as healthcare providers seek effective solutions to manage this condition.
- Advancements in Surgical Techniques:Innovations in surgical techniques, such as laparoscopic and robotic-assisted surgeries, have improved patient outcomes and reduced recovery times. The American College of Surgeons reported that minimally invasive procedures have increased by 40% over the past five years. These advancements not only enhance the effectiveness of hernia repairs but also boost the adoption of specialized mesh devices designed for these modern techniques, further propelling market growth.
- Rising Geriatric Population:The global geriatric population is projected to reach 1.7 billion in future, according to the United Nations. This demographic shift is significant, as older adults are more susceptible to hernias due to weakened muscles and connective tissues. As the population ages, the demand for surgical interventions, including ventral hernia repairs, is expected to rise, driving the market for hernia mesh devices to accommodate this growing patient base.
Market Challenges
- High Cost of Surgical Mesh Devices:The average cost of surgical mesh devices can range from USD 1,200 to USD 3,500 per procedure, which poses a financial burden on healthcare systems and patients. This high cost can limit access to necessary surgical interventions, particularly in low-income regions. As healthcare budgets tighten, the affordability of these devices remains a significant challenge, potentially hindering market growth in various regions.
- Stringent Regulatory Requirements:Regulatory bodies, such as the FDA and European Medicines Agency, impose rigorous testing and approval processes for surgical mesh devices. For instance, the FDA requires extensive clinical data to demonstrate safety and efficacy, which can delay product launches. These stringent regulations can create barriers for manufacturers, limiting innovation and market entry, particularly for smaller companies with fewer resources to navigate complex regulatory landscapes.
Global Ventral Hernia Mesh Devices Market Future Outlook
The future of the ventral hernia mesh devices market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centered care. As healthcare systems invest in innovative surgical solutions, the integration of smart technologies and bio-compatible materials is expected to enhance surgical outcomes. Additionally, the rise in outpatient procedures will likely lead to a greater demand for effective and efficient hernia repair solutions, positioning the market for sustained growth in the coming years.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets, particularly in Asia and Africa, present significant growth opportunities due to increasing healthcare investments. The World Bank estimates that healthcare spending in these regions will grow by 10% annually, creating demand for advanced surgical solutions, including ventral hernia mesh devices, as healthcare infrastructure improves and access to surgical care expands.
- Development of Bio-Compatible Materials:The ongoing research into bio-compatible materials for surgical mesh devices is a key opportunity. Innovations in materials science are leading to the development of meshes that reduce the risk of complications and improve patient outcomes. As these materials become commercially viable, they will likely attract investment and drive market growth, enhancing the safety and effectiveness of hernia repairs.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Synthetic Mesh Biologic Mesh Composite Mesh Bioabsorbable / Partially Absorbable Mesh |
| By Surgical Approach | Open Ventral Hernia Repair Laparoscopic Ventral Hernia Repair Robotic-Assisted Ventral Hernia Repair |
| By Material | Polypropylene Polyester Polytetrafluoroethylene (PTFE) ePTFE and Composite Barrier Mesh |
| By Ventral Hernia Type / Indication | Incisional Ventral Hernia Umbilical Ventral Hernia Epigastric Ventral Hernia Parastomal Ventral Hernia Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Hernia Centers / Clinics Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Distribution Channel | Direct Sales to Hospitals & ASCs Third-Party Distributors Group Purchasing Organizations (GPOs) Online / E-procurement Platforms |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Regulatory Consultants
Industry Associations (e.g., American Hernia Society)
Financial Institutions
Players Mentioned in the Report:
Medtronic plc
Ethicon Inc. (Johnson & Johnson MedTech)
Becton, Dickinson and Company (BD) C.R. Bard
W. L. Gore & Associates, Inc.
B. Braun Melsungen AG
Cook Medical LLC
Stryker Corporation
Integra LifeSciences Holdings Corporation
Medline Industries, LP
HerniaMesh S.r.l.
FEG Textiltechnik mbH
Cousin Surgery
Atrium Medical Corporation (Getinge AB)
LifeCell (Allergan Aesthetics, an AbbVie company)
TELA Bio, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Ventral Hernia Mesh Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Ventral Hernia Mesh Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Ventral Hernia Mesh Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of ventral hernias
3.1.2 Advancements in surgical techniques
3.1.3 Rising geriatric population
3.1.4 Growing demand for minimally invasive surgeries
3.2 Market Challenges
3.2.1 High cost of surgical mesh devices
3.2.2 Stringent regulatory requirements
3.2.3 Risk of complications and product recalls
3.2.4 Limited awareness in developing regions
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Development of bio-compatible materials
3.3.3 Increasing investment in healthcare infrastructure
3.3.4 Collaborations and partnerships for innovation
3.4 Market Trends
3.4.1 Shift towards absorbable mesh materials
3.4.2 Integration of smart technologies in surgical devices
3.4.3 Focus on patient-centric solutions
3.4.4 Rise in outpatient surgical procedures
3.5 Government Regulation
3.5.1 FDA guidelines on surgical mesh devices
3.5.2 CE marking requirements in Europe
3.5.3 Post-market surveillance regulations
3.5.4 Reimbursement policies for hernia repair surgeries
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Ventral Hernia Mesh Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Ventral Hernia Mesh Devices Market Segmentation
8.1 By Product Type
8.1.1 Synthetic Mesh
8.1.2 Biologic Mesh
8.1.3 Composite Mesh
8.1.4 Bioabsorbable / Partially Absorbable Mesh
8.2 By Surgical Approach
8.2.1 Open Ventral Hernia Repair
8.2.2 Laparoscopic Ventral Hernia Repair
8.2.3 Robotic-Assisted Ventral Hernia Repair
8.3 By Material
8.3.1 Polypropylene
8.3.2 Polyester
8.3.3 Polytetrafluoroethylene (PTFE)
8.3.4 ePTFE and Composite Barrier Mesh
8.4 By Ventral Hernia Type / Indication
8.4.1 Incisional Ventral Hernia
8.4.2 Umbilical Ventral Hernia
8.4.3 Epigastric Ventral Hernia
8.4.4 Parastomal Ventral Hernia
8.4.5 Others
8.5 By End-User
8.5.1 Hospitals
8.5.2 Ambulatory Surgical Centers
8.5.3 Specialty Hernia Centers / Clinics
8.5.4 Others
8.6 By Region
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa
8.7 By Distribution Channel
8.7.1 Direct Sales to Hospitals & ASCs
8.7.2 Third-Party Distributors
8.7.3 Group Purchasing Organizations (GPOs)
8.7.4 Online / E-procurement Platforms
9. Global Ventral Hernia Mesh Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Global Hernia Mesh Devices Revenue (USD Million)
9.2.4 Ventral Hernia Mesh Devices Revenue Share (%)
9.2.5 3-Year Revenue CAGR in Ventral Hernia Mesh Segment
9.2.6 Regional Revenue Mix (North America / Europe / Asia-Pacific / RoW)
9.2.7 Product Portfolio Breadth (Number of Ventral Mesh SKUs)
9.2.8 Share of Advanced/Bioabsorbable & Composite Mesh in Portfolio (%)
9.2.9 R&D Intensity (% of Revenue Spent on R&D)
9.2.10 Average Selling Price Positioning (Premium / Mid / Value)
9.2.11 Key Distribution Model (Direct vs Distributor vs GPO-driven)
9.2.12 Key Regulatory Approvals (FDA 510(k), CE Mark, etc.)
9.2.13 Reported Mesh-Related Adverse Event / Recall History
9.2.14 Strategic Partnerships & M&A Activity in Hernia Repair
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Ethicon Inc. (Johnson & Johnson MedTech)
9.5.3 Becton, Dickinson and Company (BD) – C.R. Bard
9.5.4 W. L. Gore & Associates, Inc.
9.5.5 B. Braun Melsungen AG
9.5.6 Cook Medical LLC
9.5.7 Stryker Corporation
9.5.8 Integra LifeSciences Holdings Corporation
9.5.9 Medline Industries, LP
9.5.10 HerniaMesh S.r.l.
9.5.11 FEG Textiltechnik mbH
9.5.12 Cousin Surgery
9.5.13 Atrium Medical Corporation (Getinge AB)
9.5.14 LifeCell (Allergan Aesthetics, an AbbVie company)
9.5.15 TELA Bio, Inc.
10. Global Ventral Hernia Mesh Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Facilities
10.2.2 Spending on Surgical Equipment
10.2.3 Budgeting for Training and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Surgical Complications
10.3.2 Cost Constraints
10.3.3 Availability of Skilled Personnel
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs Assessment
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Surgical Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Patient Feedback Mechanisms
10.5.4 Others
11. Global Ventral Hernia Mesh Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from medical device regulatory bodies and industry associations
- Review of published articles in medical journals focusing on ventral hernia repair techniques and mesh materials
- Examination of sales data and market trends from leading manufacturers in the ventral hernia mesh devices sector
Primary Research
- Interviews with surgeons specializing in hernia repair to understand preferences and trends in mesh selection
- Surveys with hospital procurement managers to gauge purchasing patterns and budget allocations
- Field interviews with medical device distributors to assess market dynamics and customer feedback
Validation & Triangulation
- Cross-validation of findings through multiple data sources including clinical studies and market surveys
- Triangulation of insights from primary interviews with secondary data to ensure consistency
- Sanity checks conducted through expert panel reviews comprising industry veterans and healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the global market size based on healthcare expenditure and surgical procedure volumes
- Segmentation of the market by product type, including absorbable and non-absorbable meshes
- Incorporation of regional healthcare policies and reimbursement frameworks affecting mesh usage
Bottom-up Modeling
- Collection of sales data from key manufacturers to establish baseline market volumes
- Estimation of average selling prices for different types of ventral hernia mesh devices
- Calculation of market size based on unit sales multiplied by average prices across regions
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating demographic trends, surgical rates, and technological advancements
- Scenario modeling based on potential changes in healthcare regulations and market entry of new products
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Surgeon Insights on Mesh Selection | 120 | General Surgeons, Plastic Surgeons |
| Hospital Procurement Strategies | 90 | Procurement Managers, Supply Chain Directors |
| Distributor Perspectives on Market Trends | 70 | Medical Device Distributors, Sales Representatives |
| Patient Feedback on Hernia Repair Outcomes | 110 | Post-operative Patients, Patient Advocacy Groups |
| Regulatory Insights from Healthcare Authorities | 60 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Global Ventral Hernia Mesh Devices Market?
The Global Ventral Hernia Mesh Devices Market is valued at approximately USD 1 billion, reflecting a significant growth driven by the increasing prevalence of ventral hernias, advancements in surgical techniques, and the rising adoption of minimally invasive procedures.