About the Report
Base Year 2024Saudi Arabia Ventral Hernia Mesh Devices Market Overview
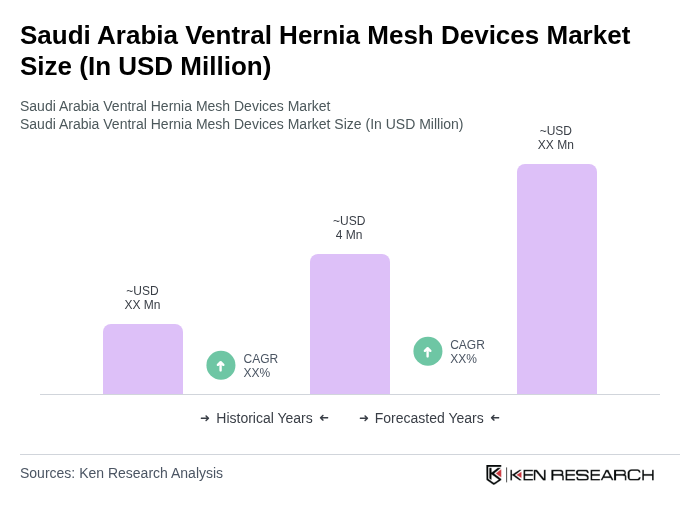
- The Saudi Arabia Ventral Hernia Mesh Devices Market is valued at approximately USD 4 million, based on a five-year historical analysis and recent country-level revenue estimates. This growth is primarily driven by the increasing prevalence of hernia cases, rising obesity and aging population, advancements in mesh materials (including lightweight composite and partially absorbable meshes), and the growing adoption of minimally invasive and laparoscopic procedures in Saudi Arabia and the wider Middle East. The market is also supported by ongoing expansion of healthcare infrastructure under Saudi Vision 2030, higher surgical volumes in tertiary hospitals, and greater access to specialized general and bariatric surgeons.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare facilities, higher concentration of tertiary and specialist hospitals, and greater availability of surgeons trained in laparoscopic and robotic hernia repair. Riyadh, being the capital, has a higher number of healthcare institutions and major referral centers, while Jeddah and Dammam serve as leading healthcare hubs in the western and eastern regions, respectively, attracting patients from surrounding provinces for complex abdominal wall and ventral hernia surgeries.
- In 2023, the Saudi Food and Drug Authority (SFDA) strengthened the regulatory framework for medical devices through instruments such as the Medical Devices Interim Regulation issued by the Saudi Food and Drug Authority and related implementing rules, which require medical devices, including hernia mesh products, to be registered in the SFDA Medical Devices Marketing Authorization system and supported by clinical evidence appropriate to the device risk class. These requirements cover conformity assessment, post?marketing surveillance, and quality management system compliance (aligned with international standards), aiming to enhance patient safety and ensure that only high?quality products are available in the market, thereby fostering trust in surgical interventions and imported hernia mesh technologies.
Saudi Arabia Ventral Hernia Mesh Devices Market Segmentation
By Mesh Type:The market is segmented into various mesh types, including Permanent Synthetic Mesh (e.g., Polypropylene, ePTFE), Partially Absorbable Composite Mesh, Fully Resorbable / Bioabsorbable Mesh, Biological Mesh (Allograft and Xenograft), 3D / Pre-shaped and Anatomical Mesh, and Others. Among these, Permanent Synthetic Mesh is the most widely used due to its durability, tensile strength, and long clinical track record in ventral and incisional hernia repair, in line with global practice where non?resorbable synthetic meshes account for the largest share of ventral hernia mesh procedures.
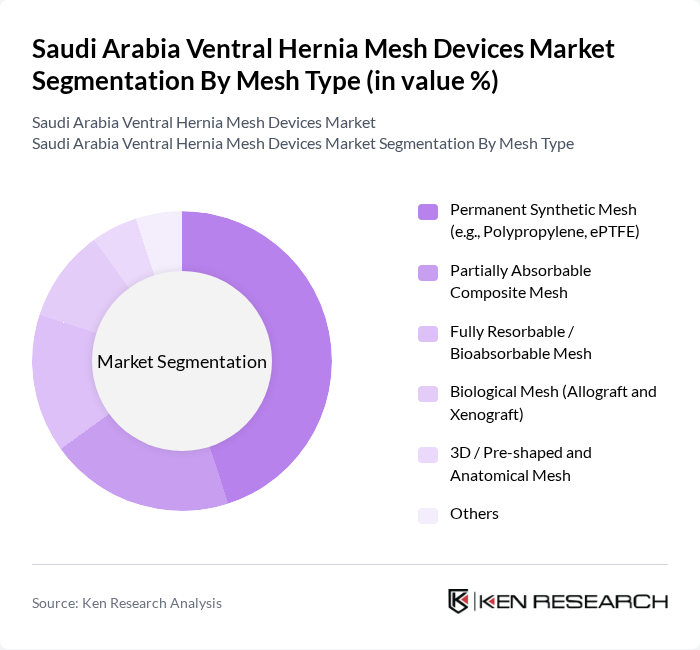
The Permanent Synthetic Mesh segment dominates the market due to its widespread acceptance among surgeons and patients alike. This type of mesh is favored for its strength, resistance to mechanical failure, and long-term performance, making it suitable for a broad range of ventral, umbilical, and incisional hernia repairs. The increasing number of abdominal surgeries linked to obesity, prior laparotomies, and bariatric procedures, combined with the preference for durable materials with predictable outcomes, contributes to its leading position. Additionally, advancements in manufacturing techniques—such as lightweight, macroporous polypropylene, composite meshes with anti?adhesive barriers, and improved sterilization and quality control—have enhanced the biocompatibility and safety profile of synthetic meshes, further driving their adoption in clinical settings.
By End-User:The market is segmented by end-user into Government Hospitals (MOH and Other Public Hospitals), Private Hospitals, Specialized Surgical & Day-Care Centers, University & Teaching Hospitals, and Others. Government Hospitals are the leading end-users due to their extensive patient base, large procedural volumes, and provision of publicly funded or subsidized healthcare services, which is consistent with broader hernia mesh utilization patterns where hospitals and large surgical centers account for the majority of mesh implants.
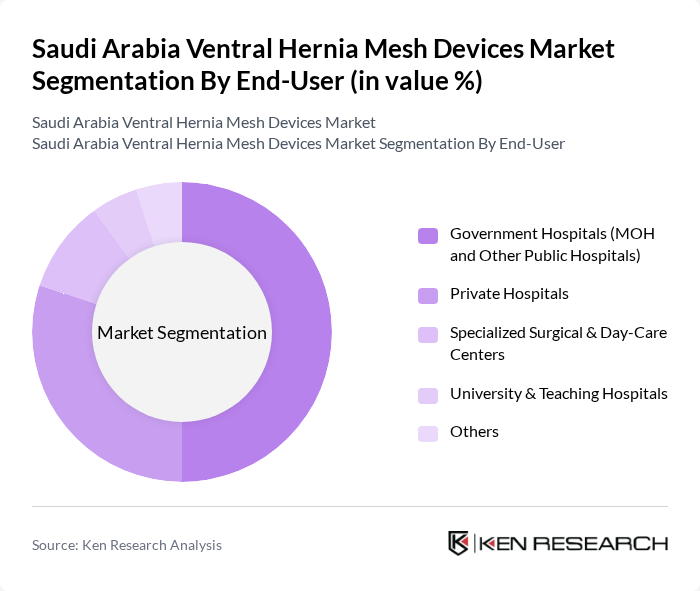
Government Hospitals are the primary end-users of ventral hernia mesh devices, accounting for a significant share of the market. This is largely due to the high volume of elective and emergency hernia surgeries performed in Ministry of Health and other public sector facilities, which cater to a broad segment of the population, including patients referred from peripheral regions. The availability of centralized funding, bulk procurement mechanisms, and investment in modern operating theatres and laparoscopic equipment in these hospitals allows for the adoption of advanced mesh technologies, further solidifying their position as market leaders. Additionally, the increasing focus on national health programs, surgical backlog reduction, and enhanced access to elective procedures under Saudi Vision 2030 supports growth in this segment.
Saudi Arabia Ventral Hernia Mesh Devices Market Competitive Landscape
The Saudi Arabia Ventral Hernia Mesh Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Ethicon Inc. (Johnson & Johnson MedTech), Becton, Dickinson and Company (BD) – C.R. Bard, W. L. Gore & Associates, Inc., B. Braun Melsungen AG, Cook Medical LLC, Integra LifeSciences Holdings Corporation, Medline Industries, LP, ConvaTec Group plc, LifeCell / Allergan Aesthetics (AbbVie Inc.), Herniamesh S.r.l., C.R. Bard (Legacy Brand Portfolio under BD), Surgical Specialties Corporation, Atrium Medical (Getinge AB), Local & Regional Distributors (e.g., Abu Dawood Medical, Tamer Group, Cigalah Group) contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Ventral Hernia Mesh Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Ventral Hernias:The prevalence of ventral hernias in Saudi Arabia is estimated to be around 5% of the population, translating to approximately 1.5 million individuals affected. This growing incidence is primarily attributed to lifestyle factors, obesity rates, and an aging population. The World Health Organization reported that obesity rates in Saudi Arabia are among the highest in the region, with adult obesity prevalence estimated at roughly one-third of the population, significantly contributing to the rise in hernia cases, thereby driving demand for mesh devices.
- Advancements in Surgical Techniques:The adoption of advanced surgical techniques, such as laparoscopic surgery, has increased significantly, with a substantial share of hernia repairs now performed using minimally invasive methods. This shift is supported by the Saudi Ministry of Health's initiatives to enhance surgical training and technology. The integration of these techniques has improved patient outcomes, reduced recovery times, and increased the demand for specialized mesh devices, further propelling market growth.
- Rising Geriatric Population:The geriatric population in Saudi Arabia is projected to increase notably in future, representing a significant demographic shift, although it remains a relatively small share of the total population compared with many high?income countries. This age group is more susceptible to ventral hernias due to factors such as weakened muscles and comorbidities. The Saudi government is investing in healthcare services for the elderly, which includes enhancing surgical options and access to hernia mesh devices, thereby driving market expansion in this segment.
Market Challenges
- High Cost of Advanced Mesh Devices:The cost of advanced ventral hernia mesh devices can range from SAR 2,000 to SAR 5,000 per unit, which poses a significant barrier to widespread adoption. Many healthcare facilities in Saudi Arabia face budget constraints, limiting their ability to procure these devices. This financial challenge is exacerbated by the limited availability of cost-effective alternatives, hindering market growth and accessibility for patients.
- Stringent Regulatory Requirements:The regulatory landscape for medical devices in Saudi Arabia is becoming increasingly stringent, with the Saudi Food and Drug Authority (SFDA) implementing rigorous approval processes aligned with international standards such as the Medical Devices Regulation framework. This includes requirements for clinical evidence and comprehensive documentation, which can delay product launches. Such regulatory hurdles can deter manufacturers from entering the market, limiting competition and innovation in the ventral hernia mesh segment.
Saudi Arabia Ventral Hernia Mesh Devices Market Future Outlook
The future of the ventral hernia mesh devices market in Saudi Arabia appears promising, driven by technological advancements and demographic shifts. The integration of digital technologies in surgical procedures is expected to enhance precision and patient outcomes. Additionally, the growing preference for outpatient surgical procedures will likely increase the demand for innovative mesh solutions. As healthcare infrastructure expands, opportunities for product customization and innovation will further support market growth, ensuring better patient care and accessibility.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Saudi government has committed substantial investments in healthcare infrastructure as part of broader national transformation initiatives, with total planned health sector spending over the medium term amounting to tens of billions in Saudi riyals, aiming to improve access to surgical services. This expansion will create opportunities for ventral hernia mesh device manufacturers to establish partnerships with hospitals and clinics, enhancing market penetration and accessibility for patients requiring surgical interventions.
- Potential for Product Innovation:There is a growing demand for customized and bioabsorbable mesh products in surgical care globally, including in Gulf Cooperation Council markets, as healthcare providers increasingly adopt advanced biomaterials to improve outcomes and reduce complications. This trend presents significant opportunities for manufacturers to innovate and develop tailored solutions that meet the specific needs of patients and healthcare providers, driving market growth and improving surgical outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Mesh Type | Permanent Synthetic Mesh (e.g., Polypropylene, ePTFE) Partially Absorbable Composite Mesh Fully Resorbable / Bioabsorbable Mesh Biological Mesh (Allograft and Xenograft) D / Pre-shaped and Anatomical Mesh Others |
| By End-User | Government Hospitals (MOH and Other Public Hospitals) Private Hospitals Specialized Surgical & Day-Care Centers University & Teaching Hospitals Others |
| By Material | Polypropylene Mesh Expanded Polytetrafluoroethylene (ePTFE) Mesh Polyester Mesh Biological (Human and Porcine / Bovine Derived) Mesh Hybrid / Composite Mesh Others |
| By Surgical Approach | Open Ventral Hernia Repair Laparoscopic Ventral Hernia Repair Robotic Ventral Hernia Repair Others |
| By Region | Central Region (Including Riyadh) Western Region (Including Makkah, Madinah, Jeddah) Eastern Region (Including Dammam, Al Khobar) Southern Region Northern Region |
| By Distribution Channel | Direct Sales to Hospitals Local Distributors & Importers Group Purchasing Organizations (GPOs) & Tender-Based Procurement Online & E-Procurement Platforms Others |
| By Clinical Indication | Primary Ventral Hernia Incisional Hernia Umbilical & Epigastric Hernia Complex Abdominal Wall Reconstruction Recurrent Ventral Hernia Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations (e.g., Saudi Medical Devices Association)
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Ethicon Inc. (Johnson & Johnson MedTech)
Becton, Dickinson and Company (BD) C.R. Bard
W. L. Gore & Associates, Inc.
B. Braun Melsungen AG
Cook Medical LLC
Integra LifeSciences Holdings Corporation
Medline Industries, LP
ConvaTec Group plc
LifeCell / Allergan Aesthetics (AbbVie Inc.)
Herniamesh S.r.l.
C.R. Bard (Legacy Brand Portfolio under BD)
Surgical Specialties Corporation
Atrium Medical (Getinge AB)
Local & Regional Distributors (e.g., Abu Dawood Medical, Tamer Group, Cigalah Group)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Ventral Hernia Mesh Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Ventral Hernia Mesh Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Ventral Hernia Mesh Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of ventral hernias
3.1.2 Advancements in surgical techniques
3.1.3 Rising geriatric population
3.1.4 Growing awareness about minimally invasive surgeries
3.2 Market Challenges
3.2.1 High cost of advanced mesh devices
3.2.2 Stringent regulatory requirements
3.2.3 Risk of complications associated with mesh implants
3.2.4 Limited reimbursement policies
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in medical technology
3.3.3 Rising demand for outpatient surgical procedures
3.3.4 Potential for product innovation and customization
3.4 Market Trends
3.4.1 Shift towards bioabsorbable mesh materials
3.4.2 Integration of digital technologies in surgical procedures
3.4.3 Growing preference for robotic-assisted surgeries
3.4.4 Increasing focus on patient-centric healthcare solutions
3.5 Government Regulation
3.5.1 Implementation of stricter quality control measures
3.5.2 Introduction of new guidelines for surgical mesh usage
3.5.3 Enhanced monitoring of post-surgical outcomes
3.5.4 Support for research and development in medical devices
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Ventral Hernia Mesh Devices Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Ventral Hernia Mesh Devices Market Segmentation
8.1 By Mesh Type
8.1.1 Permanent Synthetic Mesh (e.g., Polypropylene, ePTFE)
8.1.2 Partially Absorbable Composite Mesh
8.1.3 Fully Resorbable / Bioabsorbable Mesh
8.1.4 Biological Mesh (Allograft and Xenograft)
8.1.5 3D / Pre-shaped and Anatomical Mesh
8.1.6 Others
8.2 By End-User
8.2.1 Government Hospitals (MOH and Other Public Hospitals)
8.2.2 Private Hospitals
8.2.3 Specialized Surgical & Day-Care Centers
8.2.4 University & Teaching Hospitals
8.2.5 Others
8.3 By Material
8.3.1 Polypropylene Mesh
8.3.2 Expanded Polytetrafluoroethylene (ePTFE) Mesh
8.3.3 Polyester Mesh
8.3.4 Biological (Human and Porcine / Bovine Derived) Mesh
8.3.5 Hybrid / Composite Mesh
8.3.6 Others
8.4 By Surgical Approach
8.4.1 Open Ventral Hernia Repair
8.4.2 Laparoscopic Ventral Hernia Repair
8.4.3 Robotic Ventral Hernia Repair
8.4.4 Others
8.5 By Region
8.5.1 Central Region (Including Riyadh)
8.5.2 Western Region (Including Makkah, Madinah, Jeddah)
8.5.3 Eastern Region (Including Dammam, Al Khobar)
8.5.4 Southern Region
8.5.5 Northern Region
8.6 By Distribution Channel
8.6.1 Direct Sales to Hospitals
8.6.2 Local Distributors & Importers
8.6.3 Group Purchasing Organizations (GPOs) & Tender-Based Procurement
8.6.4 Online & E-Procurement Platforms
8.6.5 Others
8.7 By Clinical Indication
8.7.1 Primary Ventral Hernia
8.7.2 Incisional Hernia
8.7.3 Umbilical & Epigastric Hernia
8.7.4 Complex Abdominal Wall Reconstruction
8.7.5 Recurrent Ventral Hernia
8.7.6 Others
9. Saudi Arabia Ventral Hernia Mesh Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Global Hernia Mesh Revenue (Latest FY)
9.2.3 Estimated Saudi Arabia Ventral Hernia Mesh Revenue / Share
9.2.4 5-Year Revenue CAGR in Hernia / Soft Tissue Repair
9.2.5 Product Portfolio Breadth (Mesh Types & Indications Covered)
9.2.6 Regulatory Approvals & SFDA Registrations in Saudi Arabia
9.2.7 Number of SKUs / Brands Available in Saudi Arabia
9.2.8 Coverage of Key Accounts (Top MOH & Major Private Hospital Groups)
9.2.9 Share of Laparoscopic & Robotic Procedure-Focused Products
9.2.10 Average Tender Win Rate in Public-Sector Procurements
9.2.11 Average Realized Price vs Global Benchmark (Saudi Price Index)
9.2.12 Gross Margin in Hernia Mesh Portfolio
9.2.13 Local Partnership Depth (Distributors, Agents, In-country Stocking)
9.2.14 Surgeon Adoption & Preference Rating (Key Opinion Leader Feedback)
9.2.15 Post-Market Surveillance / Complication & Recall History
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Ethicon Inc. (Johnson & Johnson MedTech)
9.5.3 Becton, Dickinson and Company (BD) – C.R. Bard
9.5.4 W. L. Gore & Associates, Inc.
9.5.5 B. Braun Melsungen AG
9.5.6 Cook Medical LLC
9.5.7 Integra LifeSciences Holdings Corporation
9.5.8 Medline Industries, LP
9.5.9 ConvaTec Group plc
9.5.10 LifeCell / Allergan Aesthetics (AbbVie Inc.)
9.5.11 Herniamesh S.r.l.
9.5.12 C.R. Bard (Legacy Brand Portfolio under BD)
9.5.13 Surgical Specialties Corporation
9.5.14 Atrium Medical (Getinge AB)
9.5.15 Local & Regional Distributors (e.g., Abu Dawood Medical, Tamer Group, Cigalah Group)
10. Saudi Arabia Ventral Hernia Mesh Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Defense
10.1.3 Ministry of Education
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Hospital Infrastructure Development
10.2.2 Surgical Equipment Procurement
10.2.3 Training and Development
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Surgical Centers
10.3.3 Patients
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Surgeons
10.4.2 Hospital Administrators
10.4.3 Patients
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings
10.5.2 Improved Patient Outcomes
10.5.3 Scalability of Solutions
10.5.4 Others
11. Saudi Arabia Ventral Hernia Mesh Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare authorities in Saudi Arabia
- Review of published studies on ventral hernia repair techniques and mesh device efficacy
- Examination of regulatory frameworks and guidelines from the Saudi Food and Drug Authority (SFDA)
Primary Research
- Interviews with surgeons specializing in hernia repair across major hospitals
- Surveys with medical device distributors and suppliers in the region
- Focus groups with patients who have undergone ventral hernia repair
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews
- Triangulation of data from clinical studies and market trends
- Sanity checks through feedback from a panel of healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Saudi Arabia related to surgical procedures
- Analysis of the prevalence of ventral hernias and associated surgical interventions
- Incorporation of demographic data to assess potential market growth
Bottom-up Modeling
- Collection of sales data from leading mesh device manufacturers operating in Saudi Arabia
- Estimation of average selling prices for various types of ventral hernia mesh devices
- Volume estimates based on surgical procedure rates and device usage per procedure
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth and aging demographics
- Scenario modeling based on potential changes in healthcare policies and reimbursement rates
- Development of baseline, optimistic, and pessimistic market forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Surgeons Performing Hernia Repairs | 90 | General Surgeons, Surgical Specialists |
| Medical Device Distributors | 70 | Sales Managers, Product Specialists |
| Healthcare Administrators | 55 | Hospital Administrators, Procurement Officers |
| Patients with Ventral Hernia | 85 | Post-operative Patients, Patient Advocates |
| Regulatory Experts | 45 | Compliance Officers, Regulatory Affairs Managers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Ventral Hernia Mesh Devices Market?
The Saudi Arabia Ventral Hernia Mesh Devices Market is valued at approximately USD 4 million, driven by factors such as the increasing prevalence of hernias, advancements in mesh materials, and the growing adoption of minimally invasive surgical procedures.