Region:Global
Author(s):Dev
Product Code:KRAA2971
Pages:88
Published On:January 2026
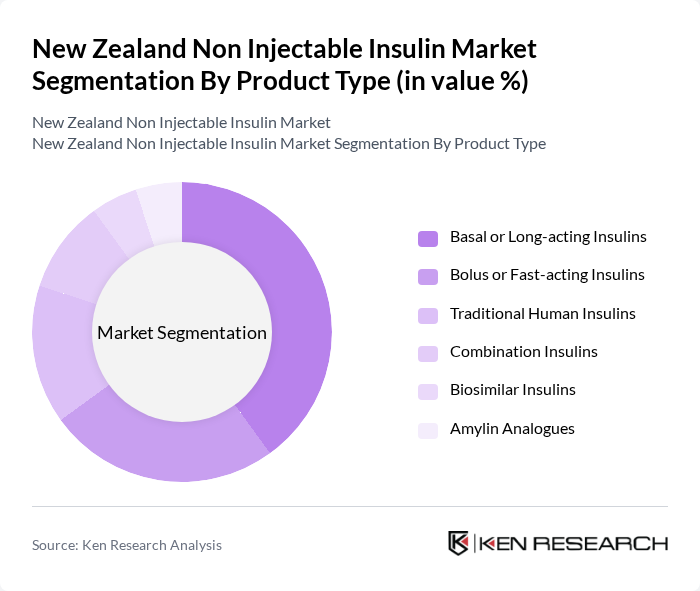
By Product Type:The product type segmentation includes various forms of non-injectable insulin, each catering to different patient needs and preferences. The subsegments are Basal or Long-acting Insulins, Bolus or Fast-acting Insulins, Traditional Human Insulins, Combination Insulins, Biosimilar Insulins, and Amylin Analogues. Among these, Basal or Long-acting Insulins dominate the market due to their convenience and effectiveness in maintaining stable blood glucose levels over extended periods. Patients prefer these formulations for their ease of use and reduced frequency of administration.
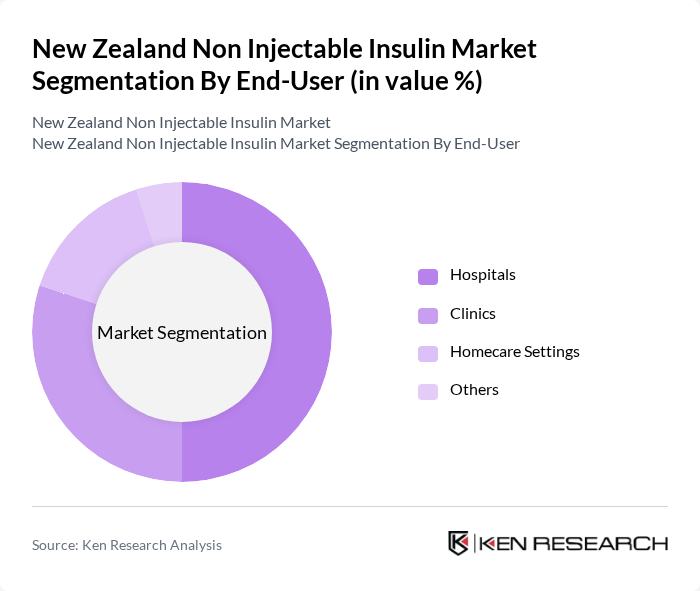
By End-User:The end-user segmentation encompasses various settings where non-injectable insulin is utilized, including Hospitals, Clinics, Homecare Settings, and Others. Hospitals are the leading end-user segment, as they provide comprehensive diabetes management services and have the necessary infrastructure to support the administration of non-injectable insulin. The increasing number of diabetes patients seeking treatment in hospitals contributes significantly to this segment's growth.
The New Zealand Non Injectable Insulin Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novo Nordisk, Sanofi, Eli Lilly and Company, Boehringer Ingelheim, AstraZeneca, Roche, Medtronic, Insulet Corporation, Tandem Diabetes Care, Zealand Pharma, Valeritas Holdings, Abbott Laboratories, Johnson & Johnson, Bayer AG, Merck & Co. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand non-injectable insulin market appears promising, driven by increasing technological advancements and a shift towards patient-centric healthcare solutions. As telehealth services expand, more patients will have access to innovative diabetes management tools. Additionally, the integration of artificial intelligence in treatment plans is expected to enhance personalized care, improving patient outcomes. These trends indicate a robust growth trajectory for non-injectable insulin products in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Basal or Long-acting Insulins Bolus or Fast-acting Insulins Traditional Human Insulins Combination Insulins Biosimilar Insulins Amylin Analogues |
| By End-User | Hospitals Clinics Homecare Settings Others |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospitals and Clinics Others |
| By Demographics | Age Group (Children, Adults, Seniors) Gender (Male, Female) Others |
| By Geography | North Island South Island Others |
| By Product Formulation | Liquid Formulations Powder Formulations Others |
| By Pricing Strategy | Premium Pricing Competitive Pricing Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Endocrinologists and Diabetes Specialists | 45 | Healthcare Providers, Clinical Researchers |
| Pharmacists in Community Settings | 80 | Pharmacy Managers, Dispensing Pharmacists |
| Patients Using Non-Injectable Insulin | 120 | Diabetes Patients, Caregivers |
| Healthcare Policy Makers | 60 | Health Economists, Policy Analysts |
| Diabetes Support Groups | 70 | Patient Advocates, Group Leaders |
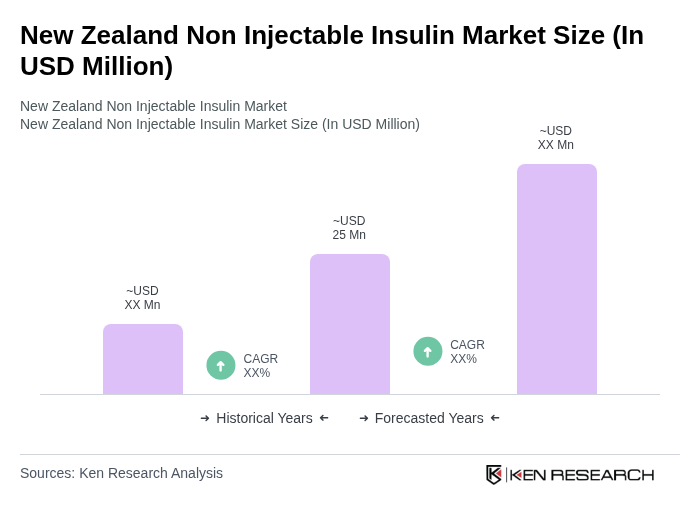
The New Zealand Non Injectable Insulin Market is valued at approximately USD 25 million, reflecting a growing demand driven by the increasing prevalence of diabetes and advancements in non-injectable insulin formulations.