About the Report
Base Year 2024Philippines Pharmacovigilance Industry Market Overview
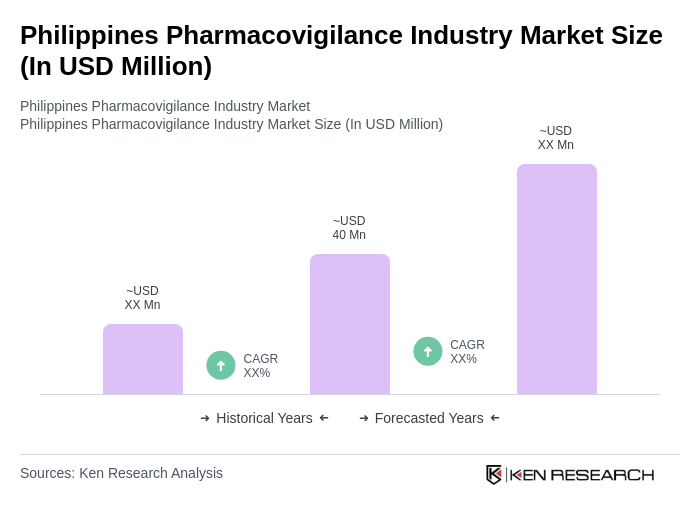
- The Philippines Pharmacovigilance Industry Market is valued at USD 40 million, based on a five-year historical analysis. This growth is primarily driven by the increasing focus on drug safety, regulatory compliance, and the rising number of adverse drug reaction reports. The market is also supported by advancements in technology and data analytics, such as the adoption of digital monitoring tools and automation platforms, which enhance the efficiency of pharmacovigilance processes. Outsourcing of safety services and the expansion of clinical trials further contribute to market growth.
- Metro Manila, Cebu, and Davao are the dominant regions in the Philippines Pharmacovigilance Industry Market. Metro Manila, being the capital, hosts numerous pharmaceutical companies and regulatory bodies, while Cebu and Davao are emerging hubs for healthcare services and research. The concentration of healthcare facilities and educational institutions in these areas fosters a conducive environment for pharmacovigilance activities.
- In 2023, the Philippines' Food and Drug Administration (FDA) implemented the "Guidelines on the Establishment of Pharmacovigilance Systems for Marketing Authorization Holders," issued by the Food and Drug Administration of the Philippines. This regulation mandates all pharmaceutical companies to establish robust pharmacovigilance systems, ensuring continuous monitoring of drug safety and efficacy. Companies are required to report adverse events within 15 days of occurrence, and compliance is essential for maintaining market authorization and public trust. The regulation covers operational requirements for reporting, documentation, and the appointment of qualified pharmacovigilance personnel.
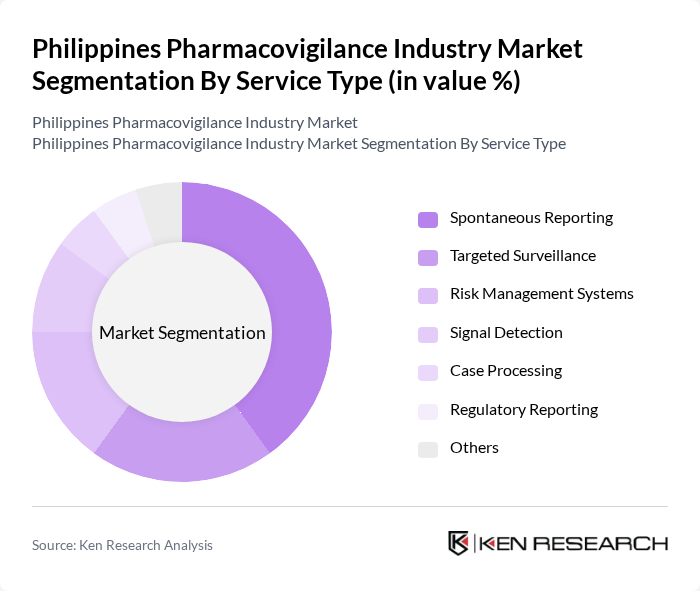
Philippines Pharmacovigilance Industry Market Segmentation
By Service Type:The service type segmentation includes various subsegments that cater to different aspects of pharmacovigilance. The key subsegments are Spontaneous Reporting, Targeted Surveillance, Risk Management Systems, Signal Detection, Case Processing, Regulatory Reporting, and Others. Among these, Spontaneous Reporting is the most dominant due to its essential role in capturing real-time data on adverse drug reactions from healthcare professionals and patients. This method is widely adopted as it allows for immediate action and risk assessment, making it a critical component of pharmacovigilance. (source)
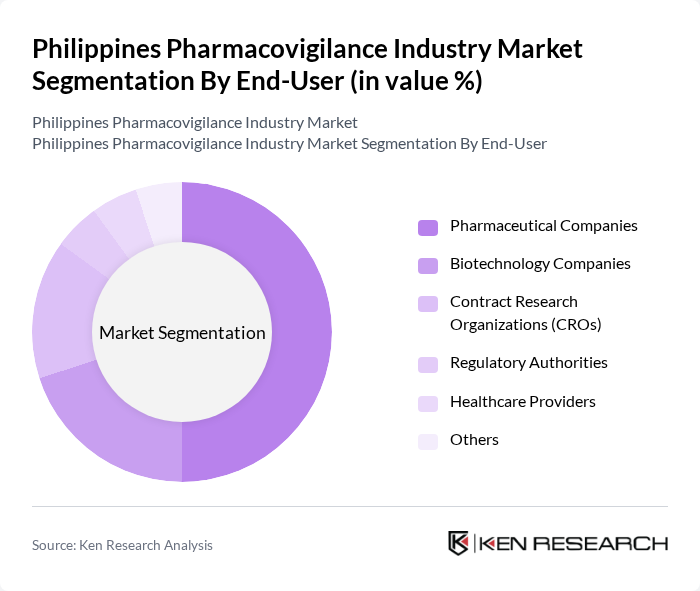
By End-User:The end-user segmentation encompasses various stakeholders involved in pharmacovigilance, including Pharmaceutical Companies, Biotechnology Companies, Contract Research Organizations (CROs), Regulatory Authorities, Healthcare Providers, and Others. Pharmaceutical Companies dominate this segment as they are primarily responsible for drug development and safety monitoring. Their extensive resources and established processes for adverse event reporting make them the leading end-users in the pharmacovigilance landscape. (source)
Philippines Pharmacovigilance Industry Market Competitive Landscape
The Philippines Pharmacovigilance Industry Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sanofi Philippines, Inc., GlaxoSmithKline Philippines, Inc., Pfizer Philippines, Inc., Merck Sharp & Dohme (MSD) Philippines, Novartis Healthcare Philippines, Inc., Roche Philippines, Inc., AstraZeneca Pharmaceuticals (Philippines), Inc., Johnson & Johnson (Philippines), Inc., Eli Lilly and Company (Philippines), Inc., Amgen Philippines, Inc., Bayer Philippines, Inc., Boehringer Ingelheim (Philippines), Inc., Takeda Healthcare Philippines, Inc., UCB Pharma (Philippines), Inc., Servier Laboratories (Philippines), Inc., IQVIA Solutions Philippines, Inc., Syneos Health Philippines, Inc., Parexel International (Philippines), Inc., Tech Mahindra Limited (Philippines), Medidata Solutions (Philippines), Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Philippines Pharmacovigilance Industry Market Industry Analysis
Growth Drivers
- Increasing Regulatory Compliance Requirements:The Philippines has seen a significant rise in regulatory compliance requirements, with the FDA implementing over 30 new guidelines in the recent past. This has led to a 15% increase in the number of pharmacovigilance reports filed, reflecting a growing commitment to drug safety. The government’s focus on aligning with international standards, such as those set by the WHO, has further propelled the need for robust pharmacovigilance systems, ensuring that patient safety remains a top priority.
- Rising Awareness of Drug Safety:Public awareness regarding drug safety has surged, with a reported 40% increase in inquiries about adverse drug reactions in the recent past. This heightened awareness is driven by media coverage and educational campaigns, leading to a greater demand for effective pharmacovigilance practices. The Philippine government has allocated PHP 500 million for public health initiatives aimed at educating citizens about drug safety, which is expected to enhance reporting and monitoring of drug-related issues significantly.
- Expansion of Pharmaceutical Market:The Philippine pharmaceutical market is projected to reach PHP 300 billion by the end of the near future, driven by a growing population and increased healthcare spending. This expansion necessitates enhanced pharmacovigilance efforts to monitor the safety of a wider array of drugs. With over 1,000 new drug registrations in the recent past, the demand for effective monitoring systems has never been higher, prompting pharmaceutical companies to invest in comprehensive pharmacovigilance frameworks to ensure compliance and safety.
Market Challenges
- Limited Resources for Pharmacovigilance:The Philippines faces significant challenges due to limited resources allocated for pharmacovigilance activities. In the recent past, only PHP 200 million was dedicated to pharmacovigilance initiatives, which is insufficient given the growing number of drugs on the market. This resource constraint hampers the ability to conduct thorough monitoring and reporting, leading to potential gaps in drug safety oversight and increased risks for patients.
- Lack of Trained Professionals:The shortage of trained pharmacovigilance professionals is a critical challenge in the Philippines. Currently, there are only about 500 certified pharmacovigilance experts in the country, which is inadequate for a market that requires extensive monitoring. This lack of expertise results in delays in reporting adverse drug reactions and compromises the overall effectiveness of pharmacovigilance systems, ultimately affecting patient safety and regulatory compliance.
Philippines Pharmacovigilance Industry Market Future Outlook
The future of the Philippines pharmacovigilance industry appears promising, driven by increasing regulatory demands and technological advancements. As the government continues to enhance its regulatory framework, the integration of digital health solutions and real-world evidence will play a crucial role in improving drug safety monitoring. Additionally, the rise of biopharmaceuticals will necessitate more robust pharmacovigilance practices, ensuring that patient safety remains at the forefront of healthcare initiatives in the country.
Market Opportunities
- Growth of Biopharmaceuticals:The biopharmaceutical sector in the Philippines is expected to grow significantly, with investments projected to reach PHP 50 billion in the near future. This growth presents an opportunity for enhanced pharmacovigilance systems to monitor the safety of innovative therapies, ensuring that emerging treatments are effectively managed and patient safety is prioritized.
- Increased Investment in Healthcare Infrastructure:The Philippine government plans to invest PHP 100 billion in healthcare infrastructure in the near future. This investment will facilitate the establishment of advanced pharmacovigilance centers, improving data collection and analysis capabilities. Enhanced infrastructure will support better monitoring of drug safety, ultimately leading to improved patient outcomes and compliance with international standards.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Service Type | Spontaneous Reporting Targeted Surveillance Risk Management Systems Signal Detection Case Processing Regulatory Reporting Others |
| By End-User | Pharmaceutical Companies Biotechnology Companies Contract Research Organizations (CROs) Regulatory Authorities Healthcare Providers Others |
| By Application | Clinical Trials (Pre-marketing) Post-Marketing Surveillance (Phase IV) Risk Assessment Signal Management Others |
| By Region | Luzon Visayas Mindanao Others |
| By Technology | Data Analytics Tools Reporting Software Database Management Systems Artificial Intelligence & Automation Cloud-Based Solutions Others |
| By Investment Source | Private Investments Government Funding International Grants Others |
| By Policy Support | Regulatory Incentives Funding for Research Training Programs Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Department of Health)
Pharmaceutical Manufacturers
Biotechnology Companies
Clinical Research Organizations
Healthcare Providers and Hospitals
Pharmacovigilance Service Providers
Insurance Companies
Players Mentioned in the Report:
Sanofi Philippines, Inc.
GlaxoSmithKline Philippines, Inc.
Pfizer Philippines, Inc.
Merck Sharp & Dohme (MSD) Philippines
Novartis Healthcare Philippines, Inc.
Roche Philippines, Inc.
AstraZeneca Pharmaceuticals (Philippines), Inc.
Johnson & Johnson (Philippines), Inc.
Eli Lilly and Company (Philippines), Inc.
Amgen Philippines, Inc.
Bayer Philippines, Inc.
Boehringer Ingelheim (Philippines), Inc.
Takeda Healthcare Philippines, Inc.
UCB Pharma (Philippines), Inc.
Servier Laboratories (Philippines), Inc.
IQVIA Solutions Philippines, Inc.
Syneos Health Philippines, Inc.
Parexel International (Philippines), Inc.
Tech Mahindra Limited (Philippines)
Medidata Solutions (Philippines), Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Philippines Pharmacovigilance Industry Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Philippines Pharmacovigilance Industry Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Philippines Pharmacovigilance Industry Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Regulatory Compliance Requirements
3.1.2 Rising Awareness of Drug Safety
3.1.3 Expansion of Pharmaceutical Market
3.1.4 Technological Advancements in Data Management
3.2 Market Challenges
3.2.1 Limited Resources for Pharmacovigilance
3.2.2 Data Privacy Concerns
3.2.3 Lack of Trained Professionals
3.2.4 Fragmented Market Structure
3.3 Market Opportunities
3.3.1 Growth of Biopharmaceuticals
3.3.2 Increased Investment in Healthcare Infrastructure
3.3.3 Collaborations with Global Pharmacovigilance Networks
3.3.4 Adoption of Artificial Intelligence in Drug Safety
3.4 Market Trends
3.4.1 Shift Towards Real-World Evidence
3.4.2 Integration of Digital Health Solutions
3.4.3 Emphasis on Patient-Centric Approaches
3.4.4 Growing Importance of Risk Management Plans
3.5 Government Regulation
3.5.1 Implementation of the FDA Act
3.5.2 Compliance with International Standards
3.5.3 Mandatory Reporting of Adverse Drug Reactions
3.5.4 Establishment of Pharmacovigilance Centers
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Philippines Pharmacovigilance Industry Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Philippines Pharmacovigilance Industry Market Segmentation
8.1 By Service Type
8.1.1 Spontaneous Reporting
8.1.2 Targeted Surveillance
8.1.3 Risk Management Systems
8.1.4 Signal Detection
8.1.5 Case Processing
8.1.6 Regulatory Reporting
8.1.7 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Companies
8.2.3 Contract Research Organizations (CROs)
8.2.4 Regulatory Authorities
8.2.5 Healthcare Providers
8.2.6 Others
8.3 By Application
8.3.1 Clinical Trials (Pre-marketing)
8.3.2 Post-Marketing Surveillance (Phase IV)
8.3.3 Risk Assessment
8.3.4 Signal Management
8.3.5 Others
8.4 By Region
8.4.1 Luzon
8.4.2 Visayas
8.4.3 Mindanao
8.4.4 Others
8.5 By Technology
8.5.1 Data Analytics Tools
8.5.2 Reporting Software
8.5.3 Database Management Systems
8.5.4 Artificial Intelligence & Automation
8.5.5 Cloud-Based Solutions
8.5.6 Others
8.6 By Investment Source
8.6.1 Private Investments
8.6.2 Government Funding
8.6.3 International Grants
8.6.4 Others
8.7 By Policy Support
8.7.1 Regulatory Incentives
8.7.2 Funding for Research
8.7.3 Training Programs
8.7.4 Others
9. Philippines Pharmacovigilance Industry Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Local Revenue (USD, latest year)
9.2.4 Revenue Growth Rate (CAGR %)
9.2.5 Market Penetration Rate (%)
9.2.6 Number of Pharmacovigilance Cases Processed Annually
9.2.7 Compliance Rate with FDA/ICH Guidelines (%)
9.2.8 Turnaround Time for Case Reporting (days)
9.2.9 Customer Retention Rate (%)
9.2.10 Adoption of Digital/AI Tools (Y/N or % of operations)
9.2.11 Operational Efficiency (Cases per FTE)
9.2.12 Innovation Index (e.g., number of new solutions launched)
9.2.13 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sanofi Philippines, Inc.
9.5.2 GlaxoSmithKline Philippines, Inc.
9.5.3 Pfizer Philippines, Inc.
9.5.4 Merck Sharp & Dohme (MSD) Philippines
9.5.5 Novartis Healthcare Philippines, Inc.
9.5.6 Roche Philippines, Inc.
9.5.7 AstraZeneca Pharmaceuticals (Philippines), Inc.
9.5.8 Johnson & Johnson (Philippines), Inc.
9.5.9 Eli Lilly and Company (Philippines), Inc.
9.5.10 Amgen Philippines, Inc.
9.5.11 Bayer Philippines, Inc.
9.5.12 Boehringer Ingelheim (Philippines), Inc.
9.5.13 Takeda Healthcare Philippines, Inc.
9.5.14 UCB Pharma (Philippines), Inc.
9.5.15 Servier Laboratories (Philippines), Inc.
9.5.16 IQVIA Solutions Philippines, Inc.
9.5.17 Syneos Health Philippines, Inc.
9.5.18 Parexel International (Philippines), Inc.
9.5.19 Tech Mahindra Limited (Philippines)
9.5.20 Medidata Solutions (Philippines), Inc.
10. Philippines Pharmacovigilance Industry Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Department of Health
10.1.2 Food and Drug Administration
10.1.3 Department of Science and Technology
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Pharmacovigilance Systems
10.2.2 Budget Allocation for Drug Safety
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Healthcare Providers
10.4 User Readiness for Adoption
10.4.1 Training and Development Needs
10.4.2 Technology Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Pharmacovigilance Outcomes
10.5.2 Expansion into New Therapeutic Areas
11. Philippines Pharmacovigilance Industry Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Partnerships with Healthcare Institutions
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
6. Customer Relationship
6.1 Loyalty Programs Development
6.2 After-sales Service Strategies
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Approaches
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Management Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors Identification
14.2 Joint Ventures Exploration
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of regulatory frameworks from the Food and Drug Administration (FDA) of the Philippines
- Review of published reports from the Department of Health (DOH) on drug safety and adverse event reporting
- Examination of industry publications and white papers on pharmacovigilance trends in Southeast Asia
Primary Research
- Interviews with pharmacovigilance officers at major pharmaceutical companies operating in the Philippines
- Surveys conducted with healthcare professionals involved in drug safety monitoring
- Focus group discussions with regulatory experts and industry stakeholders
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government reports and industry surveys
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks performed by an expert panel comprising pharmacovigilance specialists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total market size based on national healthcare expenditure and pharmaceutical sales data
- Segmentation of the market by therapeutic areas and types of adverse event reporting
- Incorporation of government initiatives aimed at enhancing drug safety and monitoring
Bottom-up Modeling
- Collection of data on the number of reported adverse drug reactions from hospitals and clinics
- Estimation of operational costs associated with pharmacovigilance activities in pharmaceutical firms
- Volume x cost analysis based on the frequency of adverse event reporting and follow-up actions
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as population growth, healthcare access, and drug consumption trends
- Scenario modeling based on potential changes in regulatory policies and public health initiatives
- Development of baseline, optimistic, and pessimistic forecasts for the market through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 100 | Pharmacovigilance Managers, Regulatory Affairs Specialists |
| Healthcare Providers | 80 | Doctors, Pharmacists, Nurses |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
| Patient Advocacy Groups | 40 | Patient Representatives, Health Advocates |
| Academic Institutions | 40 | Researchers, Professors in Pharmacology |
Frequently Asked Questions
What is the current value of the Philippines Pharmacovigilance Industry Market?
The Philippines Pharmacovigilance Industry Market is valued at approximately USD 40 million, reflecting a significant focus on drug safety, regulatory compliance, and the increasing number of adverse drug reaction reports over the past five years.