About the Report
Base Year 2024Saudi Arabia Aseptic Connectors Welders Market Overview
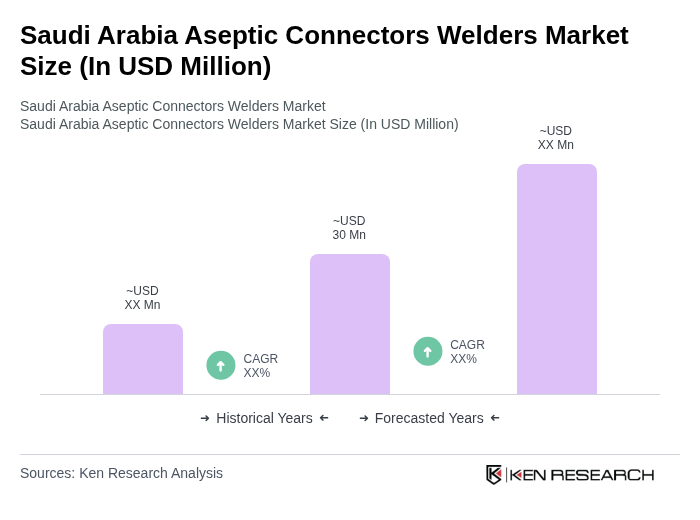
- The Saudi Arabia Aseptic Connectors Welders Market is valued at USD 30 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for sterile manufacturing processes in the biopharmaceutical and pharmaceutical industries, alongside a rising focus on safety, contamination control, and quality in drug production. The market is further supported by the expansion of single-use technologies and the adoption of advanced automation solutions in bioprocessing facilities .
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure and the presence of major pharmaceutical and biopharmaceutical companies. These cities are also focal points for government investments in healthcare and technology, further enhancing their market position. The Saudi government’s Vision 2030 initiative has accelerated investments in biomanufacturing and localizing pharmaceutical production, driving demand for aseptic processing equipment .
- The “Guidelines for Good Manufacturing Practice (GMP) for Pharmaceutical Products,” issued by the Saudi Food and Drug Authority (SFDA) in 2023, mandate the use of validated aseptic connectors and welders in biopharmaceutical manufacturing. These guidelines require manufacturers to implement internationally recognized sterile processing standards, including the use of closed-system aseptic transfer devices, to ensure product quality and patient safety .
Saudi Arabia Aseptic Connectors Welders Market Segmentation
By Product Type:
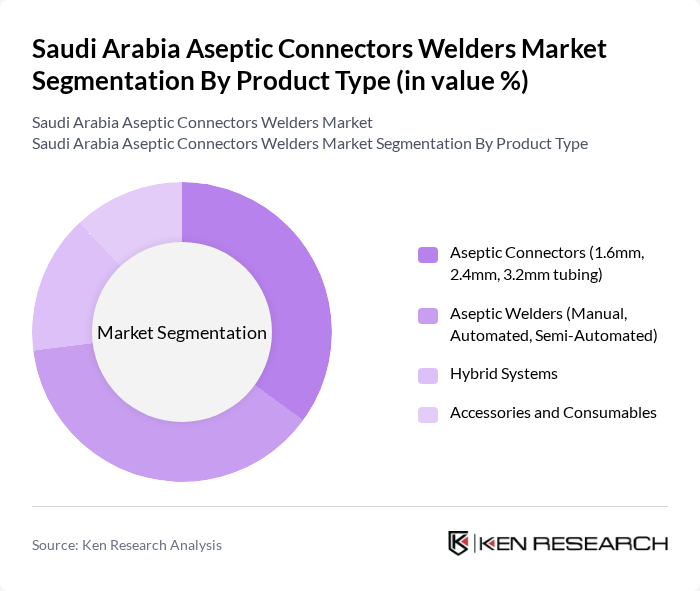
The product type segment is led by Aseptic Welders, reflecting the market’s shift toward automation and closed-system processing to minimize contamination risks and improve operational efficiency. Automated and semi-automated welders are increasingly adopted in large-scale bioprocessing, while aseptic connectors remain essential for flexible and single-use applications, especially in sterile fluid transfer. Hybrid systems and consumables support the integration of both technologies, catering to the evolving needs of biopharmaceutical manufacturers .
By End-User Industry:
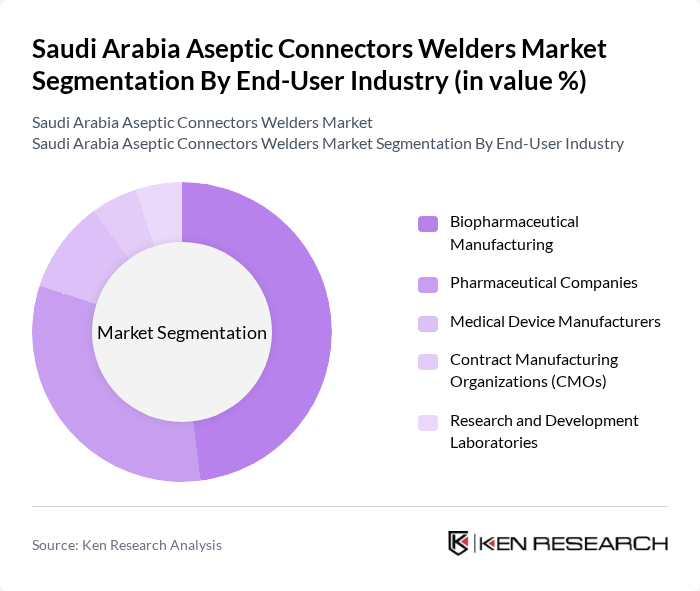
The biopharmaceutical manufacturing sector is the leading end-user, driven by the surge in biologics, biosimilars, and vaccine production. This sector’s growth is propelled by increased investments in research and development, the expansion of local manufacturing capacity, and the need for stringent aseptic processing to meet global regulatory standards. Pharmaceutical companies and medical device manufacturers are also significant contributors, with CMOs and R&D labs adopting aseptic technologies to support flexible and small-batch production .
Saudi Arabia Aseptic Connectors Welders Market Competitive Landscape
The Saudi Arabia Aseptic Connectors Welders Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sartorius AG, Merck KGaA, Danaher Corporation (Cytiva), Saint-Gobain, Dover Corporation (Colder Products Company), Watson-Marlow Fluid Technology Solutions, Terumo BCT, Inc., Liquidyne Process Technologies, Inc., MGA Technologies, LePure Biotech LLC contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Aseptic Connectors Welders Market Industry Analysis
Growth Drivers
- Increasing Demand for Sterile Environments:The biopharmaceutical manufacturing sector in Saudi Arabia is projected to grow significantly, with an estimated value of SAR 25 billion in future. This surge is driven by the rising need for sterile environments to ensure product safety and efficacy. The World Health Organization emphasizes that maintaining sterility is crucial, leading to increased investments in aseptic technologies, including connectors and welders, to meet stringent production standards.
- Technological Advancements:The Saudi aseptic connectors welders market is witnessing rapid technological advancements, with investments exceeding SAR 2 billion in R&D for innovative welding solutions. These advancements enhance the efficiency and reliability of aseptic processes, reducing contamination risks. The integration of smart technologies, such as IoT and AI, is expected to improve operational efficiency, making aseptic welding systems more attractive to manufacturers aiming for high-quality production.
- Government Initiatives Under Vision 2030:Saudi Arabia's Vision 2030 aims to diversify the economy, with healthcare and pharmaceuticals receiving significant attention. The government allocated SAR 60 billion for healthcare infrastructure improvements, fostering local manufacturing capabilities. This initiative is expected to boost the demand for aseptic connectors and welders, as local manufacturers seek to comply with international standards and enhance production capabilities in the biopharmaceutical sector.
Market Challenges
- High Initial Investment Costs:The adoption of aseptic systems in Saudi Arabia faces challenges due to high initial investment costs, which can exceed SAR 3 million for advanced systems. Many local manufacturers are hesitant to invest in these technologies, given the financial burden. This reluctance can hinder the overall growth of the aseptic connectors welders market, as companies may opt for less expensive, traditional methods that do not meet modern standards.
- Limited Local Awareness:There is a significant gap in local awareness regarding advanced aseptic technologies, with only 40% of manufacturers familiar with the latest innovations. This lack of knowledge can impede the adoption of aseptic connectors and welders, as companies may not recognize the benefits of these systems. Educational initiatives and training programs are essential to bridge this gap and promote the advantages of modern aseptic solutions in the industry.
Saudi Arabia Aseptic Connectors Welders Market Future Outlook
The future of the Saudi Arabia aseptic connectors welders market appears promising, driven by ongoing investments in healthcare and biopharmaceutical sectors. As local manufacturers increasingly adopt advanced technologies, the market is expected to witness a shift towards automation and smart solutions. Additionally, the government's commitment to enhancing healthcare infrastructure under Vision 2030 will likely create a conducive environment for growth, fostering innovation and collaboration among industry stakeholders to meet evolving market demands.
Market Opportunities
- Expansion of Biotechnology Sector:The biotechnology sector in Saudi Arabia is projected to grow to SAR 20 billion in future, presenting significant opportunities for aseptic connectors and welders. This growth is driven by increased research and development activities, necessitating advanced aseptic solutions to ensure product integrity and safety in biopharmaceutical manufacturing.
- Growing Demand for Single-Use Systems:The demand for single-use systems in pharmaceutical manufacturing is on the rise, with an expected market value of SAR 7 billion in future. This trend is driven by the need for flexibility and reduced contamination risks, creating opportunities for aseptic connectors and welders designed for single-use applications, thereby enhancing operational efficiency in production processes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Aseptic Connectors (1.6mm, 2.4mm, 3.2mm tubing) Aseptic Welders (Manual, Automated, Semi-Automated) Hybrid Systems Accessories and Consumables |
| By End-User Industry | Biopharmaceutical Manufacturing Pharmaceutical Companies Medical Device Manufacturers Contract Manufacturing Organizations (CMOs) Research and Development Laboratories |
| By Application | Sterile Fluid Transfer Systems Single-Use Bioprocess Systems Drug Delivery and Vaccine Manufacturing Biologics Production |
| By Material Composition | Plastic Aseptic Connectors (Polycarbonate, PTFE) Metal Aseptic Connectors (Stainless Steel) Composite Materials Pre-Sterilized Single-Use Components |
| By Geographic Region within Saudi Arabia | Central Region (Riyadh) Eastern Region (Dammam, Khobar) Western Region (Jeddah, Mecca) Southern Region (Abha, Jizan) |
| By Distribution Channel | Direct Sales to End-Users Authorized Distributors and Resellers Online and E-Commerce Platforms System Integrators and Solution Providers |
| By Technology Platform | Traditional Thermal Welding Laser Welding Technology Ultrasonic Welding Technology Automated Robotic Welding Systems |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers of Aseptic Connectors
Distributors and Retailers of Medical Equipment
Healthcare Providers and Hospitals
Technology Providers for Medical Devices
Industry Associations (e.g., Saudi Arabian Medical Devices Association)
Financial Institutions and Banks
Players Mentioned in the Report:
Sartorius AG
Merck KGaA
Danaher Corporation (Cytiva)
Saint-Gobain
Dover Corporation (Colder Products Company)
Watson-Marlow Fluid Technology Solutions
Terumo BCT, Inc.
Liquidyne Process Technologies, Inc.
MGA Technologies
LePure Biotech LLC
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Aseptic Connectors Welders Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Aseptic Connectors Welders Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Aseptic Connectors Welders Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for sterile environments in biopharmaceutical manufacturing
3.1.2 Technological advancements in aseptic welding and connector design
3.1.3 Rising healthcare and pharmaceutical sectors under Vision 2030
3.1.4 Government initiatives for healthcare infrastructure and local manufacturing
3.2 Market Challenges
3.2.1 High initial investment costs for aseptic systems
3.2.2 Limited local awareness of advanced aseptic technologies
3.2.3 Stringent regulatory requirements for medical device manufacturing
3.2.4 Competition from traditional welding methods and imported solutions
3.3 Market Opportunities
3.3.1 Expansion of the biotechnology and biopharmaceutical sector
3.3.2 Growing demand for single-use systems in pharmaceutical manufacturing
3.3.3 Increasing focus on contamination control and infection prevention
3.3.4 Potential for export to GCC and MENA regions
3.4 Market Trends
3.4.1 Shift towards automation and robotic welding in manufacturing
3.4.2 Adoption of smart technologies and real-time monitoring in welding
3.4.3 Emphasis on sustainability and eco-friendly welding practices
3.4.4 Rising investments in R&D for innovative aseptic solutions
3.5 Government Regulation
3.5.1 Compliance with health and safety standards (SASO, SFDA)
3.5.2 Regulations on medical device manufacturing and quality assurance
3.5.3 Guidelines for aseptic processing and contamination control
3.5.4 Incentives for local manufacturing under Vision 2030
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Aseptic Connectors Welders Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Aseptic Connectors Welders Market Segmentation
8.1 By Product Type
8.1.1 Aseptic Connectors (1.6mm, 2.4mm, 3.2mm tubing)
8.1.2 Aseptic Welders (Manual, Automated, Semi-Automated)
8.1.3 Hybrid Systems
8.1.4 Accessories and Consumables
8.2 By End-User Industry
8.2.1 Biopharmaceutical Manufacturing
8.2.2 Pharmaceutical Companies
8.2.3 Medical Device Manufacturers
8.2.4 Contract Manufacturing Organizations (CMOs)
8.2.5 Research and Development Laboratories
8.3 By Application
8.3.1 Sterile Fluid Transfer Systems
8.3.2 Single-Use Bioprocess Systems
8.3.3 Drug Delivery and Vaccine Manufacturing
8.3.4 Biologics Production
8.4 By Material Composition
8.4.1 Plastic Aseptic Connectors (Polycarbonate, PTFE)
8.4.2 Metal Aseptic Connectors (Stainless Steel)
8.4.3 Composite Materials
8.4.4 Pre-Sterilized Single-Use Components
8.5 By Geographic Region within Saudi Arabia
8.5.1 Central Region (Riyadh)
8.5.2 Eastern Region (Dammam, Khobar)
8.5.3 Western Region (Jeddah, Mecca)
8.5.4 Southern Region (Abha, Jizan)
8.6 By Distribution Channel
8.6.1 Direct Sales to End-Users
8.6.2 Authorized Distributors and Resellers
8.6.3 Online and E-Commerce Platforms
8.6.4 System Integrators and Solution Providers
8.7 By Technology Platform
8.7.1 Traditional Thermal Welding
8.7.2 Laser Welding Technology
8.7.3 Ultrasonic Welding Technology
8.7.4 Automated Robotic Welding Systems
9. Saudi Arabia Aseptic Connectors Welders Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size Classification (Large, Medium, Small)
9.2.3 Year-over-Year Revenue Growth Rate (%)
9.2.4 Market Penetration Rate in Saudi Arabia (%)
9.2.5 Customer Retention Rate (%)
9.2.6 Pricing Strategy (Premium, Mid-Range, Value)
9.2.7 Product Innovation Index (New Products Launched Annually)
9.2.8 Operational Efficiency Ratio (Cost per Unit)
9.2.9 Global Market Share Percentage (%)
9.2.10 Customer Satisfaction Score (NPS or CSAT Index)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Sartorius AG
9.5.2 Merck KGaA
9.5.3 Danaher Corporation (Cytiva)
9.5.4 Saint-Gobain
9.5.5 Dover Corporation (Colder Products Company)
9.5.6 Watson-Marlow Fluid Technology Solutions
9.5.7 Terumo BCT, Inc.
9.5.8 Liquidyne Process Technologies, Inc.
9.5.9 MGA Technologies
9.5.10 LePure Biotech LLC
10. Saudi Arabia Aseptic Connectors Welders Market End-User Analysis
10.1 Procurement Behavior of Key Ministries and Institutions
10.1.1 Ministry of Health
10.1.2 Ministry of Industry and Mineral Resources
10.1.3 Saudi Food and Drug Authority (SFDA)
10.1.4 Saudi Standards, Metrology and Quality Organization (SASO)
10.2 Corporate Spend on Infrastructure & Manufacturing
10.2.1 Healthcare Infrastructure Investments
10.2.2 Pharmaceutical and Biopharmaceutical Manufacturing Investments
10.2.3 Research and Development Funding
10.2.4 Vision 2030 Industrial Diversification Projects
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotechnology and Biopharmaceutical Firms
10.3.3 Medical Device Manufacturers
10.3.4 Contract Manufacturing Organizations (CMOs)
10.4 User Readiness for Adoption
10.4.1 Awareness of Advanced Aseptic Technologies
10.4.2 Training and Skill Development Capabilities
10.4.3 Infrastructure Readiness and Cleanroom Standards
10.4.4 Budget Allocation for Technology Upgrades
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings and Operational Efficiency Gains
10.5.2 Contamination Reduction and Quality Improvements
10.5.3 Scalability of Solutions for Production Expansion
10.5.4 Time-to-Market Acceleration for New Products
11. Saudi Arabia Aseptic Connectors Welders Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Regional Distribution Networks
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 End-User Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service and Technical Support
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging and Compliance
9.2 Export Entry Strategy
9.2.1 Target Countries (GCC and MENA Region)
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from Saudi Arabian health and medical device associations
- Market analysis publications focusing on aseptic connectors and welding technologies
- Government publications and healthcare regulations impacting aseptic connector usage
Primary Research
- Interviews with biomedical engineers specializing in aseptic connector applications
- Surveys with procurement managers in hospitals and healthcare facilities
- Field interviews with manufacturers of aseptic connectors and welding equipment
Validation & Triangulation
- Cross-validation of data from multiple industry sources and expert opinions
- Triangulation of market size estimates using sales data and regulatory insights
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of healthcare expenditure trends in Saudi Arabia related to medical devices
- Segmentation of the market by end-user categories such as hospitals and clinics
- Incorporation of growth rates from related sectors like pharmaceuticals and biotechnology
Bottom-up Modeling
- Volume estimates based on production capacities of leading aseptic connector manufacturers
- Cost analysis derived from pricing models of various welding technologies
- Estimation of market share based on sales data from key players in the sector
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating healthcare trends and technological advancements
- Scenario planning based on potential regulatory changes and market disruptions
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 100 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Manufacturers | 70 | Product Managers, R&D Engineers |
| Healthcare Facility Administrators | 60 | Facility Managers, Operations Directors |
| Biomedical Engineering Experts | 50 | Biomedical Engineers, Technical Consultants |
| Regulatory Affairs Specialists | 40 | Regulatory Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Aseptic Connectors Welders Market?
The Saudi Arabia Aseptic Connectors Welders Market is valued at approximately USD 30 million, driven by the increasing demand for sterile manufacturing processes in the biopharmaceutical and pharmaceutical industries.