About the Report
Base Year 2024Saudi Arabia Neurointerventional Devices Market Overview
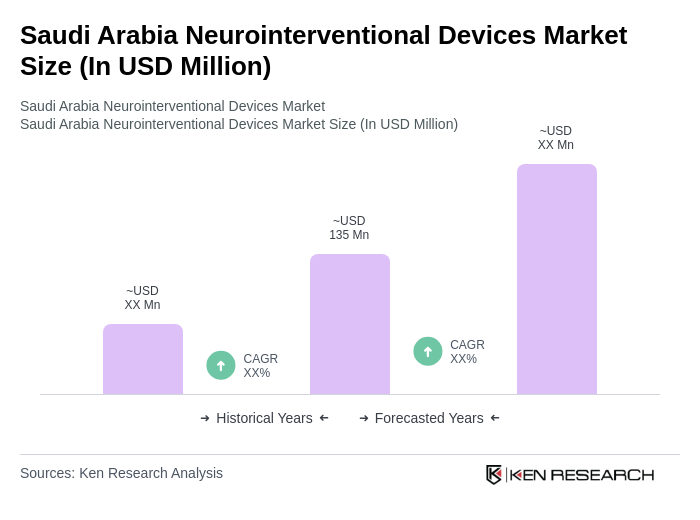
- The Saudi Arabia Neurointerventional Devices Market is valued at USD 135 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of neurological disorders, advancements in minimally invasive medical technology, and rising healthcare expenditure. The demand for innovative neurointerventional devices has surged as healthcare providers seek to improve patient outcomes and reduce recovery times. Additional growth drivers include the aging population, government investment in healthcare modernization, and the expanding private healthcare sector, which together foster increased adoption of advanced neurosurgical and interventional neurology devices .
- Key cities dominating the market include Riyadh, Jeddah, and Dammam. Riyadh serves as the capital and a major healthcare hub, hosting numerous specialized medical facilities and research institutions. Jeddah and Dammam also contribute significantly due to their advanced healthcare infrastructure and proximity to major population centers, facilitating access to neurointerventional treatments. These cities are focal points for specialist neurology centers and advanced neurosurgical procedures .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Medical Devices Interim Regulation, 2023” issued by the Saudi Food and Drug Authority. This regulation streamlines the approval process for neurointerventional devices, requiring manufacturers to obtain SFDA marketing authorization and comply with post-market surveillance and quality management standards. The initiative aims to enhance patient safety and ensure that innovative technologies are rapidly integrated into clinical practice, thereby fostering a more competitive market environment .
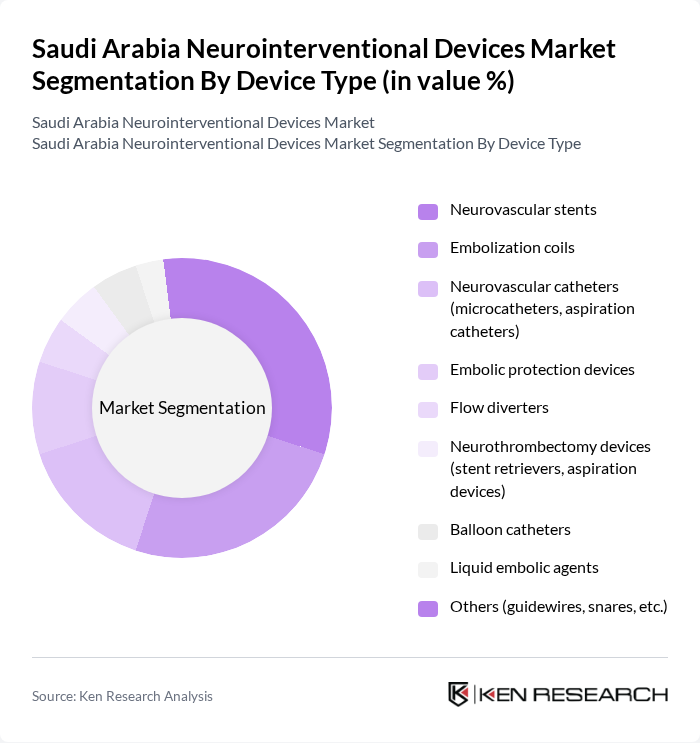
Saudi Arabia Neurointerventional Devices Market Segmentation
By Device Type:The neurointerventional devices market is segmented into various device types, including neurovascular stents, embolization coils, neurovascular catheters (microcatheters, aspiration catheters), embolic protection devices, flow diverters, neurothrombectomy devices (stent retrievers, aspiration devices), balloon catheters, liquid embolic agents, and others (guidewires, snares, etc.). Among these, neurovascular stents and embolization coils are leading the market due to their critical role in treating conditions like aneurysms and strokes. The increasing incidence of these conditions drives demand for these devices, as they are essential for effective intervention and patient recovery. The adoption of minimally invasive procedures and advanced imaging techniques further supports growth in these segments .
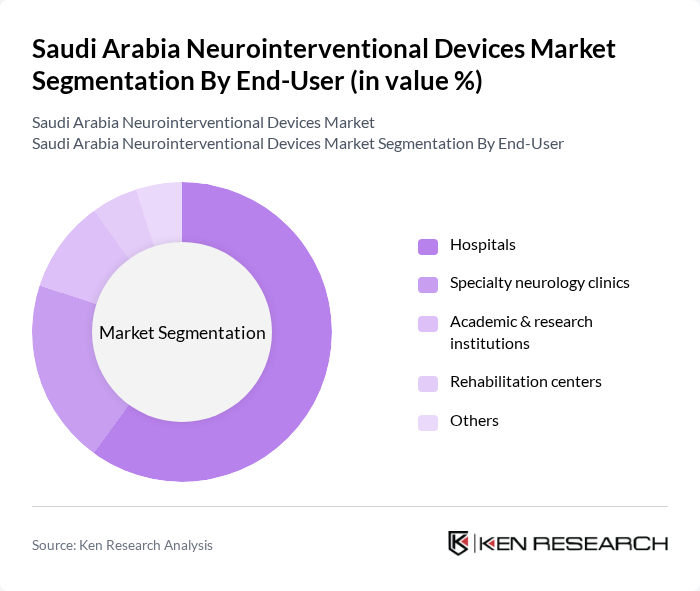
By End-User:The market is segmented by end-users, including hospitals, specialty neurology clinics, academic & research institutions, rehabilitation centers, and others. Hospitals dominate the market due to their comprehensive facilities and ability to provide a wide range of neurointerventional procedures. The increasing number of specialized neurology clinics also contributes to market growth, as they focus on advanced treatment options and patient care. The expansion of specialist neurology centers and improved reimbursement policies further drive hospital segment dominance .
Saudi Arabia Neurointerventional Devices Market Competitive Landscape
The Saudi Arabia Neurointerventional Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Stryker Corporation, Johnson & Johnson (Cerenovus), Boston Scientific Corporation, Terumo Corporation, Penumbra, Inc., MicroVention, Inc. (a Terumo Group company), Abbott Laboratories, Asahi Intecc Co., Ltd., Cook Medical, Balt Group, Phenox GmbH, Inari Medical, Inc., Acandis GmbH, AngioDynamics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Neurointerventional Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Neurological Disorders:The prevalence of neurological disorders in Saudi Arabia is rising, with approximately 1.5 million people affected by conditions such as stroke and epilepsy. According to the Saudi Ministry of Health, the incidence of stroke alone is estimated at 100 cases per 100,000 individuals annually. This growing patient population necessitates advanced neurointerventional devices, driving market demand significantly as healthcare providers seek effective treatment options to address these conditions.
- Advancements in Neurointerventional Technologies:Technological innovations in neurointerventional devices are enhancing treatment efficacy and safety. For instance, the introduction of advanced stent retrievers and embolization devices has improved outcomes in stroke management. The Saudi Food and Drug Authority (SFDA) reported a 30% increase in the approval of new neurointerventional devices from 2022 to 2023, reflecting a robust pipeline of innovative solutions that cater to the evolving needs of healthcare professionals and patients alike.
- Rising Healthcare Expenditure:Saudi Arabia's healthcare expenditure is projected to reach SAR 250 billion (approximately USD 66.67 billion) in future, driven by government initiatives to enhance healthcare services. This increase in funding supports the acquisition of advanced medical technologies, including neurointerventional devices. The Vision 2030 initiative emphasizes improving healthcare infrastructure, which further propels investments in innovative treatment modalities, thereby fostering market growth in the neurointerventional sector.
Market Challenges
- High Cost of Neurointerventional Devices:The high cost associated with neurointerventional devices poses a significant barrier to market growth. For instance, the average price of advanced neurovascular stents can exceed SAR 30,000 (approximately USD 8,000), limiting accessibility for many healthcare facilities. This financial constraint can hinder the adoption of these technologies, particularly in public hospitals where budget allocations are often restricted, impacting overall treatment availability for patients.
- Limited Access to Specialized Healthcare Facilities:Access to specialized healthcare facilities in Saudi Arabia remains a challenge, particularly in rural areas. According to the World Health Organization, only 30% of hospitals in the country are equipped with advanced neurointerventional capabilities. This disparity in healthcare access can lead to delays in treatment for patients suffering from neurological disorders, ultimately affecting patient outcomes and limiting the market's growth potential in underserved regions.
Saudi Arabia Neurointerventional Devices Market Future Outlook
The future of the neurointerventional devices market in Saudi Arabia appears promising, driven by ongoing advancements in technology and increasing healthcare investments. The integration of artificial intelligence in neurointerventional procedures is expected to enhance precision and outcomes. Additionally, the expansion of telemedicine services will facilitate remote consultations, improving access to specialized care. As the healthcare landscape evolves, a focus on patient-centric models will further shape the market, ensuring that innovative solutions meet the needs of both patients and providers effectively.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Saudi government is investing heavily in healthcare infrastructure, with plans to build over 30 new hospitals in future. This expansion will increase the availability of neurointerventional services, creating opportunities for device manufacturers to enter new markets and enhance their distribution networks, ultimately improving patient access to advanced treatments.
- Increasing Investment in Research and Development:The Saudi government allocated SAR 1.5 billion (approximately USD 400 million) for medical research in future, focusing on innovative healthcare solutions. This investment will foster collaborations between local and international companies, driving the development of cutting-edge neurointerventional devices and enhancing the overall market landscape through innovation and improved patient care.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Device Type | Neurovascular stents Embolization coils Neurovascular catheters (microcatheters, aspiration catheters) Embolic protection devices Flow diverters Neurothrombectomy devices (stent retrievers, aspiration devices) Balloon catheters Liquid embolic agents Others (guidewires, snares, etc.) |
| By End-User | Hospitals Specialty neurology clinics Academic & research institutions Rehabilitation centers Others |
| By Application | Ischemic stroke intervention Cerebral aneurysm treatment Arteriovenous malformation (AVM) & fistula treatment Carotid artery disease intervention Intracranial atherosclerotic disease Others |
| By Distribution Channel | Direct sales Medical device distributors Online sales Others |
| By Region | Central Region (Riyadh and surroundings) Eastern Region (Dammam, Al Khobar, etc.) Western Region (Jeddah, Mecca, Medina, etc.) Southern Region Northern Region Others |
| By Price Range | Low-end devices Mid-range devices Premium/high-end devices |
| By Technology | Conventional neurointerventional devices Advanced imaging-guided devices Robotic-assisted neurointerventional devices Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers
Industry Associations (e.g., Saudi Arabian Medical Devices Association)
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Stryker Corporation
Johnson & Johnson (Cerenovus)
Boston Scientific Corporation
Terumo Corporation
Penumbra, Inc.
MicroVention, Inc. (a Terumo Group company)
Abbott Laboratories
Asahi Intecc Co., Ltd.
Cook Medical
Balt Group
Phenox GmbH
Inari Medical, Inc.
Acandis GmbH
AngioDynamics, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Neurointerventional Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Neurointerventional Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Neurointerventional Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of neurological disorders
3.1.2 Advancements in neurointerventional technologies
3.1.3 Rising healthcare expenditure
3.1.4 Growing awareness and acceptance of minimally invasive procedures
3.2 Market Challenges
3.2.1 High cost of neurointerventional devices
3.2.2 Limited access to specialized healthcare facilities
3.2.3 Regulatory hurdles and compliance issues
3.2.4 Shortage of trained healthcare professionals
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in research and development
3.3.3 Collaborations with international medical device companies
3.3.4 Growing demand for advanced imaging technologies
3.4 Market Trends
3.4.1 Shift towards outpatient procedures
3.4.2 Integration of AI and machine learning in neurointerventions
3.4.3 Rise in telemedicine and remote monitoring
3.4.4 Focus on patient-centric care models
3.5 Government Regulation
3.5.1 Implementation of stricter quality control measures
3.5.2 Introduction of reimbursement policies for neurointerventional procedures
3.5.3 Establishment of guidelines for device approval
3.5.4 Promotion of public-private partnerships in healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Neurointerventional Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Neurointerventional Devices Market Segmentation
8.1 By Device Type
8.1.1 Neurovascular stents
8.1.2 Embolization coils
8.1.3 Neurovascular catheters (microcatheters, aspiration catheters)
8.1.4 Embolic protection devices
8.1.5 Flow diverters
8.1.6 Neurothrombectomy devices (stent retrievers, aspiration devices)
8.1.7 Balloon catheters
8.1.8 Liquid embolic agents
8.1.9 Others (guidewires, snares, etc.)
8.2 By End-User
8.2.1 Hospitals
8.2.2 Specialty neurology clinics
8.2.3 Academic & research institutions
8.2.4 Rehabilitation centers
8.2.5 Others
8.3 By Application
8.3.1 Ischemic stroke intervention
8.3.2 Cerebral aneurysm treatment
8.3.3 Arteriovenous malformation (AVM) & fistula treatment
8.3.4 Carotid artery disease intervention
8.3.5 Intracranial atherosclerotic disease
8.3.6 Others
8.4 By Distribution Channel
8.4.1 Direct sales
8.4.2 Medical device distributors
8.4.3 Online sales
8.4.4 Others
8.5 By Region
8.5.1 Central Region (Riyadh and surroundings)
8.5.2 Eastern Region (Dammam, Al Khobar, etc.)
8.5.3 Western Region (Jeddah, Mecca, Medina, etc.)
8.5.4 Southern Region
8.5.5 Northern Region
8.5.6 Others
8.6 By Price Range
8.6.1 Low-end devices
8.6.2 Mid-range devices
8.6.3 Premium/high-end devices
8.7 By Technology
8.7.1 Conventional neurointerventional devices
8.7.2 Advanced imaging-guided devices
8.7.3 Robotic-assisted neurointerventional devices
8.7.4 Others
9. Saudi Arabia Neurointerventional Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Saudi Arabia Neurointerventional Devices Revenue (USD, latest year)
9.2.4 Revenue Growth Rate (Saudi Arabia neurointerventional segment, %)
9.2.5 Market Share in Saudi Arabia (%)
9.2.6 Number of Neurointerventional Product Approvals (Saudi Arabia)
9.2.7 Number of Neurointerventional Procedures Supported (Saudi Arabia, latest year)
9.2.8 Distribution Network Coverage (number of cities/hospitals served)
9.2.9 R&D Investment in Neurointerventional Devices (% of revenue)
9.2.10 Product Portfolio Breadth (number of neurointerventional SKUs)
9.2.11 Local Partnerships/Distributors (number and key names)
9.2.12 Regulatory Compliance Score (SFDA approvals, certifications)
9.2.13 Customer Satisfaction Score (Saudi Arabia, latest survey)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Stryker Corporation
9.5.3 Johnson & Johnson (Cerenovus)
9.5.4 Boston Scientific Corporation
9.5.5 Terumo Corporation
9.5.6 Penumbra, Inc.
9.5.7 MicroVention, Inc. (a Terumo Group company)
9.5.8 Abbott Laboratories
9.5.9 Asahi Intecc Co., Ltd.
9.5.10 Cook Medical
9.5.11 Balt Group
9.5.12 Phenox GmbH
9.5.13 Inari Medical, Inc.
9.5.14 Acandis GmbH
9.5.15 AngioDynamics, Inc.
10. Saudi Arabia Neurointerventional Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of National Guard Health Affairs
10.1.3 Ministry of Defense
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare facilities
10.2.2 Budget allocation for medical devices
10.2.3 Funding for research and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Specialty clinics
10.3.3 Rehabilitation centers
10.4 User Readiness for Adoption
10.4.1 Training and education needs
10.4.2 Infrastructure readiness
10.4.3 Financial readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of device performance
10.5.2 Expansion into new applications
10.5.3 Long-term cost savings analysis
11. Saudi Arabia Neurointerventional Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
1.3 Value Proposition Canvas
1.4 Competitive Landscape Analysis
1.5 Customer Segmentation
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
3. Distribution Plan
3.1 Urban Retail Strategy
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitive Pricing Strategy
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Innovations
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Mitigation Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare authorities in Saudi Arabia
- Review of published articles and journals on neurointerventional devices
- Examination of trade publications and industry white papers relevant to neurointervention
Primary Research
- Interviews with neurosurgeons and interventional radiologists in leading hospitals
- Surveys with procurement managers at medical device distributors
- Field interviews with healthcare administrators overseeing neurointerventional departments
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews
- Triangulation of data from clinical studies and market trends
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure data
- Segmentation by device type, including stents, coils, and catheters
- Incorporation of demographic data and prevalence rates of neurological disorders
Bottom-up Modeling
- Collection of sales data from leading neurointerventional device manufacturers
- Estimation of market share based on unit sales and pricing strategies
- Volume x price analysis for each device category to derive total market value
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating technological advancements and healthcare policies
- Scenario modeling based on potential market disruptions and regulatory changes
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 100 | Neurosurgeons, Interventional Radiologists |
| Medical Device Distributors | 60 | Procurement Managers, Sales Directors |
| Healthcare Administrators | 50 | Hospital Administrators, Department Heads |
| Clinical Research Institutions | 40 | Clinical Researchers, Medical Device Evaluators |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Neurointerventional Devices Market?
The Saudi Arabia Neurointerventional Devices Market is valued at approximately USD 135 million, reflecting a significant growth driven by the increasing prevalence of neurological disorders and advancements in minimally invasive medical technology.