About the Report
Base Year 2024Saudi Arabia Prostate Cancer Diagnostics Market Overview
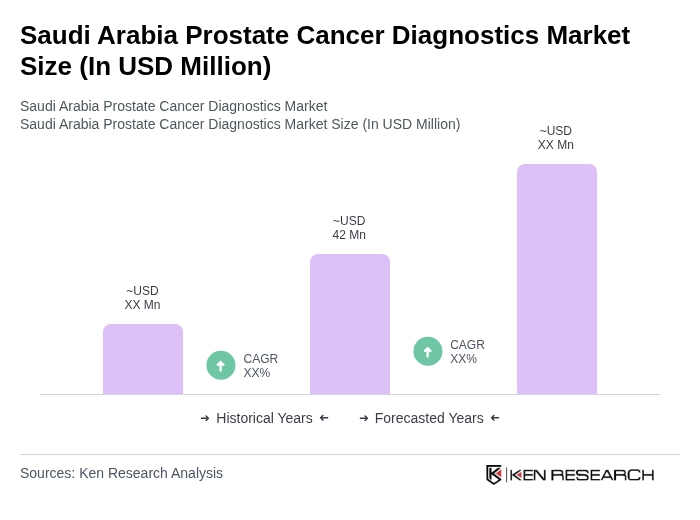
- The Saudi Arabia Prostate Cancer Diagnostics Market is valued at USD 42 million, based on the latest available market data for the diagnostics segment. Growth is primarily driven by the increasing prevalence of prostate cancer, rapid adoption of advanced diagnostic technologies such as MRI and molecular tests, and heightened public awareness regarding early detection and treatment options. The market is further supported by robust government initiatives aimed at expanding healthcare infrastructure, integrating artificial intelligence in diagnostics, and improving access to screening and specialized care services .
- Key cities such as Riyadh, Jeddah, and Dammam continue to dominate the market due to their advanced healthcare infrastructure, high concentration of specialized medical professionals, and presence of leading tertiary care hospitals. These urban centers are equipped with state-of-the-art diagnostic technologies and attract a higher patient influx, significantly contributing to overall market growth. The presence of major healthcare institutions and ongoing investment in medical technology further reinforces their market leadership .
- The National Cancer Control Program, issued by the Ministry of Health in 2023, mandates comprehensive cancer screening, early detection, and treatment protocols for major cancers, including prostate cancer. The program includes dedicated funding for advanced diagnostic technologies, standardized training for healthcare professionals, and the establishment of national screening guidelines, thereby enhancing the overall quality and accessibility of prostate cancer diagnostics across Saudi Arabia .
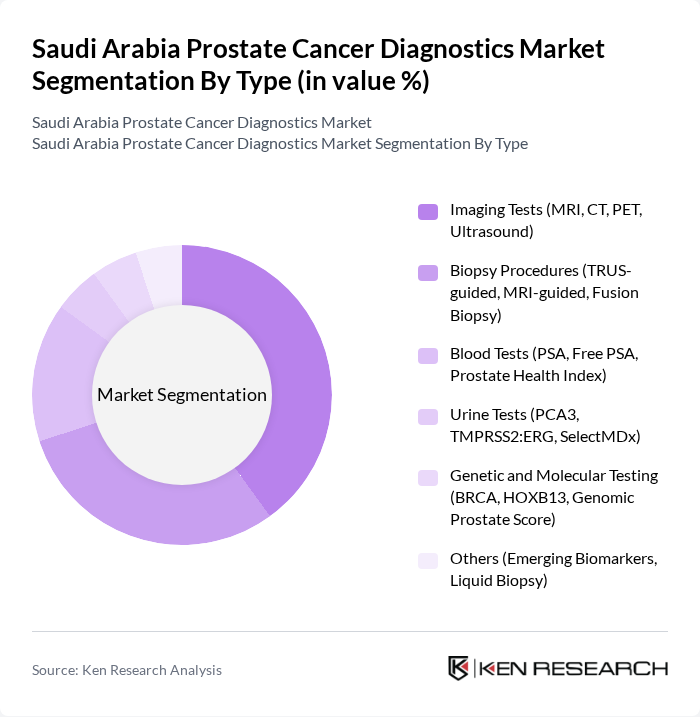
Saudi Arabia Prostate Cancer Diagnostics Market Segmentation
By Type:The market is segmented into various diagnostic types, including imaging tests, biopsy procedures, blood tests, urine tests, genetic and molecular testing, and others. Imaging tests, particularly MRI and CT, are increasingly preferred due to their non-invasive nature and ability to provide detailed visualization of prostate anatomy and pathology. Biopsy procedures, including TRUS-guided and MRI-guided biopsies, remain essential for definitive diagnosis. Blood tests, especially PSA and related indices, are widely used for initial screening and monitoring. Urine-based molecular assays and genetic testing are gaining traction as adjuncts for risk stratification and personalized management .
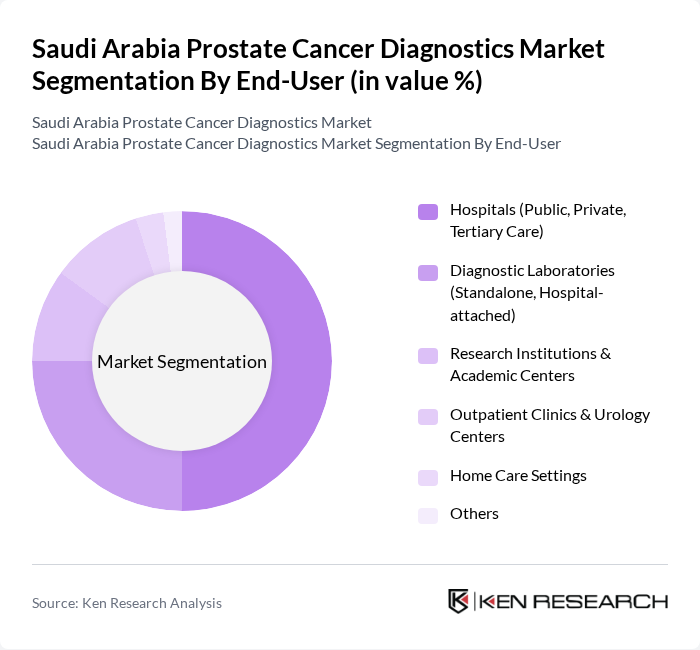
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, research institutions, outpatient clinics, home care settings, and others. Hospitals, especially tertiary care centers, are the leading end-users due to their comprehensive diagnostic and treatment capabilities, access to advanced imaging and laboratory technologies, and multidisciplinary teams. Diagnostic laboratories play a pivotal role by offering specialized testing services and supporting both hospital-based and outpatient care. Research institutions and academic centers are increasingly involved in clinical trials and translational research, while outpatient clinics and home care settings are expanding their roles through telemedicine and decentralized diagnostics .
Saudi Arabia Prostate Cancer Diagnostics Market Competitive Landscape
The Saudi Arabia Prostate Cancer Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Roche Diagnostics, Abbott Laboratories, Becton, Dickinson and Company, Hologic, Inc., Bio-Rad Laboratories, Thermo Fisher Scientific, Philips Healthcare, GE Healthcare, Agilent Technologies, Exact Sciences Corporation, Myriad Genetics, Illumina, Inc., PerkinElmer, Inc., Sysmex Corporation, Quest Diagnostics, LabCorp (Laboratory Corporation of America Holdings), Al Borg Diagnostics (Saudi Arabia), National Unified Procurement Company (NUPCO), King Faisal Specialist Hospital & Research Centre contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Prostate Cancer Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Prostate Cancer:The incidence of prostate cancer in Saudi Arabia has been rising, with approximately1,200 new cases reported annually. This increase is attributed to factors such as an aging population and lifestyle changes. The World Health Organization (WHO) estimates that prostate cancer accounts forabout 7% of all cancer cases in the country, driving demand for effective diagnostic solutions and early detection methods.
- Advancements in Diagnostic Technologies:The Saudi healthcare sector is witnessing significant technological advancements, particularly in prostate cancer diagnostics. Innovations such as MRI-targeted biopsies and liquid biopsy techniques are becoming more prevalent. In future, the market for advanced diagnostic tools was valued at approximately SAR150 million, with expectations for continued growth as hospitals and clinics adopt these technologies to improve diagnostic accuracy and patient outcomes.
- Government Initiatives for Cancer Awareness:The Saudi government has launched several initiatives aimed at increasing cancer awareness and screening. The National Cancer Control Program allocatesapproximately SAR 200 million annuallyfor public education and screening campaigns. These efforts are expected to enhance early detection rates, with a target of screening50% of the eligible population, thereby boosting the demand for prostate cancer diagnostics.
Market Challenges
- High Cost of Advanced Diagnostic Tools:The adoption of advanced diagnostic technologies is hindered by their high costs, which can exceedSAR 100,000for certain imaging equipment. This financial barrier limits access for many healthcare facilities, particularly in rural areas. As a result, the disparity in diagnostic capabilities between urban and rural regions remains a significant challenge, affecting overall patient outcomes and early detection rates.
- Lack of Awareness Among the Population:Despite government initiatives, a significant portion of the Saudi population remains unaware of prostate cancer risks and the importance of early screening. A recent survey indicated that only30%of men over 50 had undergone screening. This lack of awareness contributes to late-stage diagnoses, complicating treatment options and increasing healthcare costs, thereby posing a challenge to market growth.
Saudi Arabia Prostate Cancer Diagnostics Market Future Outlook
The future of the prostate cancer diagnostics market in Saudi Arabia appears promising, driven by technological advancements and increased government support. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency. Additionally, the expansion of telemedicine services will facilitate access to diagnostic consultations, particularly in underserved areas. As awareness campaigns continue to educate the population, early detection rates are likely to improve, positively impacting patient outcomes and market growth.
Market Opportunities
- Growing Demand for Early Detection:There is a rising demand for early detection methods, with the Saudi government aiming to increase screening rates by25%. This presents opportunities for diagnostic companies to develop and market innovative screening solutions tailored to the local population, potentially increasing market penetration and improving patient outcomes.
- Expansion of Telemedicine Services:The growth of telemedicine services in Saudi Arabia is creating new avenues for prostate cancer diagnostics. In future, it is projected that telemedicine consultations will increase by40%, allowing patients in remote areas to access diagnostic services. This trend offers significant opportunities for companies to leverage technology in providing remote diagnostic solutions and consultations.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Imaging Tests (MRI, CT, PET, Ultrasound) Biopsy Procedures (TRUS-guided, MRI-guided, Fusion Biopsy) Blood Tests (PSA, Free PSA, Prostate Health Index) Urine Tests (PCA3, TMPRSS2:ERG, SelectMDx) Genetic and Molecular Testing (BRCA, HOXB13, Genomic Prostate Score) Others (Emerging Biomarkers, Liquid Biopsy) |
| By End-User | Hospitals (Public, Private, Tertiary Care) Diagnostic Laboratories (Standalone, Hospital-attached) Research Institutions & Academic Centers Outpatient Clinics & Urology Centers Home Care Settings Others |
| By Age Group | 50 Years 60 Years 70 Years Above 70 Years |
| By Diagnostic Method | Digital Rectal Examination (DRE) Prostate-Specific Antigen (PSA) Testing MRI-guided Biopsy Transrectal Ultrasound (TRUS) Genetic/Molecular Biomarker Testing Others |
| By Region | Central Region Eastern Region Western Region Southern Region Northern Region |
| By Patient Demographics | Male Patients Family History/Genetic Risk Groups Others |
| By Insurance Coverage | Private Insurance Public Insurance Out-of-Pocket Payments Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Manufacturers and Producers of Diagnostic Equipment
Distributors and Retailers of Medical Devices
Healthcare Providers and Hospitals
Pharmaceutical Companies
Health Insurance Companies
Biotechnology Firms
Players Mentioned in the Report:
Siemens Healthineers
Roche Diagnostics
Abbott Laboratories
Becton, Dickinson and Company
Hologic, Inc.
Bio-Rad Laboratories
Thermo Fisher Scientific
Philips Healthcare
GE Healthcare
Agilent Technologies
Exact Sciences Corporation
Myriad Genetics
Illumina, Inc.
PerkinElmer, Inc.
Sysmex Corporation
Quest Diagnostics
LabCorp (Laboratory Corporation of America Holdings)
Al Borg Diagnostics (Saudi Arabia)
National Unified Procurement Company (NUPCO)
King Faisal Specialist Hospital & Research Centre
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Prostate Cancer Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Prostate Cancer Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Prostate Cancer Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of prostate cancer
3.1.2 Advancements in diagnostic technologies
3.1.3 Government initiatives for cancer awareness
3.1.4 Rising healthcare expenditure
3.2 Market Challenges
3.2.1 High cost of advanced diagnostic tools
3.2.2 Limited access to healthcare facilities in rural areas
3.2.3 Lack of awareness among the population
3.2.4 Regulatory hurdles in product approvals
3.3 Market Opportunities
3.3.1 Growing demand for early detection
3.3.2 Expansion of telemedicine services
3.3.3 Collaborations with international research institutions
3.3.4 Development of personalized medicine approaches
3.4 Market Trends
3.4.1 Shift towards non-invasive diagnostic methods
3.4.2 Integration of AI in diagnostic processes
3.4.3 Increasing focus on patient-centric care
3.4.4 Rise in home-based testing solutions
3.5 Government Regulation
3.5.1 Implementation of national cancer control programs
3.5.2 Stricter regulations on medical device approvals
3.5.3 Guidelines for prostate cancer screening
3.5.4 Policies promoting research and development in oncology
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Prostate Cancer Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Prostate Cancer Diagnostics Market Segmentation
8.1 By Type
8.1.1 Imaging Tests (MRI, CT, PET, Ultrasound)
8.1.2 Biopsy Procedures (TRUS-guided, MRI-guided, Fusion Biopsy)
8.1.3 Blood Tests (PSA, Free PSA, Prostate Health Index)
8.1.4 Urine Tests (PCA3, TMPRSS2:ERG, SelectMDx)
8.1.5 Genetic and Molecular Testing (BRCA, HOXB13, Genomic Prostate Score)
8.1.6 Others (Emerging Biomarkers, Liquid Biopsy)
8.2 By End-User
8.2.1 Hospitals (Public, Private, Tertiary Care)
8.2.2 Diagnostic Laboratories (Standalone, Hospital-attached)
8.2.3 Research Institutions & Academic Centers
8.2.4 Outpatient Clinics & Urology Centers
8.2.5 Home Care Settings
8.2.6 Others
8.3 By Age Group
8.3.1 40-50 Years
8.3.2 51-60 Years
8.3.3 61-70 Years
8.3.4 Above 70 Years
8.4 By Diagnostic Method
8.4.1 Digital Rectal Examination (DRE)
8.4.2 Prostate-Specific Antigen (PSA) Testing
8.4.3 MRI-guided Biopsy
8.4.4 Transrectal Ultrasound (TRUS)
8.4.5 Genetic/Molecular Biomarker Testing
8.4.6 Others
8.5 By Region
8.5.1 Central Region
8.5.2 Eastern Region
8.5.3 Western Region
8.5.4 Southern Region
8.5.5 Northern Region
8.6 By Patient Demographics
8.6.1 Male Patients
8.6.2 Family History/Genetic Risk Groups
8.6.3 Others
8.7 By Insurance Coverage
8.7.1 Private Insurance
8.7.2 Public Insurance
8.7.3 Out-of-Pocket Payments
8.7.4 Others
9. Saudi Arabia Prostate Cancer Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (Saudi Arabia diagnostics segment)
9.2.4 Market Penetration Rate (share of prostate cancer diagnostics in KSA)
9.2.5 Number of Prostate Cancer Diagnostic Tests Performed (annual, KSA)
9.2.6 Product Portfolio Breadth (number of prostate cancer diagnostic products)
9.2.7 Regulatory Approvals (SFDA, CE, FDA for KSA market)
9.2.8 Local Partnerships/Distribution Network Strength
9.2.9 R&D Investment in Prostate Cancer Diagnostics (as % of revenue)
9.2.10 Customer Satisfaction Index (KSA healthcare providers)
9.2.11 Pricing Strategy (premium, value-based, competitive)
9.2.12 Innovation Index (new product launches, patents in KSA)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Siemens Healthineers
9.5.2 Roche Diagnostics
9.5.3 Abbott Laboratories
9.5.4 Becton, Dickinson and Company
9.5.5 Hologic, Inc.
9.5.6 Bio-Rad Laboratories
9.5.7 Thermo Fisher Scientific
9.5.8 Philips Healthcare
9.5.9 GE Healthcare
9.5.10 Agilent Technologies
9.5.11 Exact Sciences Corporation
9.5.12 Myriad Genetics
9.5.13 Illumina, Inc.
9.5.14 PerkinElmer, Inc.
9.5.15 Sysmex Corporation
9.5.16 Quest Diagnostics
9.5.17 LabCorp (Laboratory Corporation of America Holdings)
9.5.18 Al Borg Diagnostics (Saudi Arabia)
9.5.19 National Unified Procurement Company (NUPCO)
9.5.20 King Faisal Specialist Hospital & Research Centre
10. Saudi Arabia Prostate Cancer Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Defense
10.1.4 Ministry of Interior
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Research and Development Funding
10.2.3 Public-Private Partnerships
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Centers
10.3.3 Patients
10.3.4 Healthcare Providers
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Education Needs
10.4.3 Technology Acceptance
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Patient Outcome Improvements
10.5.3 Scalability of Solutions
10.5.4 Others
11. Saudi Arabia Prostate Cancer Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and cancer registries in Saudi Arabia
- Review of academic journals and articles focusing on prostate cancer diagnostics and treatment trends
- Examination of market reports and white papers from healthcare consulting firms
Primary Research
- Interviews with oncologists and urologists specializing in prostate cancer
- Surveys conducted with hospital administrators regarding diagnostic equipment usage
- Focus groups with patients to understand their experiences and preferences in diagnostics
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data with insights from healthcare policy documents
- Sanity checks through feedback from a panel of medical professionals and industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure on cancer diagnostics
- Segmentation of the market by diagnostic methods, including imaging, biopsy, and blood tests
- Incorporation of government health initiatives aimed at early detection and screening programs
Bottom-up Modeling
- Collection of sales data from leading diagnostic equipment manufacturers in Saudi Arabia
- Estimation of market share based on the volume of diagnostic tests performed annually
- Cost analysis of diagnostic procedures to establish pricing models for various tests
Forecasting & Scenario Analysis
- Utilization of historical growth rates in prostate cancer diagnoses to project future trends
- Scenario modeling based on potential changes in healthcare policies and patient demographics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologist Insights | 60 | Oncologists, Urologists |
| Hospital Administration Feedback | 50 | Hospital Administrators, Procurement Managers |
| Patient Experience Surveys | 70 | Prostate Cancer Patients, Caregivers |
| Diagnostic Equipment Providers | 40 | Sales Managers, Product Specialists |
| Healthcare Policy Experts | 40 | Health Economists, Policy Analysts |
Frequently Asked Questions
What is the current value of the Saudi Arabia Prostate Cancer Diagnostics Market?
The Saudi Arabia Prostate Cancer Diagnostics Market is valued at approximately USD 42 million, driven by the increasing prevalence of prostate cancer and advancements in diagnostic technologies such as MRI and molecular tests.