About the Report
Base Year 2024US Antisense and RNI Therapeutic Market Overview
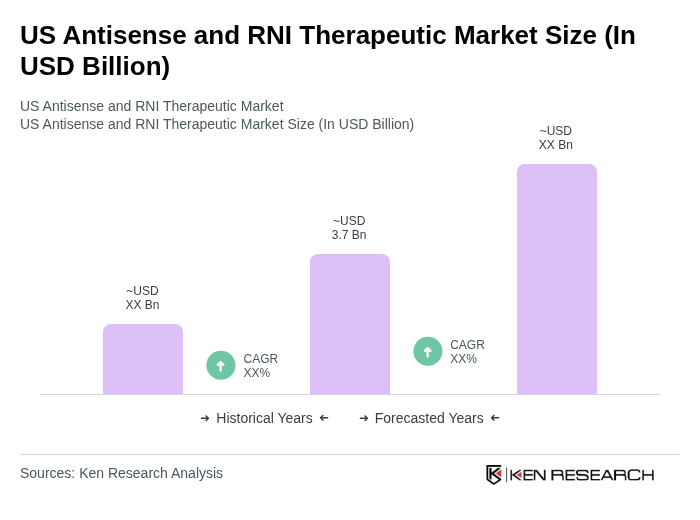
- The US Antisense and RNI Therapeutic Market is valued at USD 3.7 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in genetic research, the increasing prevalence of chronic and rare diseases, and the rising demand for personalized medicine. The market is also supported by significant investments in research and development, leading to the introduction of innovative therapies that target specific genetic disorders. Recent trends highlight the expansion of clinical trial activity, FDA approvals for RNA-based drugs, and increased partnerships between biotech firms and academic institutions, further accelerating market momentum .
- Key players in this market include major pharmaceutical hubs such as California, Massachusetts, and New York. These regions dominate due to their robust healthcare infrastructure, the presence of leading biotech firms, and strong academic institutions that foster research and innovation. The concentration of venture capital funding in these areas further accelerates the development of antisense and RNA interference therapeutics. North America, particularly the United States, holds a leading share in the global market, driven by early adoption of advanced therapies and proactive regulatory support .
- The "21st Century Cures Act," enacted by the US Congress in 2016 and implemented by the US Department of Health and Human Services, aims to accelerate the discovery, development, and delivery of new treatments. This regulation encourages innovation in gene therapy and RNA-based therapeutics by providing funding for research initiatives and streamlining the approval process for new therapies, thereby enhancing patient access to cutting-edge treatments. The Act specifically mandates the FDA to modernize clinical trial designs, support regenerative medicine, and expand the use of real-world evidence in regulatory decisions .
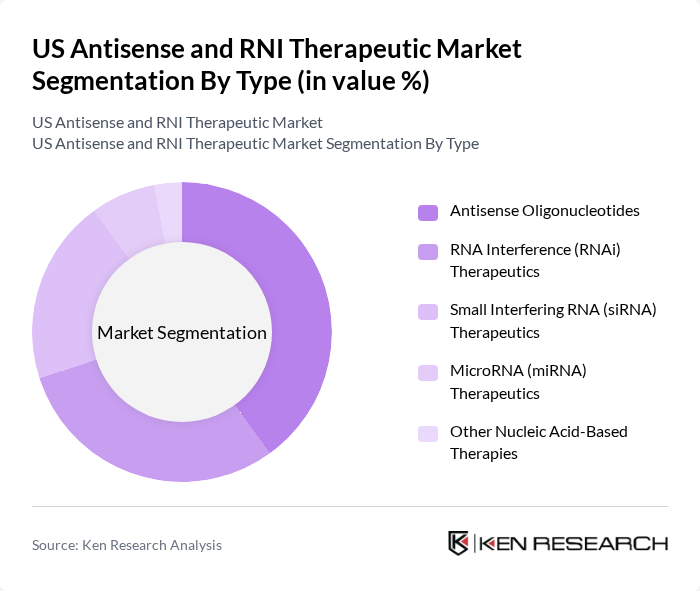
US Antisense and RNI Therapeutic Market Segmentation
By Type:The market is segmented into various types, including Antisense Oligonucleotides, RNA Interference (RNAi) Therapeutics, Small Interfering RNA (siRNA) Therapeutics, MicroRNA (miRNA) Therapeutics, and Other Nucleic Acid-Based Therapies. Among these, Antisense Oligonucleotides are leading the market due to their effectiveness in targeting specific genes associated with diseases, which has garnered significant attention from both researchers and clinicians. The growing number of clinical trials and successful product launches in this segment further solidifies its dominance.
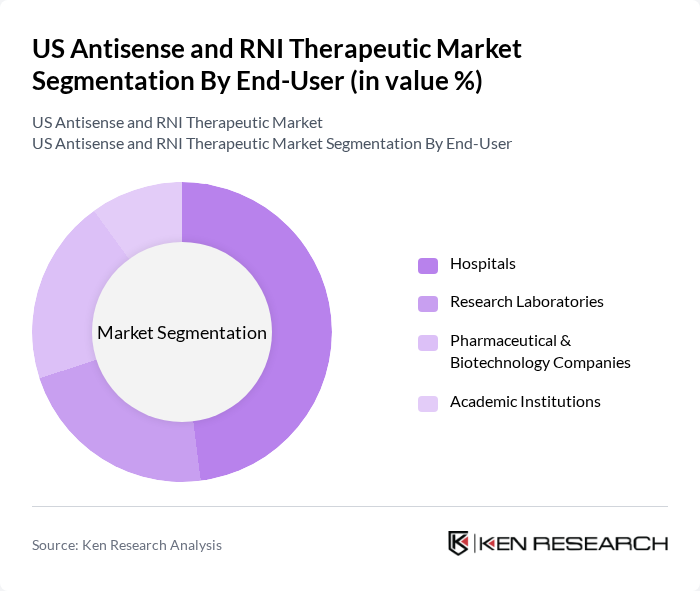
By End-User:The end-user segmentation includes Hospitals, Research Laboratories, Pharmaceutical & Biotechnology Companies, and Academic Institutions. Hospitals are the leading end-users, driven by the increasing adoption of advanced therapeutic options for treating complex diseases. The demand for personalized medicine in clinical settings has led to a surge in the utilization of antisense and RNAi therapies, making hospitals a critical segment in this market.
US Antisense and RNI Therapeutic Market Competitive Landscape
The US Antisense and RNI Therapeutic Market is characterized by a dynamic mix of regional and international players. Leading participants such as Ionis Pharmaceuticals, Alnylam Pharmaceuticals, Sarepta Therapeutics, Regulus Therapeutics, Wave Life Sciences, Dicerna Pharmaceuticals, Avidity Biosciences, Moderna, Inc., BioMarin Pharmaceutical, Pfizer Inc., Novartis AG, Roche Holding AG, Bristol-Myers Squibb, GSK (GlaxoSmithKline), Merck & Co., Inc., Sirnaomics, Inc., Arrowhead Pharmaceuticals, Silence Therapeutics, Biogen Inc., Sanofi S.A. contribute to innovation, geographic expansion, and service delivery in this space.
US Antisense and RNI Therapeutic Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders:The US has seen a significant rise in genetic disorders, with approximately 1 in 3,500 births affected by cystic fibrosis alone, according to the CDC. This growing prevalence drives demand for innovative therapies, including antisense and RNA interference (RNI) treatments. The National Institutes of Health (NIH) reported that over 7,000 rare genetic disorders exist, creating a substantial market for targeted therapies that address these conditions effectively.
- Advancements in RNA Technology:The US has invested heavily in RNA technology, with funding exceeding $1 billion for research and development. Breakthroughs in RNA-based therapies, such as mRNA vaccines, have demonstrated the potential of this technology. The National Science Foundation (NSF) reported a significant increase in RNA research publications, indicating a robust growth trajectory that supports the expansion of antisense and RNI therapeutics in clinical applications.
- Rising Investment in Biotechnology:The biotechnology sector in the US attracted over $50 billion in venture capital funding, reflecting a strong investor interest in innovative therapies. This influx of capital is crucial for the development of antisense and RNI therapeutics, enabling companies to advance their research pipelines. The Biotechnology Innovation Organization (BIO) noted that a majority of this funding is directed towards genetic and rare disease therapies, underscoring the market's growth potential.
Market Challenges
- High Development Costs:The average cost to develop a new drug in the US has reached approximately $2.6 billion, according to the Tufts Center for the Study of Drug Development. This financial burden poses a significant challenge for companies focusing on antisense and RNI therapeutics, as the lengthy development timelines and complex regulatory processes can deter investment. The high costs can limit the number of viable products entering the market, impacting overall growth.
- Regulatory Hurdles:Navigating the regulatory landscape for gene therapies can be daunting, with the FDA requiring extensive clinical trial data before approval. The FDA receives a substantial number of investigational new drug applications, highlighting the competitive nature of the market. The lengthy review process can delay product launches, and companies often face challenges in meeting stringent safety and efficacy standards, which can hinder market entry for new therapies.
US Antisense and RNI Therapeutic Market Future Outlook
The future of the US antisense and RNI therapeutic market appears promising, driven by technological advancements and increasing healthcare demands. The integration of artificial intelligence in drug discovery is expected to streamline the development process, enhancing efficiency. Additionally, the shift towards preventive healthcare will likely increase the focus on genetic therapies, as more patients seek personalized treatment options. As the market evolves, collaboration between biotech firms and research institutions will be crucial for innovation and addressing unmet medical needs.
Market Opportunities
- Expansion into Emerging Markets:The growing healthcare infrastructure in emerging markets presents significant opportunities for antisense and RNI therapeutics. Countries like India and Brazil are investing heavily in biotechnology, with healthcare spending projected to reach $300 billion in future. This expansion allows US companies to tap into new patient populations and diversify their market presence, driving growth in the sector.
- Collaborations with Research Institutions:Partnerships with academic and research institutions can accelerate the development of antisense and RNI therapies. Over 200 collaborations have been reported between biotech firms and universities, focusing on genetic research. These collaborations can enhance innovation, provide access to cutting-edge technologies, and facilitate clinical trials, ultimately leading to faster market entry for new therapies.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Antisense Oligonucleotides RNA Interference (RNAi) Therapeutics Small Interfering RNA (siRNA) Therapeutics MicroRNA (miRNA) Therapeutics Other Nucleic Acid-Based Therapies |
| By End-User | Hospitals Research Laboratories Pharmaceutical & Biotechnology Companies Academic Institutions |
| By Application | Oncology (Cancer Treatment) Neurodegenerative Disorders (e.g., Alzheimer's, Parkinson's) Cardiovascular Diseases Genetic Disorders (e.g., Duchenne Muscular Dystrophy, Spinal Muscular Atrophy) Infectious Diseases Renal Disorders Respiratory Disorders |
| By Delivery Method | Intravenous Injection Subcutaneous Injection Intrathecal Injection Oral Delivery Other Delivery Methods |
| By Therapeutic Area | Oncology Neurology Hematology Cardiometabolic & Renal Disorders Ocular Disorders Other Therapeutic Areas |
| By Region | Northeast Midwest South West |
| By Others | Emerging Technologies Combination Therapies Patient-Centric Innovations |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, National Institutes of Health)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Research Organizations
Healthcare Providers and Institutions
Health Insurance Companies
Pharmaceutical Supply Chain Stakeholders
Players Mentioned in the Report:
Ionis Pharmaceuticals
Alnylam Pharmaceuticals
Sarepta Therapeutics
Regulus Therapeutics
Wave Life Sciences
Dicerna Pharmaceuticals
Avidity Biosciences
Moderna, Inc.
BioMarin Pharmaceutical
Pfizer Inc.
Novartis AG
Roche Holding AG
Bristol-Myers Squibb
GSK (GlaxoSmithKline)
Merck & Co., Inc.
Sirnaomics, Inc.
Arrowhead Pharmaceuticals
Silence Therapeutics
Biogen Inc.
Sanofi S.A.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Antisense and RNI Therapeutic Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Antisense and RNI Therapeutic Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Antisense and RNI Therapeutic Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of genetic disorders
3.1.2 Advancements in RNA technology
3.1.3 Rising investment in biotechnology
3.1.4 Growing demand for personalized medicine
3.2 Market Challenges
3.2.1 High development costs
3.2.2 Regulatory hurdles
3.2.3 Limited awareness among healthcare providers
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with research institutions
3.3.3 Development of novel delivery systems
3.3.4 Increasing focus on orphan drugs
3.4 Market Trends
3.4.1 Rise of gene editing technologies
3.4.2 Integration of AI in drug discovery
3.4.3 Shift towards preventive healthcare
3.4.4 Growing emphasis on patient-centric approaches
3.5 Government Regulation
3.5.1 FDA guidelines for gene therapies
3.5.2 Orphan Drug Act incentives
3.5.3 Clinical trial regulations
3.5.4 Pricing and reimbursement policies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Antisense and RNI Therapeutic Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Antisense and RNI Therapeutic Market Segmentation
8.1 By Type
8.1.1 Antisense Oligonucleotides
8.1.2 RNA Interference (RNAi) Therapeutics
8.1.3 Small Interfering RNA (siRNA) Therapeutics
8.1.4 MicroRNA (miRNA) Therapeutics
8.1.5 Other Nucleic Acid-Based Therapies
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Laboratories
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Academic Institutions
8.3 By Application
8.3.1 Oncology (Cancer Treatment)
8.3.2 Neurodegenerative Disorders (e.g., Alzheimer's, Parkinson's)
8.3.3 Cardiovascular Diseases
8.3.4 Genetic Disorders (e.g., Duchenne Muscular Dystrophy, Spinal Muscular Atrophy)
8.3.5 Infectious Diseases
8.3.6 Renal Disorders
8.3.7 Respiratory Disorders
8.4 By Delivery Method
8.4.1 Intravenous Injection
8.4.2 Subcutaneous Injection
8.4.3 Intrathecal Injection
8.4.4 Oral Delivery
8.4.5 Other Delivery Methods
8.5 By Therapeutic Area
8.5.1 Oncology
8.5.2 Neurology
8.5.3 Hematology
8.5.4 Cardiometabolic & Renal Disorders
8.5.5 Ocular Disorders
8.5.6 Other Therapeutic Areas
8.6 By Region
8.6.1 Northeast
8.6.2 Midwest
8.6.3 South
8.6.4 West
8.7 By Others
8.7.1 Emerging Technologies
8.7.2 Combination Therapies
8.7.3 Patient-Centric Innovations
9. US Antisense and RNI Therapeutic Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (YoY %)
9.2.4 Market Penetration Rate (US market share %)
9.2.5 Pipeline Strength (Number of clinical-stage programs)
9.2.6 R&D Investment as a Percentage of Revenue
9.2.7 Regulatory Approval Success Rate (%)
9.2.8 Time to Market (Average years from IND to approval)
9.2.9 Commercialization Partnerships (Number of major alliances)
9.2.10 Product Portfolio Diversity (Number of approved indications)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Ionis Pharmaceuticals
9.5.2 Alnylam Pharmaceuticals
9.5.3 Sarepta Therapeutics
9.5.4 Regulus Therapeutics
9.5.5 Wave Life Sciences
9.5.6 Dicerna Pharmaceuticals
9.5.7 Avidity Biosciences
9.5.8 Moderna, Inc.
9.5.9 BioMarin Pharmaceutical
9.5.10 Pfizer Inc.
9.5.11 Novartis AG
9.5.12 Roche Holding AG
9.5.13 Bristol-Myers Squibb
9.5.14 GSK (GlaxoSmithKline)
9.5.15 Merck & Co., Inc.
9.5.16 Sirnaomics, Inc.
9.5.17 Arrowhead Pharmaceuticals
9.5.18 Silence Therapeutics
9.5.19 Biogen Inc.
9.5.20 Sanofi S.A.
10. US Antisense and RNI Therapeutic Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Contracting Practices
10.1.4 Evaluation Criteria for Therapeutics
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Biotech
10.2.2 Funding Sources
10.2.3 Infrastructure Development Needs
10.2.4 Collaboration with Private Sector
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Innovative Therapies
10.3.2 Cost Management Challenges
10.3.3 Regulatory Compliance Issues
10.3.4 Patient Education Gaps
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Integration Challenges
10.4.3 Awareness of Antisense Therapies
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Long-term Cost Savings
10.5.3 Expansion into New Indications
10.5.4 Patient Satisfaction Metrics
11. US Antisense and RNI Therapeutic Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Model Exploration
1.4 Key Partnerships Identification
1.5 Customer Segmentation Analysis
1.6 Competitive Landscape Overview
1.7 Risk Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Event Marketing Plans
2.7 Feedback and Adaptation Mechanisms
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Partnership with Healthcare Providers
3.6 Logistics and Supply Chain Management
3.7 Distribution Network Optimization
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discount and Promotion Strategies
4.7 Price Sensitivity Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends Identification
5.4 Feedback from Healthcare Professionals
5.5 Patient Needs Assessment
5.6 Market Entry Opportunities
5.7 Innovation Potential
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Engagement Strategies
6.4 Feedback Collection Mechanisms
6.5 Relationship Management Tools
6.6 Community Building Initiatives
6.7 Customer Education Programs
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
7.5 Competitive Advantages
7.6 Long-term Value Creation
7.7 Market Differentiation Strategies
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Market Research Activities
8.5 Training and Development Programs
8.6 Stakeholder Engagement
8.7 Performance Monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Fit Evaluation
10.7 Long-term Viability Analysis
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Funding Sources
11.4 Financial Projections
11.5 Risk Mitigation Strategies
11.6 Resource Allocation Plans
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Assessment Framework
12.3 Control Mechanisms
12.4 Partnership Evaluation Criteria
12.5 Long-term Strategy Alignment
12.6 Exit Strategy Considerations
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Profit Margin Projections
13.4 Revenue Growth Forecasts
13.5 Cost Management Strategies
13.6 Financial Health Indicators
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Research Collaborations
14.6 Technology Partners
14.7 Funding Partners
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from industry associations such as the Antisense Therapeutics Association
- Review of scientific literature and clinical trial data from databases like PubMed and ClinicalTrials.gov
- Examination of regulatory filings and approvals from the FDA related to antisense and RNA interference therapies
Primary Research
- Interviews with key opinion leaders (KOLs) in the field of molecular therapeutics
- Surveys targeting pharmaceutical executives involved in R&D and product development
- Focus groups with healthcare professionals to gather insights on treatment adoption and patient outcomes
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert interviews
- Triangulation of quantitative data with qualitative insights from industry experts
- Sanity checks through peer reviews and feedback from advisory panels
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market (TAM) based on healthcare expenditure and disease prevalence
- Segmentation of the market by therapeutic area, including oncology, genetic disorders, and infectious diseases
- Incorporation of trends in personalized medicine and advancements in gene therapy
Bottom-up Modeling
- Collection of sales data from leading companies in the antisense and RNAi sectors
- Estimation of market share based on product launches and pipeline analysis
- Volume and pricing analysis for existing therapies to project future revenue streams
Forecasting & Scenario Analysis
- Development of predictive models using historical growth rates and emerging market trends
- Scenario analysis based on potential regulatory changes and technological advancements
- Creation of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Therapeutics | 100 | Oncologists, Clinical Researchers |
| Genetic Disorders Treatment | 60 | Geneticists, Rare Disease Specialists |
| Infectious Disease Applications | 50 | Infectious Disease Physicians, Pharmacologists |
| Regulatory Insights | 40 | Regulatory Affairs Managers, Compliance Officers |
| Market Access Strategies | 70 | Market Access Managers, Health Economists |
Frequently Asked Questions
What is the current value of the US Antisense and RNI Therapeutic Market?
The US Antisense and RNI Therapeutic Market is valued at approximately USD 3.7 billion, driven by advancements in genetic research, the prevalence of chronic diseases, and the demand for personalized medicine.