About the Report
Base Year 2024US Cryoablation Probes Market Overview
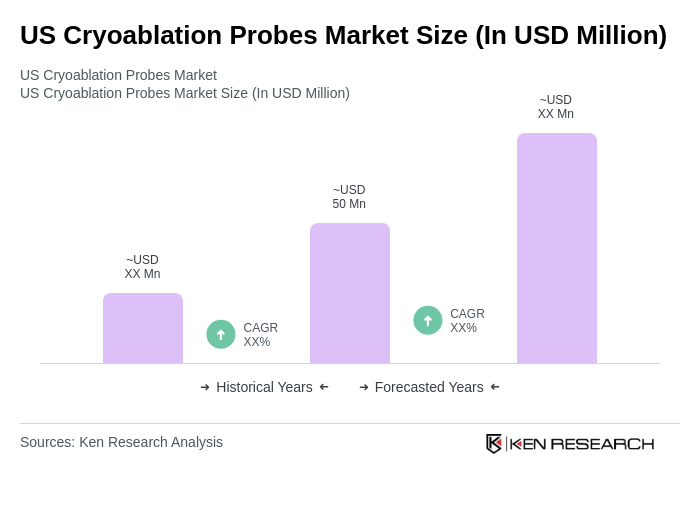
- The US Cryoablation Probes Market is valued at USD 50 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cardiovascular and oncological diseases, advancements in cryoablation technology, and a growing preference for minimally invasive procedures among healthcare providers and patients. The rising demand for effective treatment options for conditions such as cardiac arrhythmias and tumors has further fueled market expansion. Recent trends highlight the adoption of cryoablation for atrial fibrillation and oncology applications, supported by technological innovations in probe design and imaging guidance systems, which enhance procedural safety and efficacy .
- The market is dominated by key regions such as California, New York, and Texas, which are home to leading healthcare facilities and research institutions. These states benefit from a high concentration of advanced medical technology, significant healthcare expenditure, and a robust patient population, contributing to their dominance in the cryoablation probes market .
- In 2023, the US government implemented regulations to enhance the safety and efficacy of medical devices, including cryoablation probes. The Food and Drug Administration (FDA) issued updated guidance under the "Premarket Notification Submissions for Cryosurgical Medical Devices," requiring manufacturers to provide comprehensive clinical and performance data, as well as enhanced post-market surveillance. These requirements ensure that cryoablation probes meet stringent safety, efficacy, and quality standards before and after entering the US healthcare market .
US Cryoablation Probes Market Segmentation
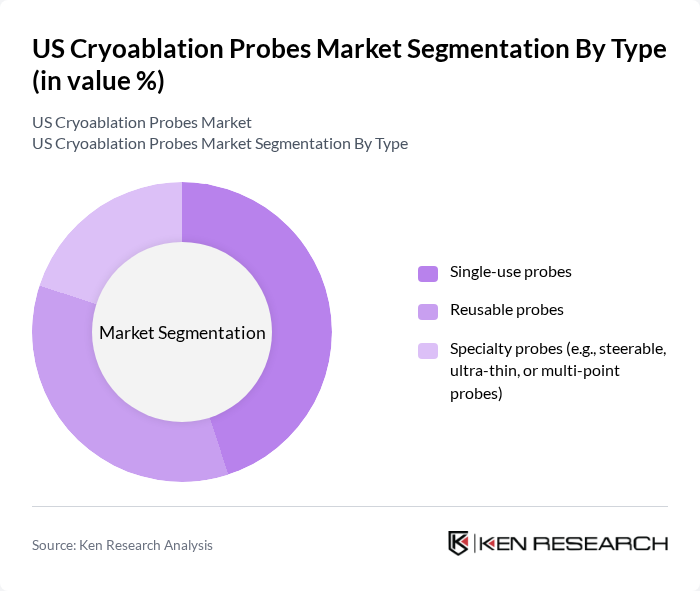
By Type:The cryoablation probes market can be segmented into three main types: Single-use probes, Reusable probes, and Specialty probes (e.g., steerable, ultra-thin, or multi-point probes). Among these, single-use probes are gaining traction due to their convenience and reduced risk of cross-contamination, making them a preferred choice in hospitals and surgical centers. Reusable probes, while cost-effective, face challenges related to sterilization and maintenance. Specialty probes are increasingly being adopted for complex procedures, enhancing precision and efficacy .
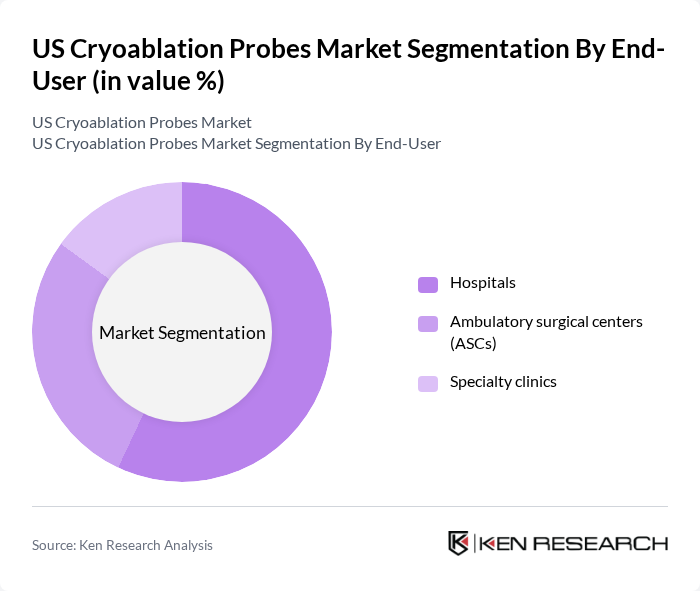
By End-User:The end-user segmentation includes Hospitals, Ambulatory surgical centers (ASCs), and Specialty clinics. Hospitals dominate the market due to their extensive resources and capabilities to perform complex procedures. ASCs are witnessing growth as they offer cost-effective and efficient care, attracting patients seeking outpatient services. Specialty clinics are also emerging as significant players, focusing on specific conditions and providing tailored treatment options .
US Cryoablation Probes Market Competitive Landscape
The US Cryoablation Probes Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Boston Scientific Corporation, Abbott Laboratories, Johnson & Johnson (Biosense Webster), AtriCure, Inc., Varian, A Siemens Healthineers Company, AngioDynamics, Inc., IceCure Medical Ltd., HealthTronics, Inc., Bruker Corporation, Erbe USA, Incorporated, Keeler USA, Merit Medical Systems, Inc., CONMED Corporation, Biotronik, Inc. contribute to innovation, geographic expansion, and service delivery in this space .
US Cryoablation Probes Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiac Arrhythmias:The prevalence of cardiac arrhythmias in the United States is projected to reach approximately 5.5 million cases in future, driven by factors such as an aging population and lifestyle changes. This surge in cases necessitates effective treatment options, including cryoablation, which is recognized for its efficacy in managing arrhythmias. The growing patient base is expected to significantly boost the demand for cryoablation probes, enhancing market growth.
- Advancements in Cryoablation Technology:Technological innovations in cryoablation, such as improved probe designs and enhanced cooling systems, are expected to increase procedure success rates. For instance, the introduction of advanced cryoablation systems has shown a reduction in procedure times by up to 30%. These advancements not only improve patient outcomes but also encourage healthcare providers to adopt cryoablation as a preferred treatment method, further driving market expansion.
- Rising Demand for Minimally Invasive Procedures:The shift towards minimally invasive surgical techniques is evident, with approximately 60% of patients preferring such options due to reduced recovery times and lower complication rates. Cryoablation procedures align with this trend, offering effective treatment with minimal invasiveness. As healthcare systems increasingly prioritize patient-centered care, the demand for cryoablation probes is expected to rise, supporting market growth in the coming years.
Market Challenges
- High Cost of Cryoablation Procedures:The average cost of cryoablation procedures can range from $15,000 to $25,000, which poses a significant barrier for many patients and healthcare providers. This high cost can limit access to treatment, particularly for uninsured or underinsured populations. As a result, the financial burden associated with cryoablation may hinder market growth, as fewer patients can afford the procedure.
- Limited Reimbursement Policies:Reimbursement policies for cryoablation procedures remain inconsistent across various insurance providers. In future, it is estimated that only about 50% of cryoablation procedures will receive full reimbursement, creating financial uncertainty for healthcare providers. This lack of comprehensive coverage can deter hospitals from investing in cryoablation technology, ultimately impacting the market's growth potential.
US Cryoablation Probes Market Future Outlook
The US cryoablation probes market is poised for significant evolution, driven by technological advancements and changing patient preferences. As healthcare systems increasingly adopt outpatient procedures, the demand for cryoablation is expected to rise. Additionally, the integration of artificial intelligence in procedural planning and execution will enhance precision and outcomes. These trends indicate a promising future for the market, with a focus on improving patient care and expanding access to innovative treatment options.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets, particularly in Asia-Pacific and Latin America, present significant growth opportunities for cryoablation probes. With increasing healthcare investments and rising awareness of advanced medical technologies, these regions are expected to see a surge in demand for cryoablation procedures, driving market expansion.
- Development of New Cryoablation Devices:The ongoing research and development of innovative cryoablation devices tailored for specific conditions, such as atrial fibrillation, are expected to create new market opportunities. These advancements can lead to improved efficacy and safety profiles, attracting more healthcare providers to adopt cryoablation technologies.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Single-use probes Reusable probes Specialty probes (e.g., steerable, ultra-thin, or multi-point probes) |
| By End-User | Hospitals Ambulatory surgical centers (ASCs) Specialty clinics |
| By Application | Cardiac arrhythmias (e.g., atrial fibrillation, ventricular tachycardia) Oncology (tumor ablation: lung, liver, kidney, prostate, breast) Pain management (nerve ablation, chronic pain) Dermatology (skin lesion ablation) |
| By Distribution Channel | Direct sales Distributors Online sales |
| By Region | Northeast Midwest South West |
| By Price Range | Low-end Mid-range High-end |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Centers for Medicare & Medicaid Services)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Regulatory Consultants
Industry Associations (e.g., American Medical Association, Medical Device Manufacturers Association)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Medtronic
Boston Scientific Corporation
Abbott Laboratories
Johnson & Johnson (Biosense Webster)
AtriCure, Inc.
Varian, A Siemens Healthineers Company
AngioDynamics, Inc.
IceCure Medical Ltd.
HealthTronics, Inc.
Bruker Corporation
Erbe USA, Incorporated
Keeler USA
Merit Medical Systems, Inc.
CONMED Corporation
Biotronik, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Cryoablation Probes Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Cryoablation Probes Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Cryoablation Probes Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiac arrhythmias
3.1.2 Advancements in cryoablation technology
3.1.3 Rising demand for minimally invasive procedures
3.1.4 Growing geriatric population
3.2 Market Challenges
3.2.1 High cost of cryoablation procedures
3.2.2 Limited reimbursement policies
3.2.3 Competition from alternative ablation techniques
3.2.4 Lack of skilled professionals
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Development of new cryoablation devices
3.3.3 Strategic partnerships and collaborations
3.3.4 Increasing awareness and education on cryoablation
3.4 Market Trends
3.4.1 Integration of AI in cryoablation procedures
3.4.2 Shift towards outpatient procedures
3.4.3 Focus on patient-centric care
3.4.4 Rise in clinical trials for new applications
3.5 Government Regulation
3.5.1 FDA guidelines on cryoablation devices
3.5.2 Regulations on medical device manufacturing
3.5.3 Compliance with safety standards
3.5.4 Reporting requirements for adverse events
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Cryoablation Probes Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Cryoablation Probes Market Segmentation
8.1 By Type
8.1.1 Single-use probes
8.1.2 Reusable probes
8.1.3 Specialty probes (e.g., steerable, ultra-thin, or multi-point probes)
8.2 By End-User
8.2.1 Hospitals
8.2.2 Ambulatory surgical centers (ASCs)
8.2.3 Specialty clinics
8.3 By Application
8.3.1 Cardiac arrhythmias (e.g., atrial fibrillation, ventricular tachycardia)
8.3.2 Oncology (tumor ablation: lung, liver, kidney, prostate, breast)
8.3.3 Pain management (nerve ablation, chronic pain)
8.3.4 Dermatology (skin lesion ablation)
8.4 By Distribution Channel
8.4.1 Direct sales
8.4.2 Distributors
8.4.3 Online sales
8.5 By Region
8.5.1 Northeast
8.5.2 Midwest
8.5.3 South
8.5.4 West
8.6 By Price Range
8.6.1 Low-end
8.6.2 Mid-range
8.6.3 High-end
8.7 Others
9. US Cryoablation Probes Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 US Cryoablation Probe Revenue (USD, latest year)
9.2.4 Revenue Growth Rate (CAGR, 3-5 years)
9.2.5 Market Share (%)
9.2.6 Number of FDA-approved cryoablation products
9.2.7 R&D Investment as % of Revenue
9.2.8 Product Portfolio Breadth (number of probe types/applications)
9.2.9 Distribution Network Coverage (US regions served)
9.2.10 Key Strategic Partnerships or Collaborations
9.2.11 Customer Satisfaction Score (Net Promoter Score or equivalent)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic
9.5.2 Boston Scientific Corporation
9.5.3 Abbott Laboratories
9.5.4 Johnson & Johnson (Biosense Webster)
9.5.5 AtriCure, Inc.
9.5.6 Varian, A Siemens Healthineers Company
9.5.7 AngioDynamics, Inc.
9.5.8 IceCure Medical Ltd.
9.5.9 HealthTronics, Inc.
9.5.10 Bruker Corporation
9.5.11 Erbe USA, Incorporated
9.5.12 Keeler USA
9.5.13 Merit Medical Systems, Inc.
9.5.14 CONMED Corporation
9.5.15 Biotronik, Inc.
10. US Cryoablation Probes Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for medical devices
10.1.2 Decision-making processes
10.1.3 Evaluation criteria for procurement
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in healthcare infrastructure
10.2.2 Funding for medical technology
10.2.3 Trends in corporate healthcare spending
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges faced by hospitals
10.3.2 Issues in outpatient settings
10.3.3 Needs of specialty clinics
10.4 User Readiness for Adoption
10.4.1 Training and education requirements
10.4.2 Technology acceptance levels
10.4.3 Infrastructure readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion into new applications
10.5.3 Long-term benefits analysis
11. US Cryoablation Probes Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Business model development
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from medical device associations and healthcare market research firms
- Review of published articles in peer-reviewed journals focusing on cryoablation technologies and applications
- Examination of regulatory filings and guidelines from the FDA and other relevant health authorities
Primary Research
- Interviews with leading medical professionals specializing in oncology and cardiology
- Surveys conducted with hospital procurement departments regarding cryoablation probe usage
- Field interviews with biomedical engineers involved in the development of cryoablation devices
Validation & Triangulation
- Cross-validation of findings through multiple data sources including clinical trial results and market surveys
- Triangulation of data from healthcare providers, manufacturers, and regulatory bodies
- Sanity checks through expert panel reviews comprising industry veterans and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and investment in medical technologies
- Segmentation by application areas such as oncology, cardiology, and pain management
- Incorporation of trends in minimally invasive procedures and technological advancements in cryoablation
Bottom-up Modeling
- Volume estimates derived from sales data of leading cryoablation probe manufacturers
- Operational cost analysis based on pricing models and reimbursement rates for cryoablation procedures
- Calculation of market size based on the number of procedures performed annually and average device usage per procedure
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as aging population, cancer incidence rates, and healthcare policies
- Scenario modeling based on potential advancements in cryoablation technology and shifts in treatment paradigms
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Departments | 60 | Oncologists, Medical Directors |
| Cardiology Clinics | 50 | Cardiologists, Interventional Specialists |
| Hospital Procurement Teams | 45 | Procurement Managers, Supply Chain Coordinators |
| Biomedical Engineering Firms | 40 | Product Development Engineers, R&D Managers |
| Healthcare Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
Frequently Asked Questions
What is the current value of the US Cryoablation Probes Market?
The US Cryoablation Probes Market is valued at approximately USD 50 million, reflecting a five-year historical analysis. This growth is driven by the rising prevalence of cardiovascular and oncological diseases and advancements in cryoablation technology.