About the Report
Base Year 2024US Pharmaceutical Regulatory Affairs Market Overview
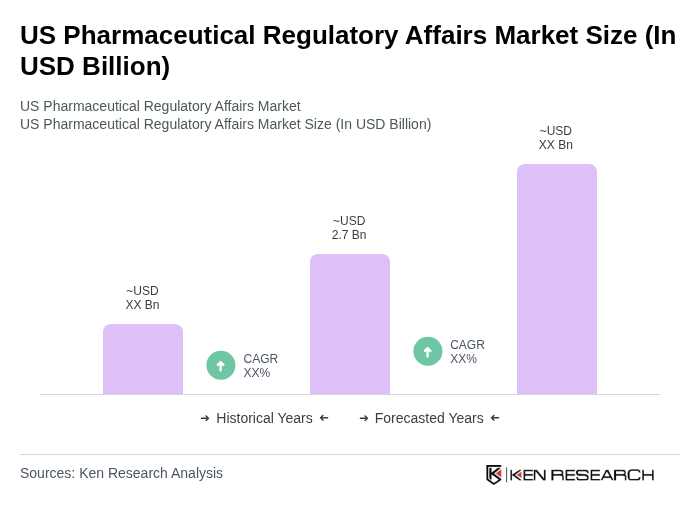
- The US Pharmaceutical Regulatory Affairs Market is valued at USD 2.7 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing complexity of regulatory requirements, the rise in drug development activities, and the growing emphasis on compliance and safety in the pharmaceutical industry. Major drivers include the expansion of biologics and personalized medicine, the proliferation of advanced therapies, and the growing trend toward regulatory affairs outsourcing to manage evolving compliance demands .
- Key players in this market are concentrated in major cities such as New York, San Francisco, and Boston, which dominate due to their high density of pharmaceutical companies, research institutions, and regulatory agencies. These cities serve as hubs for innovation and possess robust infrastructure supporting drug development and regulatory processes .
- The regulatory framework governing pharmaceutical affairs in the US is primarily defined by the Federal Food, Drug, and Cosmetic Act (FD&C Act), administered by the US Food and Drug Administration (FDA). The FD&C Act, most recently amended by the FDA Reauthorization Act of 2017, outlines requirements for drug approval, clinical trials, labeling, and post-market surveillance. Compliance with these regulations is mandatory for all pharmaceutical manufacturers and sponsors seeking market authorization in the United States .
US Pharmaceutical Regulatory Affairs Market Segmentation
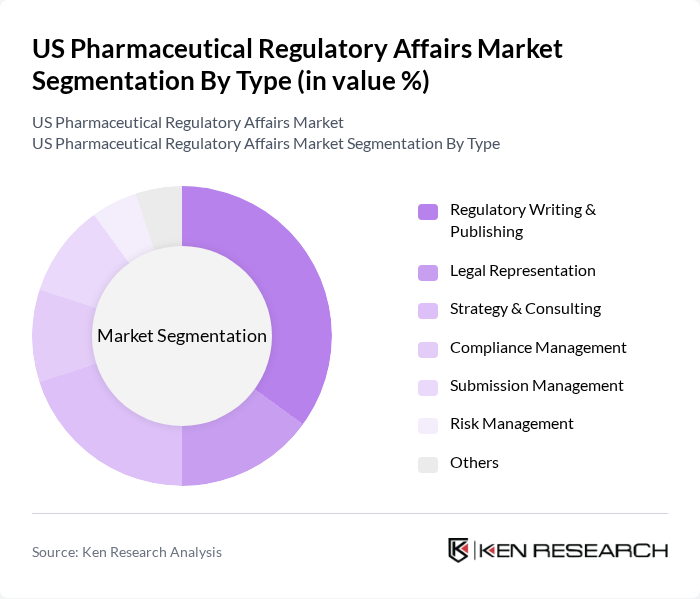
By Type:The market is segmented into various types, including Regulatory Writing & Publishing, Legal Representation, Strategy & Consulting, Compliance Management, Submission Management, Risk Management, and Others. Each of these segments plays a crucial role in ensuring that pharmaceutical products meet regulatory standards and are safe for public use. Among these, Regulatory Writing & Publishing is the most dominant segment, driven by the increasing need for comprehensive documentation and submission of regulatory filings. Outsourcing of regulatory affairs services, particularly in regulatory writing and publishing, is a key trend as companies seek specialized expertise to navigate complex and evolving requirements .
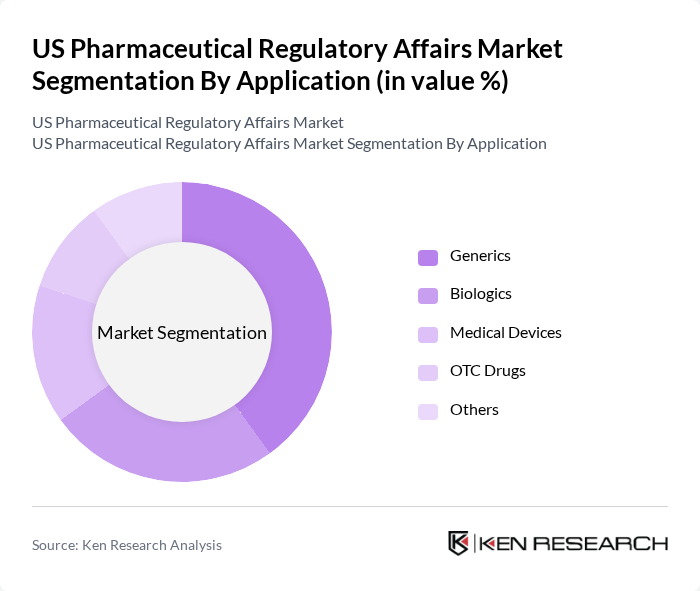
By Application:The market is further segmented by application into Generics, Biologics, Medical Devices, OTC Drugs, and Others. The Generics segment is currently leading the market due to the increasing demand for cost-effective medication options and the expiration of patents for several blockbuster drugs. This trend is reinforced by the growing focus on biosimilars and advanced therapies, as well as the expansion of regulatory requirements for medical devices and combination products .
US Pharmaceutical Regulatory Affairs Market Competitive Landscape
The US Pharmaceutical Regulatory Affairs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Parexel International Corporation, Labcorp Drug Development (formerly Covance Inc.), Charles River Laboratories International, Inc., ICON plc, Medpace Holdings, Inc., PPD, Inc. (part of Thermo Fisher Scientific), Syneos Health, Inc., WuXi AppTec Co., Ltd., Freyr Solutions, PharmaLex GmbH, NDA Group AB, BlueReg Group, VCLS (Voisin Consulting Life Sciences), Accenture plc, Deloitte Touche Tohmatsu Limited contribute to innovation, geographic expansion, and service delivery in this space.
US Pharmaceutical Regulatory Affairs Market Industry Analysis
Growth Drivers
- Increasing R&D Investments:In future, the pharmaceutical industry is projected to invest approximately $200 billion in research and development (R&D) activities, reflecting a 5% increase from the previous year. This surge is driven by the need for innovative therapies and the development of complex biologics. The National Institutes of Health (NIH) reported that funding for biomedical research reached approximately $45 billion, emphasizing the critical role of R&D in advancing drug discovery and regulatory compliance.
- Rising Demand for Biologics:The biologics market is expected to reach approximately $400 billion in future, driven by an increasing prevalence of chronic diseases and a growing aging population. According to the FDA, biologics accounted for approximately 40% of new drug approvals in recent years, highlighting their significance in modern therapeutics. This demand necessitates robust regulatory frameworks to ensure safety and efficacy, thereby propelling the growth of regulatory affairs services in the pharmaceutical sector.
- Technological Advancements in Drug Development:The integration of advanced technologies, such as artificial intelligence (AI) and machine learning, is revolutionizing drug development processes. In future, it is estimated that AI-driven solutions will reduce drug development timelines by approximately 30%, significantly impacting regulatory strategies. The FDA has recognized over 100 AI-based tools, enhancing the efficiency of regulatory submissions and compliance, thus driving the demand for regulatory affairs expertise.
Market Challenges
- Stringent Regulatory Compliance:The regulatory landscape is becoming increasingly complex, with the FDA implementing more rigorous compliance standards. In future, the FDA is expected to conduct over 1,000 inspections, focusing on data integrity and clinical trial transparency. This heightened scrutiny poses challenges for pharmaceutical companies, as non-compliance can lead to significant financial penalties, estimated at approximately $1 billion annually across the industry, impacting overall market growth.
- High Costs of Drug Development:The average cost of developing a new drug has escalated to approximately $2.6 billion in future, driven by extensive clinical trials and regulatory hurdles. The Tufts Center for the Study of Drug Development reported that only approximately 12% of drugs entering clinical trials receive FDA approval, underscoring the financial risks involved. These high costs can deter investment in innovative therapies, posing a significant challenge to the regulatory affairs market.
US Pharmaceutical Regulatory Affairs Market Future Outlook
The US pharmaceutical regulatory affairs market is poised for significant evolution, driven by the increasing emphasis on patient-centric approaches and the integration of real-world evidence in regulatory processes. As companies adapt to these trends, the demand for regulatory expertise will grow, particularly in the context of mergers and acquisitions. Furthermore, the ongoing reforms in drug approval processes will likely enhance efficiency, fostering innovation and collaboration within the industry, ultimately benefiting patient access to new therapies.
Market Opportunities
- Growth in Personalized Medicine:The personalized medicine market is projected to reach approximately $100 billion in future, driven by advancements in genomics and biotechnology. This growth presents opportunities for regulatory affairs professionals to navigate the complexities of personalized therapies, ensuring compliance with evolving regulations and facilitating market access for innovative treatments.
- Expansion of Regulatory Consulting Services:As pharmaceutical companies face increasing regulatory challenges, the demand for regulatory consulting services is expected to rise. In future, the regulatory consulting market is anticipated to grow to approximately $10 billion, providing opportunities for firms to offer specialized expertise in navigating compliance, thereby enhancing their competitive advantage in the industry.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Regulatory Writing & Publishing Legal Representation Strategy & Consulting Compliance Management Submission Management Risk Management Others |
| By Application | Generics Biologics Medical Devices OTC Drugs Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Medical Device Manufacturers Contract Research Organizations (CROs) Others |
| By Service Provider | In-house Outsourcing |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Immunology Others |
| By Region | Northeast Midwest South West |
| By Company Size | Large Enterprises Medium Enterprises Small Enterprises Others |
| By Policy Support | Government Grants Tax Incentives Regulatory Exemptions Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Drug Enforcement Administration)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Research Organizations
Clinical Research Institutions
Pharmaceutical Trade Associations
Health Insurance Providers
Players Mentioned in the Report:
Parexel International Corporation
Labcorp Drug Development (formerly Covance Inc.)
Charles River Laboratories International, Inc.
ICON plc
Medpace Holdings, Inc.
PPD, Inc. (part of Thermo Fisher Scientific)
Syneos Health, Inc.
WuXi AppTec Co., Ltd.
Freyr Solutions
PharmaLex GmbH
NDA Group AB
BlueReg Group
VCLS (Voisin Consulting Life Sciences)
Accenture plc
Deloitte Touche Tohmatsu Limited
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Pharmaceutical Regulatory Affairs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Pharmaceutical Regulatory Affairs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Pharmaceutical Regulatory Affairs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing R&D Investments
3.1.2 Rising Demand for Biologics
3.1.3 Expanding Global Market Access
3.1.4 Technological Advancements in Drug Development
3.2 Market Challenges
3.2.1 Stringent Regulatory Compliance
3.2.2 High Costs of Drug Development
3.2.3 Market Competition and Saturation
3.2.4 Intellectual Property Issues
3.3 Market Opportunities
3.3.1 Growth in Personalized Medicine
3.3.2 Expansion of Regulatory Consulting Services
3.3.3 Increased Focus on Rare Diseases
3.3.4 Adoption of Digital Health Technologies
3.4 Market Trends
3.4.1 Shift Towards Patient-Centric Approaches
3.4.2 Integration of Artificial Intelligence in Regulatory Processes
3.4.3 Emphasis on Real-World Evidence
3.4.4 Growing Importance of Regulatory Affairs in M&A
3.5 Government Regulation
3.5.1 FDA Modernization Act
3.5.2 Drug Approval Process Reforms
3.5.3 Orphan Drug Act
3.5.4 21st Century Cures Act
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Pharmaceutical Regulatory Affairs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Pharmaceutical Regulatory Affairs Market Segmentation
8.1 By Type
8.1.1 Regulatory Writing & Publishing
8.1.2 Legal Representation
8.1.3 Strategy & Consulting
8.1.4 Compliance Management
8.1.5 Submission Management
8.1.6 Risk Management
8.1.7 Others
8.2 By Application
8.2.1 Generics
8.2.2 Biologics
8.2.3 Medical Devices
8.2.4 OTC Drugs
8.2.5 Others
8.3 By End-User
8.3.1 Pharmaceutical Companies
8.3.2 Biotechnology Firms
8.3.3 Medical Device Manufacturers
8.3.4 Contract Research Organizations (CROs)
8.3.5 Others
8.4 By Service Provider
8.4.1 In-house
8.4.2 Outsourcing
8.5 By Therapeutic Area
8.5.1 Oncology
8.5.2 Cardiovascular
8.5.3 Neurology
8.5.4 Infectious Diseases
8.5.5 Immunology
8.5.6 Others
8.6 By Region
8.6.1 Northeast
8.6.2 Midwest
8.6.3 South
8.6.4 West
8.7 By Company Size
8.7.1 Large Enterprises
8.7.2 Medium Enterprises
8.7.3 Small Enterprises
8.7.4 Others
8.8 By Policy Support
8.8.1 Government Grants
8.8.2 Tax Incentives
8.8.3 Regulatory Exemptions
8.8.4 Others
9. US Pharmaceutical Regulatory Affairs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue (USD, latest fiscal year)
9.2.4 Revenue Growth Rate (%)
9.2.5 Market Penetration (Number of US FDA submissions supported annually)
9.2.6 Client Portfolio Diversification (Pharma, Biotech, Device, etc.)
9.2.7 Regulatory Compliance Rate (Successful submissions/Total submissions)
9.2.8 Service Diversification Index (Breadth of regulatory services offered)
9.2.9 Client Satisfaction Score (Survey/Net Promoter Score)
9.2.10 Innovation Index (Investment in digital/AI regulatory solutions)
9.2.11 Employee Headcount (US-based regulatory professionals)
9.2.12 Years of US Regulatory Affairs Experience
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Parexel International Corporation
9.5.2 Labcorp Drug Development (formerly Covance Inc.)
9.5.3 Charles River Laboratories International, Inc.
9.5.4 ICON plc
9.5.5 Medpace Holdings, Inc.
9.5.6 PPD, Inc. (part of Thermo Fisher Scientific)
9.5.7 Syneos Health, Inc.
9.5.8 WuXi AppTec Co., Ltd.
9.5.9 Freyr Solutions
9.5.10 PharmaLex GmbH
9.5.11 NDA Group AB
9.5.12 BlueReg Group
9.5.13 VCLS (Voisin Consulting Life Sciences)
9.5.14 Accenture plc
9.5.15 Deloitte Touche Tohmatsu Limited
10. US Pharmaceutical Regulatory Affairs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Federal Agencies
10.1.2 State Health Departments
10.1.3 Public Health Organizations
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Regulatory Compliance
10.2.2 Spending on Technology Upgrades
10.2.3 Budget Allocation for Training
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotech Firms
10.3.3 Medical Device Manufacturers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Regulatory Changes
10.4.2 Training and Support Needs
10.4.3 Technology Adoption Rates
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Compliance Efficiency
10.5.2 Cost Savings Analysis
10.5.3 User Feedback and Improvement
10.5.4 Others
11. US Pharmaceutical Regulatory Affairs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of FDA regulatory guidelines and updates on pharmaceutical approvals
- Review of industry reports from organizations such as the Pharmaceutical Research and Manufacturers of America (PhRMA)
- Examination of market trends and forecasts from reputable pharmaceutical market research publications
Primary Research
- Interviews with regulatory affairs professionals in leading pharmaceutical companies
- Surveys targeting compliance officers and quality assurance managers
- Field interviews with industry experts and consultants specializing in pharmaceutical regulations
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including trade publications and regulatory filings
- Triangulation of qualitative insights from interviews with quantitative data from market reports
- Sanity checks conducted through expert panel reviews to ensure data accuracy and relevance
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on total pharmaceutical sales and regulatory compliance costs
- Segmentation of the market by therapeutic areas and regulatory pathways
- Incorporation of government initiatives and funding for pharmaceutical innovation
Bottom-up Modeling
- Collection of data on the number of new drug applications and their approval rates
- Estimation of costs associated with regulatory submissions and compliance for various drug categories
- Volume x cost analysis based on historical data of pharmaceutical product launches
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as R&D spending and regulatory changes
- Scenario modeling based on potential shifts in FDA policies and global market dynamics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Regulatory Compliance | 120 | Regulatory Affairs Managers, Compliance Officers |
| Clinical Trials Management | 90 | Clinical Research Coordinators, Project Managers |
| Drug Approval Processes | 60 | Regulatory Submissions Specialists, Quality Assurance Managers |
| Market Access Strategies | 50 | Market Access Managers, Health Economists |
| Pharmaceutical Policy Impact | 40 | Policy Analysts, Industry Consultants |
Frequently Asked Questions
What is the current value of the US Pharmaceutical Regulatory Affairs Market?
The US Pharmaceutical Regulatory Affairs Market is valued at approximately USD 2.7 billion, reflecting significant growth driven by increasing regulatory complexities, drug development activities, and a focus on compliance and safety within the pharmaceutical industry.