About the Report
Base Year 2024Oman Pharmaceutical Regulatory Affairs Market Overview
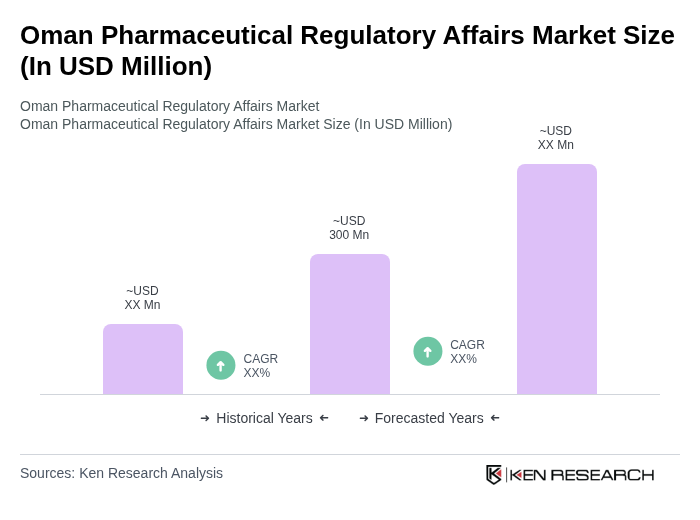
- The Oman Pharmaceutical Regulatory Affairs Market is valued at USD 300 million, based on a five-year historical analysis. This growth is primarily driven by increasing healthcare expenditure, a rising prevalence of chronic diseases, an aging population, and the government's focus on enhancing healthcare infrastructure. Additional drivers include economic diversification, increased demand for generics, advancements in biotechnology, and foreign investments. The regulatory framework has evolved to support the introduction of innovative pharmaceutical products, ensuring safety and efficacy in the market.
- Muscat, the capital city, is the dominant hub for pharmaceutical activities in Oman due to its strategic location, advanced healthcare facilities, and a concentration of regulatory bodies. Other notable cities include Salalah and Sohar, which are emerging as key players in the pharmaceutical landscape, driven by investments in healthcare and pharmaceutical manufacturing.
- TheMinisterial Decision No. 41/2023 issued by the Ministry of Healthestablished a regulation mandating that all pharmaceutical products must undergo a comprehensive review process before market entry. This regulation requires manufacturers and importers to submit detailed technical dossiers, clinical data, and quality certifications for evaluation by the Directorate General of Pharmaceutical Affairs and Drug Control. The regulation aims to enhance the safety and efficacy of drugs available to the public, ensuring that all products meet stringent quality standards and comply with international best practices.
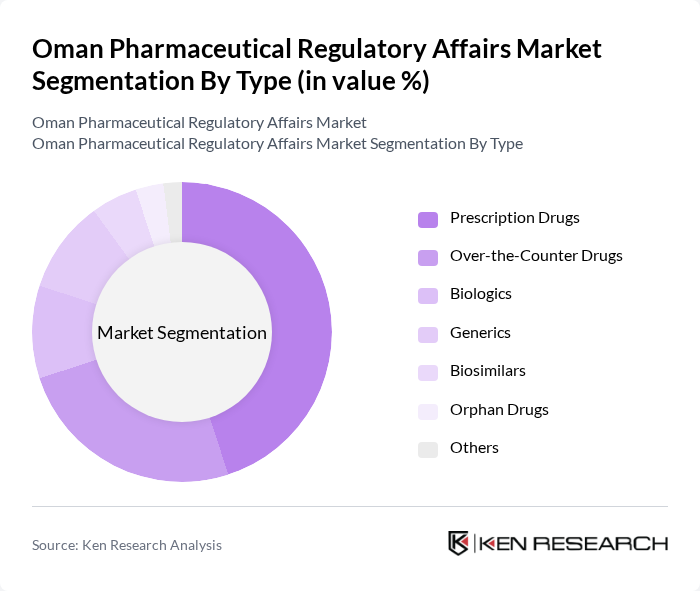
Oman Pharmaceutical Regulatory Affairs Market Segmentation
By Type:The market is segmented into various types, including Prescription Drugs, Over-the-Counter Drugs, Biologics, Generics, Biosimilars, Orphan Drugs, and Others.Prescription Drugsdominate the market due to the increasing prevalence of chronic diseases, the growing demand for effective treatment options, and a preference for branded medications. The trend towards personalized medicine, the introduction of innovative therapies, and the rising adoption of generics further bolster the demand for prescription medications.
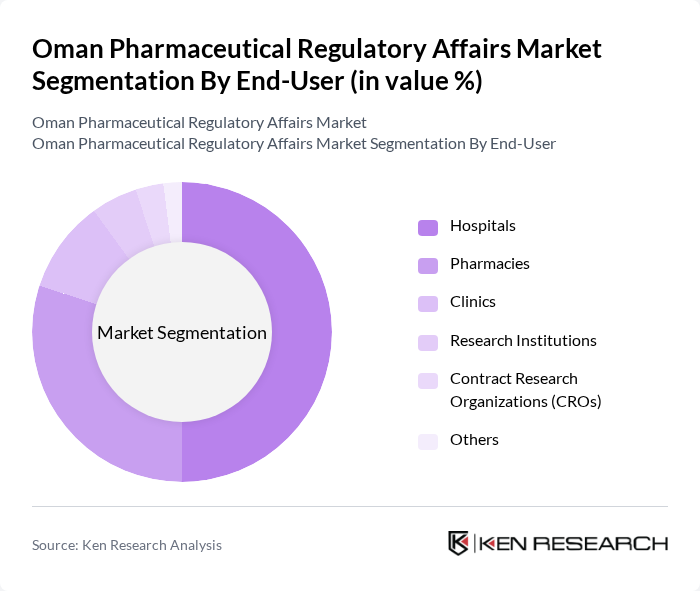
By End-User:The end-user segmentation includes Hospitals, Pharmacies, Clinics, Research Institutions, Contract Research Organizations (CROs), and Others.Hospitalsare the leading end-users, driven by the increasing number of patients requiring advanced medical care, the growing demand for specialized treatments, and substantial government investment in healthcare infrastructure. The expansion of healthcare facilities and the rising number of outpatient services also contribute to the dominance of hospitals in the pharmaceutical regulatory landscape.
Oman Pharmaceutical Regulatory Affairs Market Competitive Landscape
The Oman Pharmaceutical Regulatory Affairs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oman Pharmaceutical Products Co. LLC, Gulf Pharmaceutical Industries (Julphar), Dhofar Pharmaceutical Industries LLC, Muscat Pharmacy & Stores LLC, Al Hikma Pharmaceuticals PLC, United Pharmaceutical Manufacturing Co. LLC, Oman Medical Products Co. LLC, Al Nahda Pharmaceuticals LLC, Aster DM Healthcare, Badr Al Samaa Group of Hospitals, Al Muheet Pharmaceutical Co. LLC, Oman Drug Store LLC, Al Maharah Pharmaceuticals LLC, Al Salam Pharmaceutical Co. LLC, Novartis AG, Sanofi S.A., GlaxoSmithKline plc, Bayer AG, Cipla Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
Oman Pharmaceutical Regulatory Affairs Market Industry Analysis
Growth Drivers
- Increasing Demand for Pharmaceuticals:The pharmaceutical market in Oman is projected to reach OMR 1.2 billion in future, driven by a growing population of approximately 5.1 million. The rise in chronic diseases, such as diabetes and cardiovascular conditions, has led to an increased demand for medications. Additionally, the World Health Organization reported that Oman’s healthcare expenditure is expected to grow by approximately 7 percent annually, further fueling the need for pharmaceutical products and regulatory compliance.
- Government Initiatives for Healthcare Improvement:The Omani government has allocated OMR 1.5 billion for healthcare in future, focusing on enhancing healthcare services and infrastructure. Initiatives such as the National Health Strategy aim to improve access to medicines and healthcare services. This investment is expected to create a favorable environment for pharmaceutical companies, encouraging compliance with regulatory standards and fostering growth in the regulatory affairs sector.
- Rising Investment in Research and Development:Oman’s investment in pharmaceutical R&D is projected to reach OMR 200 million in future, reflecting a commitment to innovation. The government is promoting partnerships between local universities and pharmaceutical companies to enhance research capabilities. This focus on R&D is expected to lead to the development of new drugs and therapies, necessitating robust regulatory frameworks to ensure compliance and safety in the market.
Market Challenges
- Stringent Regulatory Compliance Requirements:The pharmaceutical sector in Oman faces stringent regulatory compliance requirements, which can be a significant barrier for companies. The Ministry of Health has implemented rigorous guidelines for drug approval, requiring extensive documentation and clinical trial data. This complexity can lead to delays in product launches, with an average approval time of 12-18 months, impacting market entry and competitiveness for new entrants.
- Limited Awareness of Regulatory Processes:Many local pharmaceutical companies lack comprehensive knowledge of regulatory processes, which can hinder their ability to navigate compliance effectively. A survey by the Omani Pharmaceutical Association indicated that over 60 percent of companies reported challenges in understanding regulatory requirements. This gap in knowledge can lead to non-compliance, resulting in penalties and potential market exclusion, further complicating the regulatory landscape.
Oman Pharmaceutical Regulatory Affairs Market Future Outlook
The future of the Oman pharmaceutical regulatory affairs market appears promising, driven by increasing healthcare investments and a focus on innovation. As the government continues to enhance healthcare infrastructure, the demand for regulatory compliance will rise. Additionally, the integration of digital technologies in regulatory processes is expected to streamline approvals and improve efficiency. This evolving landscape will likely attract both local and international players, fostering a competitive environment that prioritizes patient safety and product efficacy.
Market Opportunities
- Growth in Biopharmaceuticals:The biopharmaceutical sector in Oman is anticipated to grow significantly, with investments projected at OMR 50 million in future. This growth presents opportunities for regulatory affairs professionals to develop frameworks that ensure compliance with international standards, facilitating the introduction of innovative therapies and enhancing patient access to advanced treatments.
- Digital Transformation in Regulatory Affairs:The adoption of digital tools in regulatory affairs is expected to revolutionize compliance processes in Oman. With an estimated investment of OMR 30 million in digital solutions in future, companies can leverage technology to streamline submissions and enhance data management, ultimately improving regulatory efficiency and reducing time-to-market for new products.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Prescription Drugs Over-the-Counter Drugs Biologics Generics Biosimilars Orphan Drugs Others |
| By End-User | Hospitals Pharmacies Clinics Research Institutions Contract Research Organizations (CROs) Others |
| By Product Category | Cardiovascular Drugs Anti-Infectives Oncology Drugs Neurology Drugs Endocrinology (e.g., Diabetes Drugs) Respiratory Drugs Gastrointestinal Drugs Others |
| By Distribution Channel | Direct Sales Wholesalers Online Pharmacies Retail Pharmacies Hospital Pharmacies Others |
| By Regulatory Compliance Level | Fully Compliant Partially Compliant Non-Compliant Others |
| By Therapeutic Area | Diabetes Respiratory Gastrointestinal Oncology Cardiovascular Infectious Diseases Others |
| By Market Maturity | Emerging Market Established Market Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman Pharmaceutical Regulatory Authority)
Pharmaceutical Manufacturers and Producers
Distributors and Wholesalers
Clinical Research Organizations (CROs)
Pharmaceutical Trade Associations
Healthcare Providers and Institutions
Insurance Companies and Payers
Players Mentioned in the Report:
Oman Pharmaceutical Products Co. LLC
Gulf Pharmaceutical Industries (Julphar)
Dhofar Pharmaceutical Industries LLC
Muscat Pharmacy & Stores LLC
Al Hikma Pharmaceuticals PLC
United Pharmaceutical Manufacturing Co. LLC
Oman Medical Products Co. LLC
Al Nahda Pharmaceuticals LLC
Aster DM Healthcare
Badr Al Samaa Group of Hospitals
Al Muheet Pharmaceutical Co. LLC
Oman Drug Store LLC
Al-Fouzan Trading & Contracting Co. (remove, not relevant; replace with: Pfizer Inc.)
Al Maharah Pharmaceuticals LLC
Al Salam Pharmaceutical Co. LLC
Novartis AG
Sanofi S.A.
GlaxoSmithKline plc
Bayer AG
Cipla Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Pharmaceutical Regulatory Affairs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Pharmaceutical Regulatory Affairs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Pharmaceutical Regulatory Affairs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Pharmaceuticals
3.1.2 Government Initiatives for Healthcare Improvement
3.1.3 Rising Investment in Research and Development
3.1.4 Expansion of Healthcare Infrastructure
3.2 Market Challenges
3.2.1 Stringent Regulatory Compliance Requirements
3.2.2 Limited Awareness of Regulatory Processes
3.2.3 High Costs of Compliance
3.2.4 Market Entry Barriers for New Players
3.3 Market Opportunities
3.3.1 Growth in Biopharmaceuticals
3.3.2 Digital Transformation in Regulatory Affairs
3.3.3 Collaborations with International Regulatory Bodies
3.3.4 Expansion into Emerging Markets
3.4 Market Trends
3.4.1 Increasing Focus on Patient-Centric Regulations
3.4.2 Adoption of Advanced Technologies in Compliance
3.4.3 Shift Towards Personalized Medicine
3.4.4 Growing Importance of Sustainability in Pharmaceuticals
3.5 Government Regulation
3.5.1 New Drug Approval Processes
3.5.2 Pharmacovigilance Requirements
3.5.3 Pricing and Reimbursement Policies
3.5.4 Import and Export Regulations
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Pharmaceutical Regulatory Affairs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Pharmaceutical Regulatory Affairs Market Segmentation
8.1 By Type
8.1.1 Prescription Drugs
8.1.2 Over-the-Counter Drugs
8.1.3 Biologics
8.1.4 Generics
8.1.5 Biosimilars
8.1.6 Orphan Drugs
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Pharmacies
8.2.3 Clinics
8.2.4 Research Institutions
8.2.5 Contract Research Organizations (CROs)
8.2.6 Others
8.3 By Product Category
8.3.1 Cardiovascular Drugs
8.3.2 Anti-Infectives
8.3.3 Oncology Drugs
8.3.4 Neurology Drugs
8.3.5 Endocrinology (e.g., Diabetes Drugs)
8.3.6 Respiratory Drugs
8.3.7 Gastrointestinal Drugs
8.3.8 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Wholesalers
8.4.3 Online Pharmacies
8.4.4 Retail Pharmacies
8.4.5 Hospital Pharmacies
8.4.6 Others
8.5 By Regulatory Compliance Level
8.5.1 Fully Compliant
8.5.2 Partially Compliant
8.5.3 Non-Compliant
8.5.4 Others
8.6 By Therapeutic Area
8.6.1 Diabetes
8.6.2 Respiratory
8.6.3 Gastrointestinal
8.6.4 Oncology
8.6.5 Cardiovascular
8.6.6 Infectious Diseases
8.6.7 Others
8.7 By Market Maturity
8.7.1 Emerging Market
8.7.2 Established Market
8.7.3 Others
9. Oman Pharmaceutical Regulatory Affairs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Regulatory Submission Success Rate
9.2.6 Time to Regulatory Approval
9.2.7 Number of Regulatory Filings per Year
9.2.8 Compliance Audit Pass Rate
9.2.9 Product Portfolio Breadth (by type/therapeutic area)
9.2.10 Local vs. International Regulatory Expertise
9.2.11 Market Share Percentage
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Oman Pharmaceutical Products Co. LLC
9.5.2 Gulf Pharmaceutical Industries (Julphar)
9.5.3 Dhofar Pharmaceutical Industries LLC
9.5.4 Muscat Pharmacy & Stores LLC
9.5.5 Al Hikma Pharmaceuticals PLC
9.5.6 United Pharmaceutical Manufacturing Co. LLC
9.5.7 Oman Medical Products Co. LLC
9.5.8 Al Nahda Pharmaceuticals LLC
9.5.9 Aster DM Healthcare
9.5.10 Badr Al Samaa Group of Hospitals
9.5.11 Al Muheet Pharmaceutical Co. LLC
9.5.12 Oman Drug Store LLC
9.5.13 Al-Fouzan Trading & Contracting Co. (remove, not relevant; replace with: Pfizer Inc.)
9.5.14 Al Maharah Pharmaceuticals LLC
9.5.15 Al Salam Pharmaceutical Co. LLC
9.5.16 Novartis AG
9.5.17 Sanofi S.A.
9.5.18 GlaxoSmithKline plc
9.5.19 Bayer AG
9.5.20 Cipla Ltd.
10. Oman Pharmaceutical Regulatory Affairs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Commerce and Industry
10.1.3 Ministry of Finance
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Pharmaceutical Manufacturing Facilities
10.2.3 Research and Development Expenditure
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Pharmacies
10.3.3 Clinics
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Education Needs
10.4.2 Technology Adoption Rates
10.4.3 Regulatory Awareness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback Mechanisms
10.5.3 Scalability of Solutions
10.5.4 Others
11. Oman Pharmaceutical Regulatory Affairs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of regulatory frameworks from the Oman Ministry of Health and the Directorate General of Pharmaceutical Affairs
- Review of market reports and publications from the World Health Organization (WHO) and the Gulf Cooperation Council (GCC)
- Examination of pharmaceutical import/export data from Oman’s National Centre for Statistics and Information
Primary Research
- Interviews with regulatory affairs professionals in leading pharmaceutical companies operating in Oman
- Surveys targeting pharmacists and healthcare providers to understand market dynamics and regulatory challenges
- Field interviews with key stakeholders in the pharmaceutical supply chain, including distributors and wholesalers
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including industry reports and expert opinions
- Triangulation of regulatory changes with market trends and sales data
- Sanity checks conducted through expert panel reviews comprising regulatory consultants and industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total pharmaceutical market size based on national healthcare expenditure and population demographics
- Segmentation of the market by therapeutic categories and product types, including prescription and over-the-counter medications
- Incorporation of government healthcare initiatives and their impact on pharmaceutical demand
Bottom-up Modeling
- Collection of sales data from major pharmaceutical distributors and retailers in Oman
- Estimation of market share for local versus international pharmaceutical companies
- Volume and pricing analysis based on product categories and distribution channels
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating population growth, disease prevalence, and healthcare spending trends
- Scenario modeling based on potential regulatory changes and their impact on market access
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 50 | Regulatory Affairs Managers, Quality Assurance Officers |
| Healthcare Providers | 50 | Pharmacists, Hospital Administrators |
| Distributors and Wholesalers | 40 | Supply Chain Managers, Sales Directors |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Market Research Experts | 40 | Industry Analysts, Market Strategists |
Frequently Asked Questions
What is the current value of the Oman Pharmaceutical Regulatory Affairs Market?
The Oman Pharmaceutical Regulatory Affairs Market is valued at approximately USD 300 million, reflecting a five-year historical analysis. This growth is attributed to increased healthcare expenditure, a rising prevalence of chronic diseases, and government initiatives to enhance healthcare infrastructure.