About the Report
Base Year 2024APAC Biosimulation Market Overview
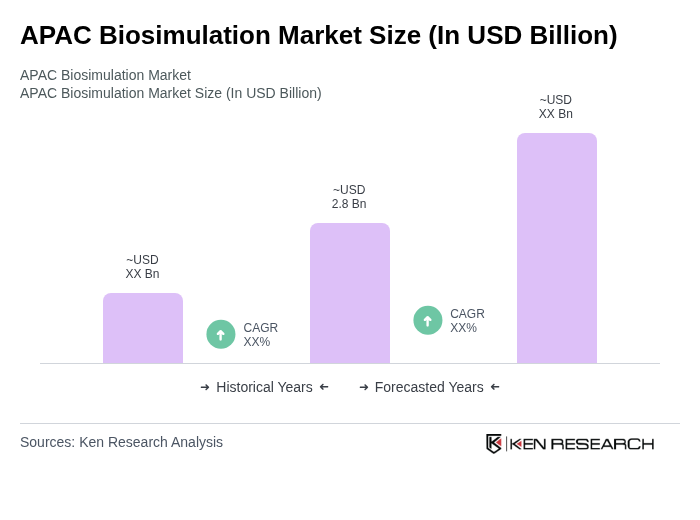
- The APAC Biosimulation Market is valued at USD 2.8 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in drug development processes, increasing demand for personalized medicine, and the rising adoption of in silico modeling techniques. The market is also supported by the growing need for cost-effective and time-efficient solutions in clinical trials and regulatory submissions. Recent trends include the integration of artificial intelligence, cloud-based simulation platforms, and the expansion of biosimulation applications into clinical decision support and real-world data analytics, further accelerating adoption across the region .
- Key players in this market includeChina, Japan, and India, which dominate due to their robust pharmaceutical and biotechnology sectors. China leads with significant investments in research and development, particularly in precision and personalized medicine. Japan is recognized for its regulatory harmonization and technological innovation in drug discovery, while India benefits from a large pool of skilled professionals and a rapidly growing number of biotech firms, making it a competitive player in the biosimulation landscape .
- In 2023, the Indian government implemented theNational Biotechnology Development Strategy (NBDS), 2021–2025issued by the Department of Biotechnology, Ministry of Science & Technology. This strategy aims to enhance the biosimulation capabilities of local firms, including a funding allocation of INR 200 billion to support research, development, and the adoption of advanced biosimulation technologies. The NBDS outlines operational priorities such as strengthening translational research infrastructure, promoting industry-academia collaboration, and incentivizing the adoption of digital and computational tools in drug development .
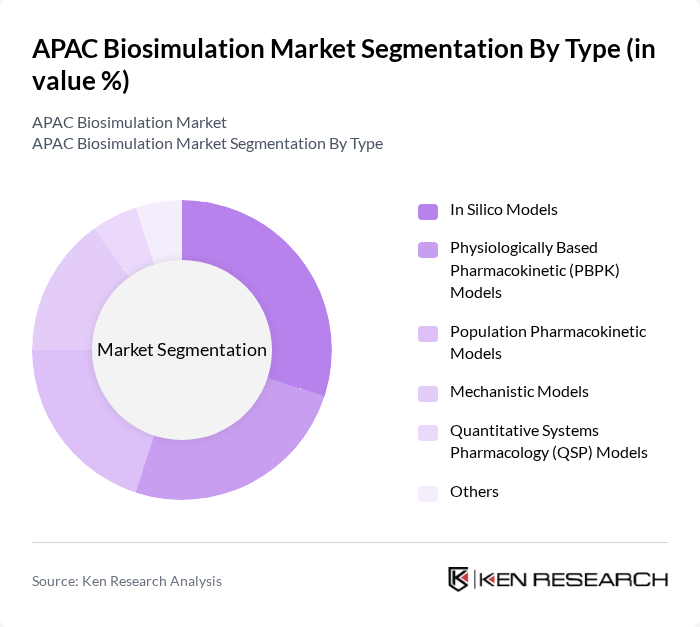
APAC Biosimulation Market Segmentation
By Type:The market is segmented into various types of biosimulation models, including In Silico Models, Physiologically Based Pharmacokinetic (PBPK) Models, Population Pharmacokinetic Models, Mechanistic Models, Quantitative Systems Pharmacology (QSP) Models, and Others. Each of these models serves distinct purposes in drug development and research, with varying levels of complexity and application. Integrated software platforms, especially those supporting PBPK and QSP modeling, are seeing the highest adoption due to their ability to streamline regulatory submissions and optimize clinical trial design .
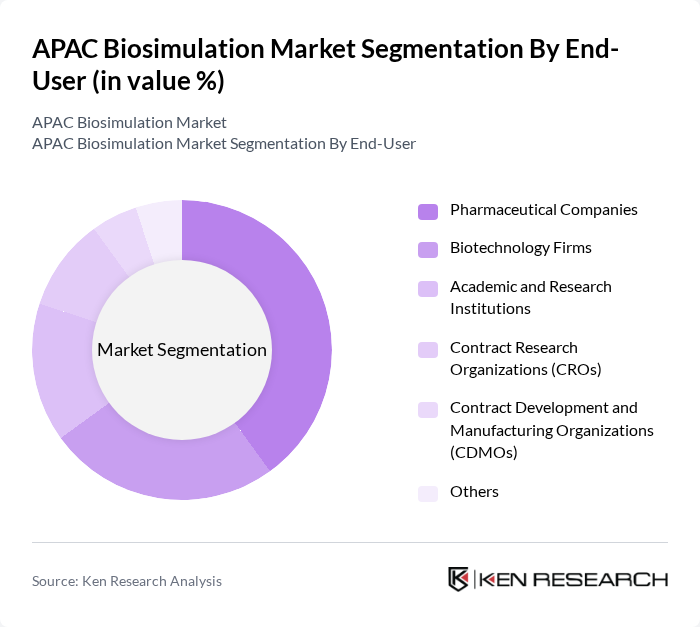
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Academic and Research Institutions, Contract Research Organizations (CROs), Contract Development and Manufacturing Organizations (CDMOs), and Others. Each segment plays a crucial role in utilizing biosimulation technologies for various applications in drug development and research. Pharmaceutical and biotechnology companies are the primary adopters, leveraging biosimulation to accelerate drug discovery, reduce development costs, and improve clinical outcomes .
APAC Biosimulation Market Competitive Landscape
The APAC Biosimulation Market is characterized by a dynamic mix of regional and international players. Leading participants such as Certara, Simulations Plus, Dassault Systèmes (BIOVIA), Schrödinger, InSilico Medicine, BioSymetrics, ChemAxon, Genedata, Optibrium, Medidata Solutions (Dassault Systèmes), ACD/Labs (Advanced Chemistry Development), Biorelate, GSK (GlaxoSmithKline), Takeda Pharmaceutical Company, WuXi AppTec contribute to innovation, geographic expansion, and service delivery in this space.
APAC Biosimulation Market Industry Analysis
Growth Drivers
- Increasing Demand for Drug Development Efficiency:The APAC region is witnessing a surge in drug development efficiency, driven by the need to reduce time-to-market. In future, the average time for drug development is projected to be around 10 to 15 years, with biosimulation tools potentially reducing this by up to 30%. This efficiency is crucial as the global pharmaceutical market is expected to reach $1.6 trillion, emphasizing the need for faster, cost-effective solutions in drug development.
- Rising Adoption of Personalized Medicine:The personalized medicine market in APAC is anticipated to grow significantly, with an estimated value of $120 billion in future. This growth is fueled by advancements in genomics and biotechnology, which necessitate the use of biosimulation to tailor treatments to individual patient profiles. As healthcare systems increasingly focus on personalized approaches, the demand for biosimulation tools that support these initiatives is expected to rise correspondingly.
- Advancements in Computational Technologies:The rapid evolution of computational technologies is a key driver for the biosimulation market in APAC. In future, investments in AI and machine learning within the healthcare sector are projected to exceed $35 billion. These technologies enhance the accuracy and efficiency of biosimulation models, enabling researchers to simulate complex biological processes and predict drug interactions more effectively, thus fostering innovation in drug development.
Market Challenges
- High Initial Investment Costs:One of the significant barriers to the adoption of biosimulation in APAC is the high initial investment required. The average cost of implementing biosimulation software and infrastructure can range from $600,000 to $2.5 million, depending on the complexity of the systems. This financial burden can deter smaller pharmaceutical companies and research institutions from adopting these advanced technologies, limiting market growth.
- Limited Awareness Among Stakeholders:Despite the potential benefits, there remains a significant knowledge gap regarding biosimulation among stakeholders in the APAC region. A recent survey indicated that over 65% of pharmaceutical companies are unaware of the capabilities and advantages of biosimulation. This lack of awareness hinders the widespread adoption of these technologies, slowing down innovation and efficiency improvements in drug development processes.
APAC Biosimulation Market Future Outlook
The future of the APAC biosimulation market appears promising, driven by technological advancements and increasing regulatory support. As the region embraces cloud-based solutions and integrates AI into biosimulation processes, the efficiency and accuracy of drug development are expected to improve significantly. Furthermore, the rise of virtual clinical trials will likely reshape traditional methodologies, allowing for more flexible and cost-effective approaches to drug testing and approval, ultimately enhancing patient outcomes and market competitiveness.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets in APAC, such as India and Vietnam, present significant opportunities for biosimulation companies. With increasing investments in healthcare infrastructure, these regions are poised to adopt advanced technologies, including biosimulation, to enhance drug development processes and improve healthcare outcomes.
- Collaborations with Academic Institutions:Collaborating with academic institutions can drive innovation in biosimulation tools. By leveraging research capabilities and expertise, companies can develop cutting-edge solutions that address specific industry needs, fostering a more robust biosimulation ecosystem in the APAC region and enhancing overall market growth.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | In Silico Models Physiologically Based Pharmacokinetic (PBPK) Models Population Pharmacokinetic Models Mechanistic Models Quantitative Systems Pharmacology (QSP) Models Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) Contract Development and Manufacturing Organizations (CDMOs) Others |
| By Region | China Japan India South Korea Australia & New Zealand ASEAN Rest of Asia Pacific |
| By Application | Drug Discovery Toxicology Studies Clinical Trials Simulation Regulatory Submissions Precision Medicine Others |
| By Technology | Software Solutions Cloud-Based Platforms Simulation Tools Data Analytics Solutions Artificial Intelligence/Machine Learning Integration Others |
| By Investment Source | Private Investments Government Funding Venture Capital Public-Private Partnerships (PPP) Others |
| By Policy Support | Research Grants Tax Incentives Regulatory Frameworks Subsidies for Technology Adoption Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Institutes of Health, Food and Drug Administration)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations
Healthcare Providers and Hospitals
Medical Device Manufacturers
Biotechnology Research Firms
Insurance Companies and Payers
Players Mentioned in the Report:
Certara
Simulations Plus
Dassault Systemes (BIOVIA)
Schrodinger
InSilico Medicine
BioSymetrics
ChemAxon
Genedata
Optibrium
Medidata Solutions (Dassault Systemes)
ACD/Labs (Advanced Chemistry Development)
Biorelate
GSK (GlaxoSmithKline)
Takeda Pharmaceutical Company
WuXi AppTec
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Biosimulation Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Biosimulation Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Biosimulation Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for drug development efficiency
3.1.2 Rising adoption of personalized medicine
3.1.3 Advancements in computational technologies
3.1.4 Growing regulatory support for biosimulation
3.2 Market Challenges
3.2.1 High initial investment costs
3.2.2 Limited awareness among stakeholders
3.2.3 Data privacy and security concerns
3.2.4 Integration with existing systems
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with academic institutions
3.3.3 Development of innovative biosimulation tools
3.3.4 Increasing focus on sustainable practices
3.4 Market Trends
3.4.1 Shift towards cloud-based biosimulation solutions
3.4.2 Integration of AI and machine learning
3.4.3 Growing emphasis on real-world evidence
3.4.4 Rise of virtual clinical trials
3.5 Government Regulation
3.5.1 Guidelines for biosimulation in drug approval
3.5.2 Support for research and development initiatives
3.5.3 Regulations on data usage and sharing
3.5.4 Incentives for technology adoption
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Biosimulation Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Biosimulation Market Segmentation
8.1 By Type
8.1.1 In Silico Models
8.1.2 Physiologically Based Pharmacokinetic (PBPK) Models
8.1.3 Population Pharmacokinetic Models
8.1.4 Mechanistic Models
8.1.5 Quantitative Systems Pharmacology (QSP) Models
8.1.6 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Academic and Research Institutions
8.2.4 Contract Research Organizations (CROs)
8.2.5 Contract Development and Manufacturing Organizations (CDMOs)
8.2.6 Others
8.3 By Region
8.3.1 China
8.3.2 Japan
8.3.3 India
8.3.4 South Korea
8.3.5 Australia & New Zealand
8.3.6 ASEAN
8.3.7 Rest of Asia Pacific
8.4 By Application
8.4.1 Drug Discovery
8.4.2 Toxicology Studies
8.4.3 Clinical Trials Simulation
8.4.4 Regulatory Submissions
8.4.5 Precision Medicine
8.4.6 Others
8.5 By Technology
8.5.1 Software Solutions
8.5.2 Cloud-Based Platforms
8.5.3 Simulation Tools
8.5.4 Data Analytics Solutions
8.5.5 Artificial Intelligence/Machine Learning Integration
8.5.6 Others
8.6 By Investment Source
8.6.1 Private Investments
8.6.2 Government Funding
8.6.3 Venture Capital
8.6.4 Public-Private Partnerships (PPP)
8.6.5 Others
8.7 By Policy Support
8.7.1 Research Grants
8.7.2 Tax Incentives
8.7.3 Regulatory Frameworks
8.7.4 Subsidies for Technology Adoption
8.7.5 Others
9. APAC Biosimulation Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (APAC region)
9.2.4 Number of APAC Clients/Projects
9.2.5 R&D Expenditure as % of Revenue
9.2.6 Market Penetration Rate (APAC)
9.2.7 Product Portfolio Breadth (e.g., PBPK, QSP, AI integration)
9.2.8 Regulatory Approvals/Collaborations in APAC
9.2.9 Customer Retention Rate
9.2.10 Average Deal Size (APAC)
9.2.11 Return on Investment (ROI)
9.2.12 Market Share Percentage (APAC)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Certara
9.5.2 Simulations Plus
9.5.3 Dassault Systèmes (BIOVIA)
9.5.4 Schrödinger
9.5.5 InSilico Medicine
9.5.6 BioSymetrics
9.5.7 ChemAxon
9.5.8 Genedata
9.5.9 Optibrium
9.5.10 Medidata Solutions (Dassault Systèmes)
9.5.11 ACD/Labs (Advanced Chemistry Development)
9.5.12 Biorelate
9.5.13 GSK (GlaxoSmithKline)
9.5.14 Takeda Pharmaceutical Company
9.5.15 WuXi AppTec
10. APAC Biosimulation Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Procurement Channels
10.1.4 Evaluation Criteria for Vendors
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns by Sector
10.2.3 Impact of Economic Conditions
10.2.4 Future Spending Projections
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Implementation
10.3.2 Technical Support Needs
10.3.3 Cost Management Issues
10.3.4 Regulatory Compliance Concerns
10.4 User Readiness for Adoption
10.4.1 Training and Support Requirements
10.4.2 Technology Familiarity Levels
10.4.3 Infrastructure Readiness
10.4.4 Attitudes Towards Innovation
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Opportunities for Upscaling
10.5.4 Long-term Value Realization
11. APAC Biosimulation Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Customer Segmentation
1.5 Key Partnerships
1.6 Cost Structure Analysis
1.7 Competitive Advantage
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from biotechnology and pharmaceutical associations in the APAC region
- Review of academic journals and publications focusing on biosimulation technologies and applications
- Examination of market trends and forecasts from government and regulatory bodies related to drug development
Primary Research
- Interviews with R&D leaders at pharmaceutical companies utilizing biosimulation
- Surveys with clinical trial managers to understand the adoption of biosimulation tools
- Field interviews with software developers specializing in biosimulation platforms
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of quantitative data from surveys with qualitative insights from expert interviews
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall pharmaceutical market size in APAC as a baseline for biosimulation market potential
- Segmentation of the market by therapeutic areas and types of biosimulation applications
- Incorporation of growth rates from related sectors such as personalized medicine and digital health
Bottom-up Modeling
- Collection of data on the number of clinical trials utilizing biosimulation across major APAC markets
- Estimation of revenue generated from biosimulation software and services based on firm-level data
- Volume x pricing analysis for biosimulation tools and consulting services
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as regulatory changes and technological advancements
- Scenario modeling based on varying levels of market adoption and investment in biosimulation
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical R&D Departments | 100 | R&D Directors, Clinical Trial Managers |
| Biosimulation Software Providers | 60 | Product Managers, Software Developers |
| Regulatory Affairs Specialists | 50 | Regulatory Affairs Managers, Compliance Officers |
| Healthcare Technology Consultants | 40 | Consultants, Market Analysts |
| Academic Researchers in Biosimulation | 40 | Professors, Research Scientists |
Frequently Asked Questions
What is the current value of the APAC Biosimulation Market?
The APAC Biosimulation Market is valued at approximately USD 2.8 billion, driven by advancements in drug development, personalized medicine demand, and the adoption of in silico modeling techniques.