Region:Asia
Author(s):Dev
Product Code:KRAC2799
Pages:91
Published On:October 2025
By Biomarker Type:The market is segmented into various biomarker types, including Alpha-Synuclein (?-Syn), Neurofilament Light Chain (NfL), Phosphorylated Tau (p-Tau), and Other Neurodegeneration Markers. Each of these biomarkers plays a crucial role in the diagnosis and monitoring of Parkinson's disease, with specific applications in clinical settings.
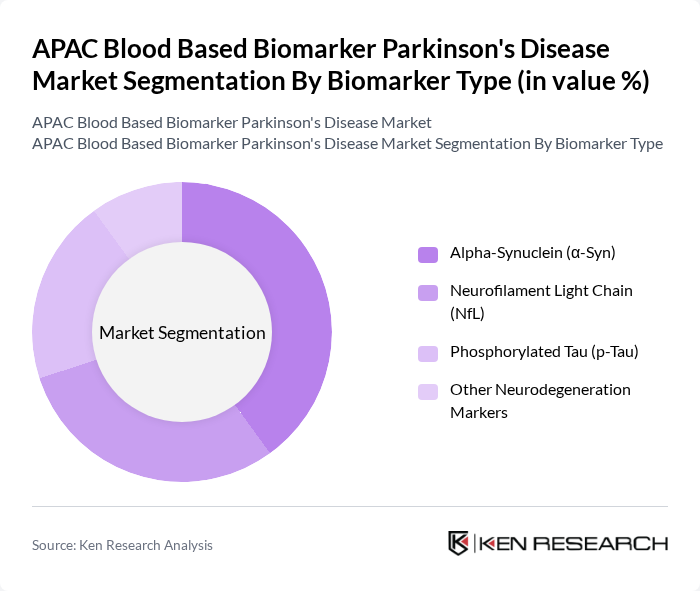
The Alpha-Synuclein (?-Syn) biomarker is currently dominating the market due to its critical role in the pathophysiology of Parkinson's disease. Its presence in blood samples has been linked to disease onset and progression, making it a focal point for both research and clinical diagnostics. The increasing awareness among healthcare professionals regarding the importance of early diagnosis and the ability to monitor disease progression effectively has led to a surge in demand for tests utilizing this biomarker.
By Technology Platform:The market is segmented by technology platforms, including Immunoassay-Based, Mass Spectrometry-Based, Genomic & Proteomic Analysis, and Multiplex Assay Technologies. Each platform offers unique advantages in terms of sensitivity, specificity, and throughput for biomarker detection.
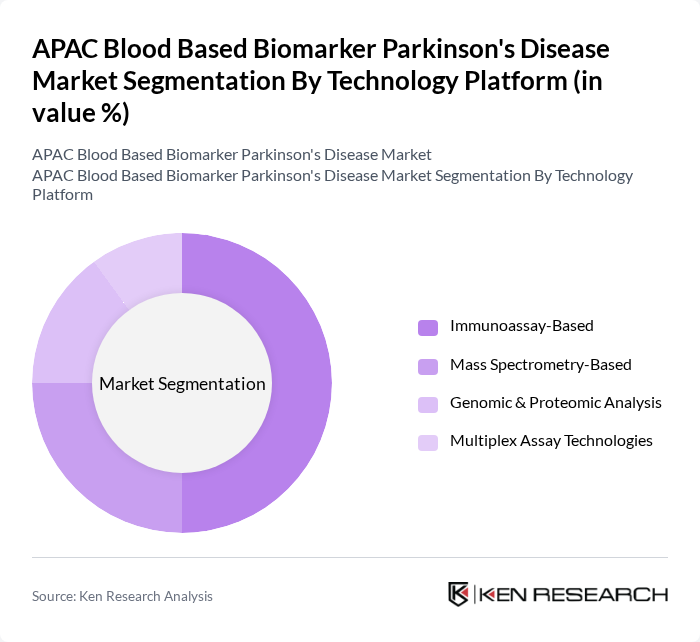
The Immunoassay-Based technology platform is leading the market due to its established use in clinical laboratories and its ability to provide rapid and reliable results. This platform is favored for its ease of use and cost-effectiveness, making it accessible for widespread clinical application. The growing trend towards personalized medicine and the need for precise diagnostic tools further bolster the demand for immunoassay-based tests in the context of Parkinson's disease.
The APAC Blood Based Biomarker Parkinson's Disease Market is characterized by a dynamic mix of regional and international players. Leading participants such as F. Hoffmann-La Roche Ltd (Roche Diagnostics), Sysmex Corporation, Proteomedix AG, MiRXES Pte Ltd, Eisai Co. Ltd, Thermo Fisher Scientific Inc., Siemens Healthineers AG, Abbott Laboratories, Bio-Rad Laboratories Inc., Nutech Cancer Biomarkers India Pvt Ltd, Creative Diagnostics, Minomic International Limited, QIAGEN N.V., Illumina Inc., PerkinElmer Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC blood-based biomarker market for Parkinson's Disease appears promising, driven by ongoing advancements in technology and increasing healthcare investments. As the region's population ages, the demand for innovative diagnostic solutions will likely rise. Furthermore, the integration of artificial intelligence in biomarker analysis is expected to enhance diagnostic accuracy and efficiency. Collaborative efforts between biotech firms and research institutions will also play a crucial role in accelerating the development of personalized medicine approaches tailored to individual patient needs.
| Segment | Sub-Segments |
|---|---|
| By Biomarker Type | Alpha-Synuclein (?-Syn) Neurofilament Light Chain (NfL) Phosphorylated Tau (p-Tau) Other Neurodegeneration Markers |
| By Technology Platform | Immunoassay-Based Mass Spectrometry-Based Genomic & Proteomic Analysis Multiplex Assay Technologies |
| By Application | Early Diagnosis Disease Progression Monitoring Treatment Response Evaluation Differential Diagnosis from Other Neurodegenerative Diseases |
| By End-User | Hospitals and Medical Centers Diagnostic Laboratories Research Institutions and Academic Centers Clinical Trial Organizations |
| By Region | North Asia (Japan, South Korea, China) Southeast Asia (Thailand, Vietnam, Indonesia, Philippines) South Asia (India, Bangladesh) Oceania |
| By Distribution Channel | Direct Sales to Healthcare Facilities Diagnostic Laboratory Partnerships Online Clinical Diagnostic Platforms Contract Research Organization (CRO) Networks |
| By Regulatory Approval Status | FDA/EMA Approved Regional Regulatory Approved (China NMPA, India CDSCO) Under Clinical Validation Research Use Only (RUO) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurologists and Movement Disorder Specialists | 120 | Neurologists, Clinical Researchers |
| Laboratory Managers and Technicians | 90 | Laboratory Directors, Biomedical Technologists |
| Healthcare Policy Makers | 60 | Health Administrators, Policy Analysts |
| Patients and Caregivers | 80 | Parkinson's Patients, Caregivers |
| Biomarker Research Scientists | 70 | Research Scientists, Clinical Trial Coordinators |
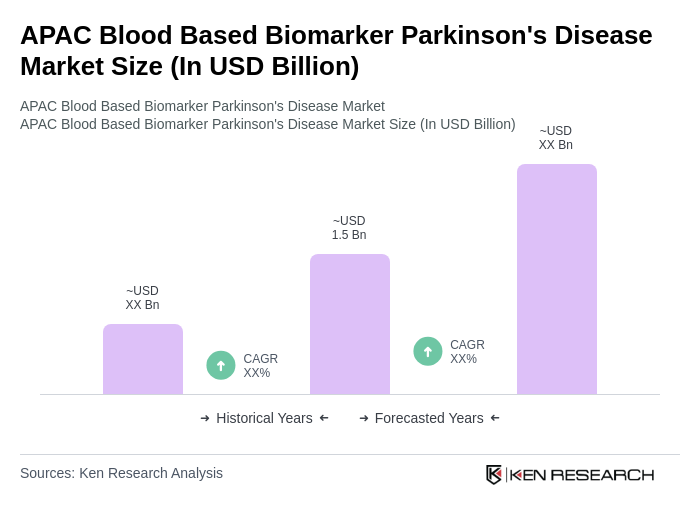
The APAC Blood Based Biomarker Parkinson's Disease Market is valued at approximately USD 1.5 billion, driven by the increasing prevalence of Parkinson's disease and advancements in biomarker research, which enhance diagnostic accuracy and disease monitoring.