About the Report
Base Year 2024APAC Influenza Diagnostics Market Overview
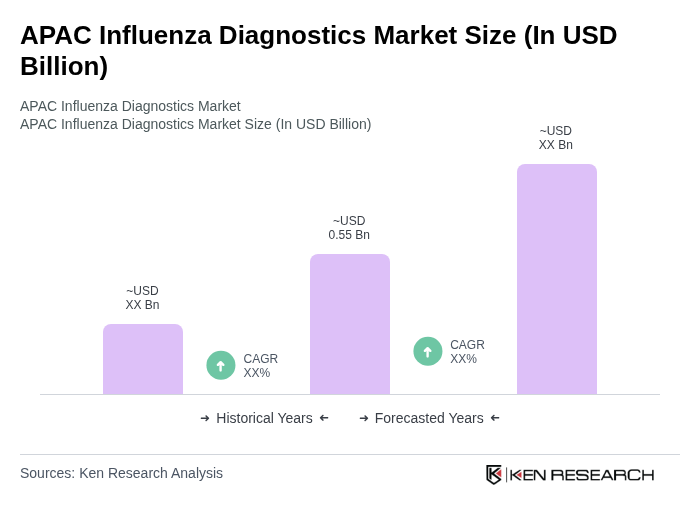
- The APAC Influenza Diagnostics Market is valued at USD 0.55 billion, based on a five?year historical analysis. This growth is primarily driven by the increasing prevalence of seasonal and zoonotic influenza outbreaks, heightened awareness regarding early diagnosis, and advancements in molecular and point?of?care diagnostic technologies. The market has seen a surge in demand for rapid and accurate testing methods, including rapid antigen assays and molecular point?of?care platforms, which are crucial for effective disease management and control in both hospital and decentralized settings.
- Key players in this market include China, Japan, and India, which dominate due to their large populations, expanding healthcare infrastructure, and significant investments in diagnostic and life?science technologies. These countries have also experienced recurrent influenza activity and heightened preparedness efforts, prompting governments and healthcare providers to prioritize surveillance networks, laboratory capacity, and rapid diagnostic solutions to enhance public health responses.
- In India, influenza diagnosis in hospitals and clinics during peak flu seasons is guided by national standards under the Laboratory Biosafety Guidelines, 2023, issued by the Ministry of Health and Family Welfare and the Indian Council of Medical Research (ICMR), which mandate appropriate biosafety practices, specimen handling, and use of validated in?vitro diagnostic tests for respiratory pathogens including influenza in clinical and public health laboratories. These requirements aim to support timely and reliable influenza diagnosis, safeguard laboratory and clinical staff, and strengthen health?system capacity to manage seasonal and pandemic influenza burdens.
APAC Influenza Diagnostics Market Segmentation
By Test Type:The market is segmented into various test types, including Rapid Influenza Diagnostic Tests (RIDTs), Reverse Transcription Polymerase Chain Reaction (RT?PCR) Tests, Molecular Point?of?Care Tests, Viral Culture Tests, and Immunofluorescence and Other Traditional Tests. Each of these test types plays a crucial role in diagnosing influenza, with varying levels of accuracy, speed, and complexity.
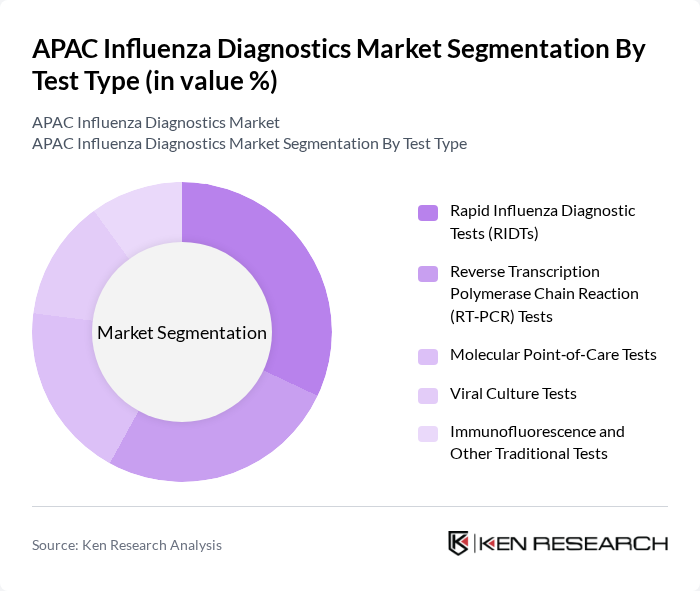
The Rapid Influenza Diagnostic Tests (RIDTs) segment is currently dominating the market due to their ease of use, quick turnaround time, and ability to provide results typically within 15–30 minutes. This rapid testing capability is particularly appealing in clinical and emergency settings where timely diagnosis is critical for effective patient management and initiation of antiviral therapy. Additionally, the increasing adoption of RIDTs and rapid antigen assays in decentralized and point?of?care environments, including outpatient clinics and urgent care centers, has further propelled their market share, as healthcare providers seek efficient solutions to manage seasonal influenza surges and emerging respiratory threats.
By End-User:The market is segmented by end-users, including Hospitals, Diagnostic and Reference Laboratories, Point?of?Care Testing (POCT) Settings and Clinics, Home Care and Over?the?Counter Testing, and Research and Academic Institutes. Each end-user category has distinct needs and preferences regarding influenza diagnostics, ranging from high?throughput molecular platforms in centralized labs to portable, user?friendly assays for near?patient and home use.
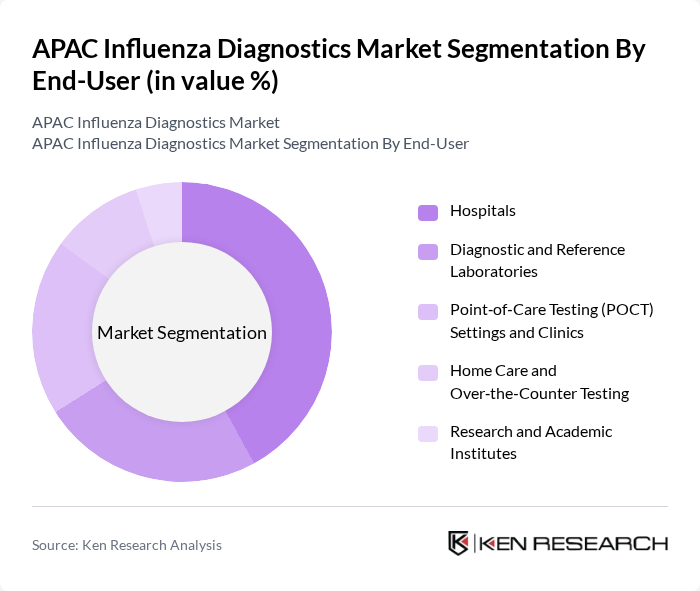
Hospitals are the leading end-user segment in the influenza diagnostics market, primarily due to their high patient volumes and the critical need for accurate and timely diagnosis in emergency and inpatient settings. Hospitals often have the resources to invest in advanced diagnostic technologies, such as high?sensitivity RT?PCR platforms and integrated respiratory panels, which enhances their ability to manage severe influenza cases and co?infections effectively. Furthermore, the integration of laboratory?based and point?of?care testing into hospital workflows is essential for improving patient outcomes, optimizing bed management, and controlling the spread of influenza within healthcare facilities and the wider community.
APAC Influenza Diagnostics Market Competitive Landscape
The APAC Influenza Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as F. Hoffmann?La Roche Ltd, Abbott Laboratories, Siemens Healthineers AG, Thermo Fisher Scientific Inc., bioMérieux SA, Cepheid (Danaher Corporation), QuidelOrtho Corporation, Hologic, Inc., Becton, Dickinson and Company (BD), GenMark Diagnostics (Roche), Luminex Corporation (DiaSorin S.p.A.), PerkinElmer, Inc. (Revvity, Inc.), Agilent Technologies, Inc., Eiken Chemical Co., Ltd., Meridian Bioscience, Inc. (A Diebold Nixdorf Company), Beijing Wantai Biological Pharmacy Co., Ltd., Guangzhou Wondfo Biotech Co., Ltd., Seegene Inc., SD Biosensor, Inc., Sysmex Corporation contribute to innovation, geographic expansion, and service delivery in this space.
APAC Influenza Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Influenza Outbreaks:The APAC region has witnessed a significant rise in influenza cases, with the World Health Organization reporting over 1.5 million confirmed cases in recent seasons. This surge has heightened the demand for effective diagnostic solutions. Countries like India and China have experienced seasonal outbreaks, prompting healthcare systems to prioritize influenza diagnostics. The increasing incidence of influenza not only drives demand for testing but also emphasizes the need for timely interventions to mitigate public health risks.
- Advancements in Diagnostic Technologies:The APAC influenza diagnostics market is benefiting from rapid technological advancements, particularly in molecular diagnostics. For instance, the introduction of PCR-based tests has reduced the time to diagnosis to under two hours, enhancing patient management. According to a report by the Asia-Pacific Medical Technology Association, the market for molecular diagnostics is projected to reach $5.5 billion in future, driven by innovations that improve accuracy and speed, thus fostering greater adoption in clinical settings.
- Rising Healthcare Expenditure in APAC:Healthcare spending in the APAC region is projected to reach $3 trillion in future, according to the World Bank. This increase is largely driven by government initiatives aimed at improving healthcare infrastructure and access to medical technologies. Countries like Japan and South Korea are leading in healthcare investments, which directly supports the growth of the influenza diagnostics market. Enhanced funding allows for the procurement of advanced diagnostic tools, ultimately improving disease management and patient outcomes.
Market Challenges
- High Cost of Advanced Diagnostic Equipment:The cost of advanced diagnostic equipment remains a significant barrier in the APAC influenza diagnostics market. For instance, high-throughput PCR machines can exceed $120,000, limiting access for smaller healthcare facilities. This financial constraint is particularly pronounced in developing nations, where healthcare budgets are constrained. As a result, many facilities may opt for less accurate, traditional testing methods, which can hinder effective disease management and control efforts.
- Limited Access to Healthcare in Rural Areas:Access to healthcare services in rural areas of APAC is a persistent challenge, with approximately 65% of the population in these regions lacking adequate healthcare facilities. According to the Asian Development Bank, rural healthcare infrastructure is often underfunded, leading to delays in diagnosis and treatment. This disparity in access can result in higher morbidity rates during influenza outbreaks, as timely diagnostics are crucial for effective intervention and management of the disease.
APAC Influenza Diagnostics Market Future Outlook
The APAC influenza diagnostics market is poised for significant transformation, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the expansion of telemedicine services will facilitate remote testing and consultations. As governments prioritize public health initiatives, the focus on rapid diagnostic tests will likely grow, ensuring timely interventions during influenza outbreaks and improving overall healthcare outcomes in the region.
Market Opportunities
- Expansion of Telemedicine Services:The rise of telemedicine presents a unique opportunity for influenza diagnostics. With an estimated 35% increase in telehealth consultations in future, healthcare providers can leverage remote diagnostics to reach underserved populations. This approach not only enhances access to testing but also streamlines patient management, ultimately improving health outcomes during influenza seasons.
- Development of Rapid Diagnostic Tests:The demand for rapid diagnostic tests is on the rise, with the market for these tests projected to grow significantly. Innovations in test design, such as lateral flow assays, are expected to reduce diagnosis time to under 25 minutes. This rapid turnaround is crucial for effective outbreak management, allowing healthcare providers to initiate timely treatment and containment measures, thereby enhancing public health responses.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Test Type | Rapid Influenza Diagnostic Tests (RIDTs) Reverse Transcription Polymerase Chain Reaction (RT?PCR) Tests Molecular Point?of?Care Tests Viral Culture Tests Immunofluorescence and Other Traditional Tests |
| By End-User | Hospitals Diagnostic and Reference Laboratories Point?of?Care Testing (POCT) Settings and Clinics Home Care and Over?the?Counter Testing Research and Academic Institutes |
| By Country | China Japan India South Korea Australia & New Zealand ASEAN (Indonesia, Thailand, Malaysia, Singapore, Vietnam, Others) Rest of APAC |
| By Technology | Molecular Diagnostics Immunoassay?based Rapid Tests Next?Generation Sequencing (NGS) Other Emerging and Hybrid Technologies |
| By Application | Clinical Diagnostics Public Health Surveillance Research and Drug/Vaccine Development Occupational and Travel Health Screening |
| By Setting of Care | Centralized Laboratory Testing Decentralized and Near?Patient Testing Retail and Pharmacy?based Testing Mobile and Community Health Programs |
| By Payer Type | Public and Social Health Insurance Private Insurance Out?of?Pocket Payments Donor?funded and NGO?supported Programs |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., World Health Organization, Ministry of Health in various APAC countries)
Manufacturers and Producers of Diagnostic Kits
Distributors and Retailers of Medical Devices
Public Health Organizations
Healthcare Providers and Hospitals
Industry Associations (e.g., Asia Pacific Society of Clinical Microbiology and Infection)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
F. HoffmannLa Roche Ltd
Abbott Laboratories
Siemens Healthineers AG
Thermo Fisher Scientific Inc.
bioMerieux SA
Cepheid (Danaher Corporation)
QuidelOrtho Corporation
Hologic, Inc.
Becton, Dickinson and Company (BD)
GenMark Diagnostics (Roche)
Luminex Corporation (DiaSorin S.p.A.)
PerkinElmer, Inc. (Revvity, Inc.)
Agilent Technologies, Inc.
Eiken Chemical Co., Ltd.
Meridian Bioscience, Inc. (A Diebold Nixdorf Company)
Beijing Wantai Biological Pharmacy Co., Ltd.
Guangzhou Wondfo Biotech Co., Ltd.
Seegene Inc.
SD Biosensor, Inc.
Sysmex Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. APAC Influenza Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 APAC Influenza Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. APAC Influenza Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of influenza outbreaks
3.1.2 Advancements in diagnostic technologies
3.1.3 Rising healthcare expenditure in APAC
3.1.4 Growing awareness about early diagnosis
3.2 Market Challenges
3.2.1 High cost of advanced diagnostic equipment
3.2.2 Limited access to healthcare in rural areas
3.2.3 Stringent regulatory requirements
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Development of rapid diagnostic tests
3.3.3 Collaborations with public health organizations
3.3.4 Increasing investment in R&D for influenza diagnostics
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of AI in diagnostic processes
3.4.3 Growing demand for home testing kits
3.4.4 Focus on personalized medicine approaches
3.5 Government Regulation
3.5.1 Approval processes for diagnostic devices
3.5.2 Guidelines for influenza surveillance
3.5.3 Funding for public health initiatives
3.5.4 Regulations on data privacy in health diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. APAC Influenza Diagnostics Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. APAC Influenza Diagnostics Market Segmentation
8.1 By Test Type
8.1.1 Rapid Influenza Diagnostic Tests (RIDTs)
8.1.2 Reverse Transcription Polymerase Chain Reaction (RT?PCR) Tests
8.1.3 Molecular Point?of?Care Tests
8.1.4 Viral Culture Tests
8.1.5 Immunofluorescence and Other Traditional Tests
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic and Reference Laboratories
8.2.3 Point?of?Care Testing (POCT) Settings and Clinics
8.2.4 Home Care and Over?the?Counter Testing
8.2.5 Research and Academic Institutes
8.3 By Country
8.3.1 China
8.3.2 Japan
8.3.3 India
8.3.4 South Korea
8.3.5 Australia & New Zealand
8.3.6 ASEAN (Indonesia, Thailand, Malaysia, Singapore, Vietnam, Others)
8.3.7 Rest of APAC
8.4 By Technology
8.4.1 Molecular Diagnostics
8.4.2 Immunoassay?based Rapid Tests
8.4.3 Next?Generation Sequencing (NGS)
8.4.4 Other Emerging and Hybrid Technologies
8.5 By Application
8.5.1 Clinical Diagnostics
8.5.2 Public Health Surveillance
8.5.3 Research and Drug/Vaccine Development
8.5.4 Occupational and Travel Health Screening
8.6 By Setting of Care
8.6.1 Centralized Laboratory Testing
8.6.2 Decentralized and Near?Patient Testing
8.6.3 Retail and Pharmacy?based Testing
8.6.4 Mobile and Community Health Programs
8.7 By Payer Type
8.7.1 Public and Social Health Insurance
8.7.2 Private Insurance
8.7.3 Out?of?Pocket Payments
8.7.4 Donor?funded and NGO?supported Programs
9. APAC Influenza Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 APAC Influenza Diagnostics Revenue (USD Million)
9.2.3 APAC Revenue CAGR in Influenza Diagnostics (Historical & Forecast)
9.2.4 APAC Market Share in Influenza Diagnostics (%)
9.2.5 Product Portfolio Breadth (Test Types and Technologies Covered)
9.2.6 Geographic Coverage within APAC (Number of Countries/Markets Served)
9.2.7 Installed Base of Instruments and Analyzers in APAC
9.2.8 Volume of Influenza Tests Performed Annually in APAC
9.2.9 Time?to?Result of Key Influenza Assays (Average Minutes)
9.2.10 Regulatory Approvals in APAC (e.g., PMDA, NMPA, CDSCO, TGA)
9.2.11 R&D Intensity in Respiratory / Influenza Diagnostics (% of Revenue)
9.2.12 Pricing Positioning of Key Assays (Premium / Mid?range / Value)
9.2.13 Channel Strength (Direct vs Distributor Network Coverage)
9.2.14 Strategic Partnerships and Local Manufacturing Presence in APAC
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 F. Hoffmann?La Roche Ltd
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers AG
9.5.4 Thermo Fisher Scientific Inc.
9.5.5 bioMérieux SA
9.5.6 Cepheid (Danaher Corporation)
9.5.7 QuidelOrtho Corporation
9.5.8 Hologic, Inc.
9.5.9 Becton, Dickinson and Company (BD)
9.5.10 GenMark Diagnostics (Roche)
9.5.11 Luminex Corporation (DiaSorin S.p.A.)
9.5.12 PerkinElmer, Inc. (Revvity, Inc.)
9.5.13 Agilent Technologies, Inc.
9.5.14 Eiken Chemical Co., Ltd.
9.5.15 Meridian Bioscience, Inc. (A Diebold Nixdorf Company)
9.5.16 Beijing Wantai Biological Pharmacy Co., Ltd.
9.5.17 Guangzhou Wondfo Biotech Co., Ltd.
9.5.18 Seegene Inc.
9.5.19 SD Biosensor, Inc.
9.5.20 Sysmex Corporation
10. APAC Influenza Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Diagnostic Infrastructure
10.2.2 Spending on Research and Development
10.2.3 Budget for Training and Capacity Building
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Diagnostic Accuracy
10.3.2 Issues with Supply Chain Management
10.3.3 Barriers to Technology Adoption
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Diagnostic Outcomes
10.5.2 Expansion into New Applications
10.5.3 Long-term Cost Savings
11. APAC Influenza Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Model Exploration
1.4 Key Partnerships Identification
1.5 Customer Segmentation Analysis
1.6 Competitive Landscape Overview
1.7 Risk Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Customer Engagement Tactics
2.7 Performance Metrics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Partnership with Healthcare Providers
3.6 Logistics and Supply Chain Management
3.7 Performance Evaluation
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discounting Strategies
4.7 Pricing Optimization Techniques
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
5.4 Feedback Mechanisms
5.5 Innovation Opportunities
5.6 Market Entry Barriers
5.7 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Systems
6.4 Relationship Management Tools
6.5 Engagement Strategies
6.6 Retention Strategies
6.7 Performance Metrics
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Approaches
7.4 Competitive Differentiation
7.5 Value Delivery Mechanisms
7.6 Performance Metrics
7.7 Future Outlook
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
8.5 Market Research Activities
8.6 Performance Monitoring
8.7 Continuous Improvement Processes
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
10.5 Risk Assessment
10.6 Performance Metrics
10.7 Future Outlook
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Financial Projections
11.4 Risk Assessment
11.5 Performance Metrics
11.6 Future Outlook
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnership Evaluation
12.3 Risk Mitigation Strategies
12.4 Performance Metrics
12.5 Future Outlook
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
13.3 Financial Projections
13.4 Risk Assessment
13.5 Performance Metrics
13.6 Future Outlook
14. Potential Partner List
14.1 Distributors
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations such as WHO and CDC regarding influenza trends in the APAC region
- Review of market studies and white papers from industry associations and research firms focusing on diagnostics technologies
- Examination of government health statistics and epidemiological data to understand influenza prevalence and testing rates
Primary Research
- Interviews with key opinion leaders in the healthcare sector, including virologists and public health officials
- Surveys conducted with laboratory managers and diagnostic product distributors to gather insights on market needs and challenges
- Focus group discussions with healthcare providers to assess the adoption of influenza diagnostic tools and technologies
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including market reports and expert interviews
- Triangulation of quantitative data from surveys with qualitative insights from expert interviews
- Sanity checks through peer reviews and feedback from industry experts to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall healthcare expenditure in APAC countries and its allocation to diagnostic services
- Analysis of the influenza incidence rates and corresponding diagnostic testing rates across different demographics
- Incorporation of government initiatives and funding for influenza prevention and control programs
Bottom-up Modeling
- Collection of sales data from leading diagnostic manufacturers and distributors in the APAC region
- Estimation of market share based on product types, including rapid tests, PCR tests, and serological tests
- Volume and pricing analysis to derive revenue estimates for each diagnostic segment
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market growth based on historical data trends
- Scenario modeling based on potential influenza outbreaks and their impact on diagnostic demand
- Development of best-case, worst-case, and most-likely scenarios to account for uncertainties in healthcare policies and technology adoption
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Laboratories | 60 | Laboratory Directors, Epidemiologists |
| Private Diagnostic Centers | 70 | Operations Managers, Clinical Pathologists |
| Healthcare Providers (Hospitals) | 90 | Infectious Disease Specialists, Hospital Administrators |
| Government Health Agencies | 40 | Public Health Officials, Policy Makers |
| Diagnostic Equipment Manufacturers | 50 | Product Managers, Sales Executives |
Frequently Asked Questions
What is the current value of the APAC Influenza Diagnostics Market?
The APAC Influenza Diagnostics Market is valued at approximately USD 0.55 billion, driven by the increasing prevalence of influenza outbreaks and advancements in diagnostic technologies, particularly in molecular and point-of-care testing methods.