APAC Intrauterine Devices Market Overview
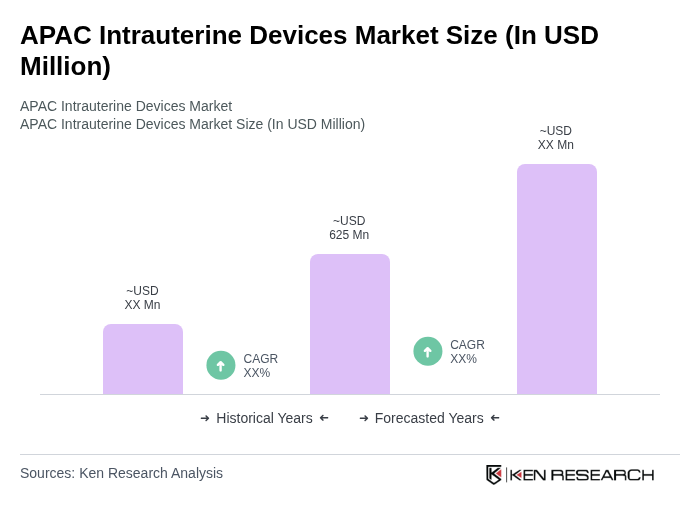
- The APAC Intrauterine Devices Market is valued at USD 625 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of family planning, rising healthcare expenditure, and the growing acceptance of long-term contraceptive methods among women. The market has seen a significant uptick in demand due to the emphasis on reproductive health and the availability of various IUD options, supported by technological advancements such as reusable and postpartum IUDs.
- Countries such as China and India dominate the market due to their large populations and increasing healthcare investments. China accounts for approximately USD 290 million in market value, while India represents USD 220 million, collectively driving regional growth. Additionally, Japan and South Korea are significant players, driven by advanced healthcare infrastructure and a high level of awareness regarding reproductive health. The urbanization and changing societal norms in Southeast Asia also contribute to the market's growth in these regions.
- Government initiatives and public health programs play a crucial role in market expansion. Multiple governments and non-profit organizations across the APAC region are actively promoting the use of IUDs in conjunction with educating populations about birth control measures and family planning. These coordinated efforts, combined with technological improvements that have resulted in effective contraception with minimal adverse effects, are expected to propel market growth throughout the forecast period.
APAC Intrauterine Devices Market Segmentation
By Type:The market is segmented into various types of intrauterine devices, including Copper IUDs, Hormonal IUDs, Non-hormonal IUDs, and Others. Among these, Copper IUDs remain widely used due to their effectiveness and long duration of action. Hormonal IUDs are gaining popularity as they offer additional benefits such as reduced menstrual bleeding. Non-hormonal IUDs cater to a niche market, while the 'Others' category includes newer innovations in IUD technology.
By End-User:The end-user segmentation includes Hospitals, Clinics, Home care settings, and Others. Hospitals are the primary end-users due to their comprehensive healthcare services and facilities for IUD insertion. Clinics also play a significant role, especially in urban areas where women seek family planning services. Home care settings are emerging as a viable option for IUD management, while the 'Others' category includes community health organizations and NGOs.
APAC Intrauterine Devices Market Competitive Landscape
The APAC Intrauterine Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Bayer AG, CooperSurgical, Inc., Merck & Co., Inc., Teva Pharmaceutical Industries Ltd., HRA Pharma, Pregna International Limited, Ocon Medical Ltd., Tatum Medical, DKT International, Ferring Pharmaceuticals, Mylan N.V., Hologic, Inc., Semaine, Ameda, Inc., Becton, Dickinson and Company, Shanghai Dahua Medical Apparatus Co., Ltd., Hangzhou Kangnai Medical Instrument Co., Ltd., Jiangsu Yuyue Medical Equipment & Supply Co., Ltd., Medisafe International, Meditech International contribute to innovation, geographic expansion, and service delivery in this space.
APAC Intrauterine Devices Market Industry Analysis
Growth Drivers
- Increasing Awareness of Reproductive Health:The APAC region has seen a significant rise in reproductive health awareness, with over 60% of women aged 15-49 now knowledgeable about various contraceptive methods, according to the World Health Organization. This awareness is bolstered by educational campaigns, which have increased access to information. In future, the region is expected to allocate approximately $1.5 billion towards reproductive health initiatives, further driving the adoption of intrauterine devices (IUDs) as a reliable contraceptive option.
- Rising Demand for Long-term Contraceptive Solutions:The demand for long-term contraceptive solutions is surging, with IUD usage projected to increase by 20% in the APAC region in future. This trend is driven by a growing preference for methods that require minimal maintenance. In countries like India and Indonesia, where the population is expected to reach 1.6 billion and 290 million respectively, the need for effective family planning solutions is critical, making IUDs an attractive option for many women.
- Government Initiatives Promoting Family Planning:Governments across the APAC region are increasingly investing in family planning initiatives, with funding expected to exceed $1 billion in future. Countries like Thailand and Vietnam have implemented policies to enhance access to contraceptives, including IUDs. These initiatives aim to reduce maternal mortality rates and improve overall reproductive health, thereby fostering a conducive environment for the growth of the IUD market in the region.
Market Challenges
- Cultural Resistance to Contraceptive Methods:Cultural attitudes towards contraceptive methods pose significant challenges in the APAC region. In many countries, traditional beliefs and stigma surrounding contraceptive use hinder acceptance. For instance, in rural areas of Pakistan, only 30% of women use any form of contraception, with IUDs being particularly underutilized. This cultural resistance can limit market penetration and growth opportunities for IUD manufacturers.
- Regulatory Hurdles in Product Approval:The regulatory landscape for contraceptives in the APAC region is complex, with varying standards across countries. For example, the approval process for new IUDs can take up to 24 months in India, compared to 12 months in Japan. These regulatory hurdles can delay product launches and increase costs for manufacturers, ultimately impacting market growth and innovation in the IUD sector.
APAC Intrauterine Devices Market Future Outlook
The future of the APAC intrauterine devices market appears promising, driven by increasing investments in women's health and a shift towards personalized contraceptive solutions. As governments and healthcare providers collaborate to enhance access to reproductive health services, the adoption of IUDs is expected to rise. Additionally, the growing trend of telehealth consultations will facilitate better education and access to IUDs, further supporting market growth in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets in Southeast Asia present significant opportunities for IUD manufacturers. With a combined population of over 650 million, countries like Myanmar and Cambodia are experiencing increased demand for effective contraceptive solutions. Targeted marketing and distribution strategies can capitalize on this growing need, potentially increasing market share for IUDs in these regions.
- Development of Innovative IUD Products:There is a growing opportunity for the development of innovative IUD products that cater to diverse consumer needs. For instance, the introduction of non-hormonal IUDs and those with extended duration of effectiveness can attract a broader user base. Research and development investments in this area could lead to significant advancements, enhancing the appeal of IUDs in the competitive contraceptive market.