Region:Asia
Author(s):Dev
Product Code:KRAA3944
Pages:99
Published On:January 2026
By Device Type:The device type segmentation includes various technologies used in the treatment of uterine fibroids. Surgical technologies, including laparoscopic and hysteroscopic systems, remain widely used in high-volume markets such as China and other APAC countries. Uterine Artery Embolization Devices are important in interventional radiology settings and are favored for their minimally invasive nature and effectiveness in reducing fibroid size in women seeking uterus-preserving options. Radiofrequency Ablation Devices and Magnetic Resonance-Guided Focused Ultrasound (MRgFUS)/High-Intensity Focused Ultrasound (HIFU) Systems are gaining traction in APAC tertiary centers as non-invasive or minimally invasive approaches offering shorter recovery times and reduced hospital stays. The market is also supported by Hysteroscopic and Laparoscopic Myomectomy Devices, which continue to be preferred for surgical removal of fibroids, particularly in women with multiple or large fibroids and in centers where advanced image-guided technologies are less accessible.

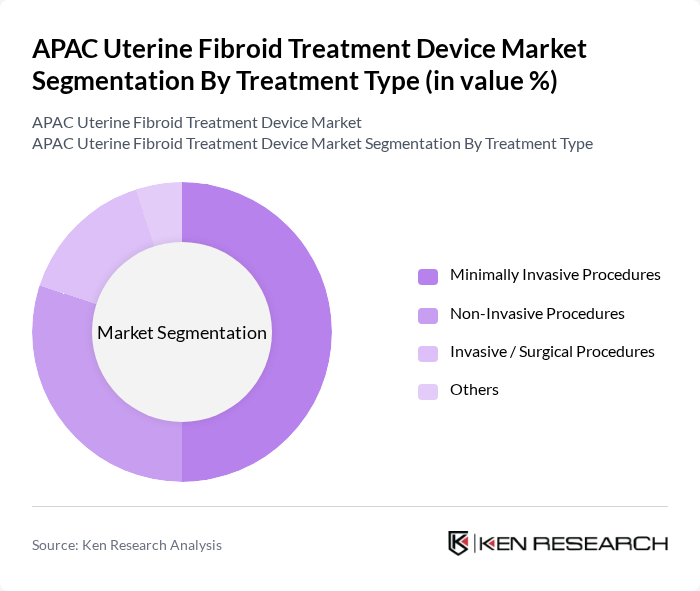
By Treatment Type:The treatment type segmentation encompasses various approaches to managing uterine fibroids. Minimally Invasive Procedures, including laparoscopic myomectomy, hysteroscopic resection, uterine artery embolization, and radiofrequency ablation, are the most popular due to reduced recovery time, lower complication rates, and strong clinical preference for uterus-preserving options. Non-Invasive Procedures, particularly MR-guided focused ultrasound and HIFU, are also gaining traction in APAC as technological advances and expanding access to advanced imaging systems improve their effectiveness and availability. Invasive/Surgical Procedures, such as open myomectomy and hysterectomy, remain relevant for complex cases, large fibroid burden, or where other treatments are ineffective, especially in settings with limited access to advanced minimally invasive technology. Other treatment types, including hybrid approaches and adjunctive pharmacologic support used perioperatively with devices, are less commonly utilized but contribute to comprehensive management strategies.
The APAC Uterine Fibroid Treatment Device Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Johnson & Johnson (Ethicon), Siemens Healthineers AG, GE HealthCare Technologies Inc., Hologic, Inc., Olympus Corporation, Stryker Corporation, FUJIFILM Holdings Corporation, Canon Medical Systems Corporation, Philips Healthcare (Royal Philips), B. Braun Melsungen AG, Cook Medical LLC, Merit Medical Systems, Inc., Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC uterine fibroid treatment device market appears promising, driven by technological innovations and increasing healthcare investments. As the demand for personalized medicine grows, treatment options will likely become more tailored to individual patient needs. Additionally, the integration of digital health technologies, such as telemedicine and mobile health applications, will enhance patient engagement and access to care, ultimately improving treatment outcomes and market dynamics in the region.
| Segment | Sub-Segments |
|---|---|
| By Device Type | Uterine Artery Embolization Devices Radiofrequency Ablation Devices Magnetic Resonance-Guided Focused Ultrasound (MRgFUS) Systems High-Intensity Focused Ultrasound (HIFU) Systems Hysteroscopic and Laparoscopic Myomectomy Devices Hysterectomy Devices and Systems Others |
| By Treatment Type | Minimally Invasive Procedures Non-Invasive Procedures Invasive / Surgical Procedures Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Application | Symptomatic Uterine Fibroids Asymptomatic Uterine Fibroids Infertility Associated with Fibroids Others |
| By Patient Demographics | Age Group Socioeconomic Status Urban vs Rural Population Others |
| By Country | China India Japan South Korea Australia & New Zealand Rest of APAC |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gynecological Clinics | 120 | Gynecologists, Clinic Managers |
| Hospitals with Uterine Fibroid Treatment Programs | 100 | Surgeons, Hospital Administrators |
| Patient Advocacy Groups | 60 | Patient Representatives, Health Educators |
| Medical Device Distributors | 80 | Sales Managers, Product Specialists |
| Health Insurance Providers | 70 | Policy Analysts, Claims Managers |
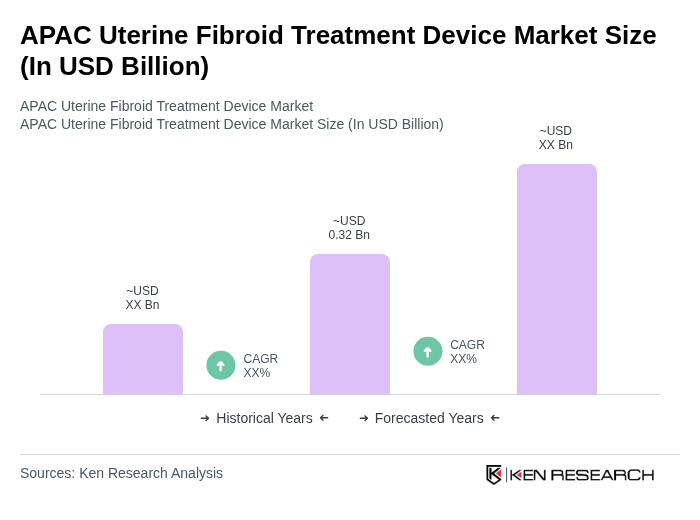
The APAC Uterine Fibroid Treatment Device Market is valued at approximately USD 0.32 billion, representing a significant share of the global market, which is estimated at around USD 1.2 billion. This growth is driven by increasing prevalence and advancements in treatment technologies.