About the Report
Base Year 2024Listen to the audio summary
Australia Dermal Toxicity Testing Market Overview
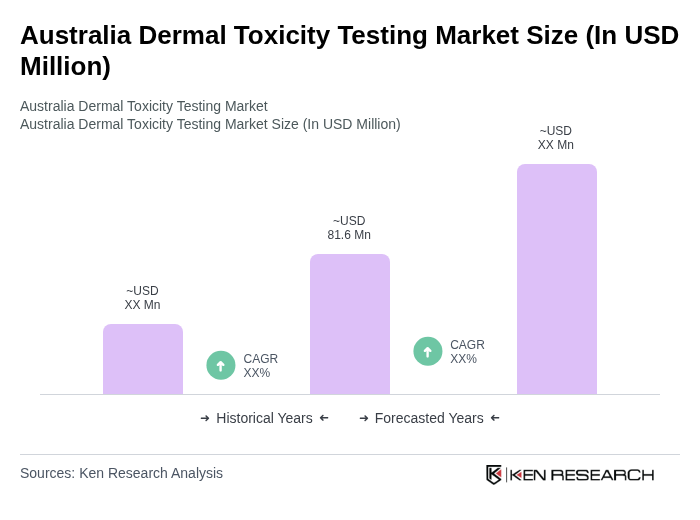
- The Australia Dermal Toxicity Testing Market is valued at approximately USD 81.6 million, based on a five-year historical analysis. This growth is primarily driven by increasing regulatory scrutiny on cosmetic and pharmaceutical products, alongside rising consumer awareness regarding product safety and efficacy. The demand for reliable testing methods has surged, leading to advancements in testing technologies and methodologies, with in-vitro methods gaining significant traction due to ethical advantages and regulatory acceptance by bodies such as the OECD.
- Key cities such as Sydney, Melbourne, and Brisbane dominate the market due to their robust pharmaceutical and cosmetic industries. These urban centers are home to numerous research institutions and testing laboratories, fostering innovation and collaboration. The concentration of industry players and regulatory bodies in these regions further enhances their market leadership.
- The National Industrial Chemicals Notification and Assessment Scheme (NICNAS), issued by the Australian Department of Climate Change, Energy, Environment and Water, mandates comprehensive safety assessments for new chemical substances. This regulation aims to ensure that all industrial chemicals, including those used in cosmetics and personal care products, meet stringent safety standards before they can be marketed, with compliance requirements extending to hazard classification and labeling of chemical products.
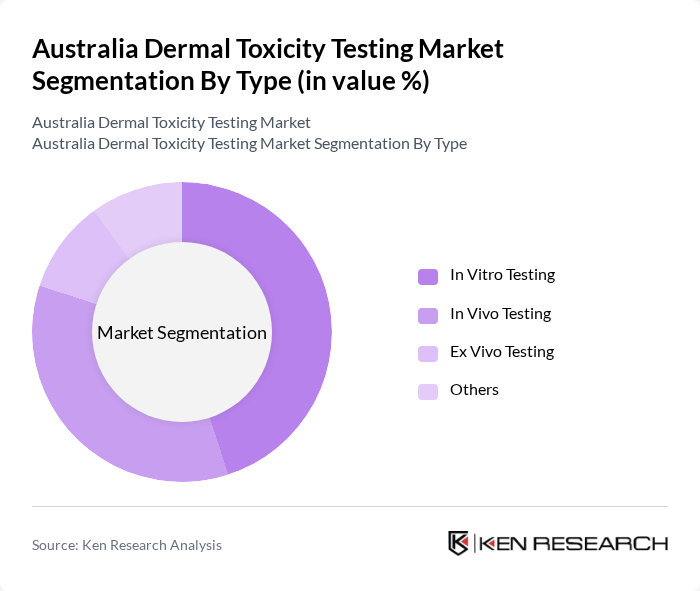
Australia Dermal Toxicity Testing Market Segmentation
By Type:The market is segmented into various testing types, including In Vitro Testing, In Vivo Testing, Ex Vivo Testing, and Others. In Vitro Testing is gaining traction due to its ethical advantages and cost-effectiveness, commanding approximately 49.5% of the global dermal toxicity testing market share, while In Vivo Testing remains essential for comprehensive safety assessments. Ex Vivo Testing is utilized for specific applications, and the "Others" category includes emerging methodologies such as in-silico assays, which are recording the fastest growth rates in the sector.
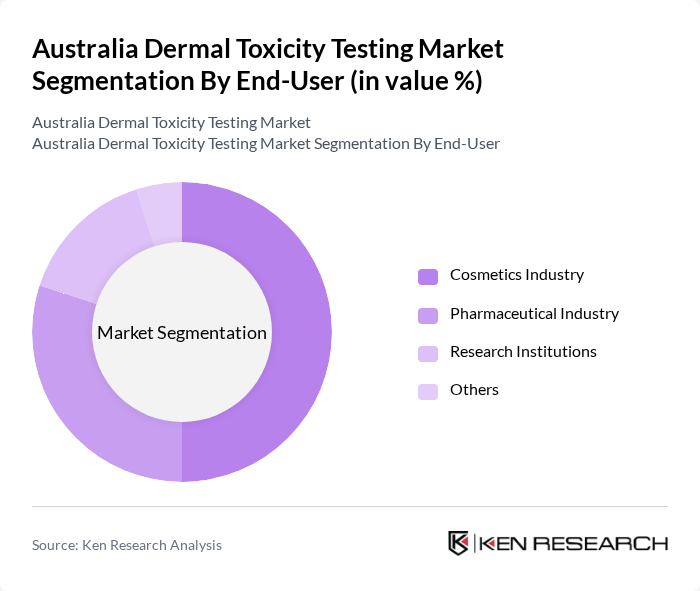
By End-User:The primary end-users of dermal toxicity testing services include the Cosmetics Industry, Pharmaceutical Industry, Research Institutions, and Others. The Cosmetics Industry is the largest segment, driven by stringent regulations and consumer demand for safe products, while skin sensitization testing represents over 41.2% of the global market share due to regulatory requirements. The Pharmaceutical Industry also significantly contributes to the market, focusing on drug safety and efficacy, with the healthcare sector accounting for substantial market share.
Australia Dermal Toxicity Testing Market Competitive Landscape
The Australia Dermal Toxicity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as ChemCentre, Eurofins Scientific, SGS Australia, Intertek Group, Charles River Laboratories, Covance, Q2 Solutions, BioReliance, Labcorp, Toxikon, Medpace, WIL Research, Envigo, Charles Lawrence International, Axiom Laboratories contribute to innovation, geographic expansion, and service delivery in this space.
Australia Dermal Toxicity Testing Market Industry Analysis
Growth Drivers
- Increasing Regulatory Requirements:The Australian government has implemented stringent regulations for dermal toxicity testing, driven by the need for consumer safety. In future, the Therapeutic Goods Administration (TGA) is expected to enforce compliance with updated guidelines, impacting over 1,300 cosmetic products. This regulatory landscape compels companies to invest in comprehensive testing, ensuring that products meet safety standards, thereby driving market growth significantly.
- Rising Awareness of Skin Safety:With a growing consumer focus on skin health, the demand for safe cosmetic products is surging. In future, the Australian skincare market is projected to reach AUD 3.9 billion, reflecting a 12% increase from the previous year. This heightened awareness encourages manufacturers to prioritize dermal toxicity testing, ensuring their products are safe and effective, thus propelling market expansion.
- Advancements in Testing Technologies:The development of innovative testing technologies, such as in vitro methods, is revolutionizing the dermal toxicity testing landscape. In future, investments in these technologies are expected to exceed AUD 180 million, enhancing testing efficiency and accuracy. This shift not only reduces reliance on animal testing but also aligns with consumer preferences for ethical practices, further stimulating market growth.
Market Challenges
- High Costs of Testing Procedures:The financial burden associated with dermal toxicity testing remains a significant challenge for companies. In future, the average cost of comprehensive testing procedures is estimated to be around AUD 55,000 per product. This high expenditure can deter smaller firms from entering the market, limiting competition and innovation within the industry.
- Limited Availability of Skilled Professionals:The dermal toxicity testing sector faces a shortage of qualified professionals, which hampers growth. In future, it is estimated that Australia will have a deficit of approximately 1,200 toxicologists and dermatologists. This scarcity not only affects the quality of testing but also delays product launches, posing a significant challenge for companies aiming to comply with regulatory standards.
Australia Dermal Toxicity Testing Market Future Outlook
The future of the Australia dermal toxicity testing market appears promising, driven by technological advancements and evolving consumer preferences. As the demand for safe and effective cosmetic products continues to rise, companies are likely to invest in innovative testing methods. Additionally, the integration of artificial intelligence in toxicity assessments is expected to enhance accuracy and efficiency. These trends will shape the market landscape, fostering a more competitive environment while addressing regulatory and ethical concerns.
Market Opportunities
- Expansion of E-commerce in Cosmetics:The growth of e-commerce platforms is creating new opportunities for dermal toxicity testing. In future, online cosmetic sales in Australia are projected to reach AUD 1.5 billion, prompting brands to ensure product safety through rigorous testing. This trend encourages companies to invest in testing services to build consumer trust and enhance their market presence.
- Development of Alternative Testing Methods:The increasing demand for cruelty-free products is driving the development of alternative testing methods. In future, investments in non-animal testing technologies are expected to surpass AUD 100 million. This shift not only aligns with consumer preferences but also opens new avenues for companies to innovate and differentiate their products in a competitive market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | In Vitro Testing In Vivo Testing Ex Vivo Testing Others |
| By End-User | Cosmetics Industry Pharmaceutical Industry Research Institutions Others |
| By Product Type | Skincare Products Haircare Products Makeup Products Others |
| By Testing Method | Patch Testing Repeated Insult Patch Testing (RIPT) Human Repeat Insult Patch Test (HRIPT) Others |
| By Region | New South Wales Victoria Queensland Others |
| By Application | Dermatological Products Household Products Industrial Products Others |
| By Regulatory Compliance | TGA Compliance APVMA Compliance NICNAS Compliance Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, National Industrial Chemicals Notification and Assessment Scheme)
Manufacturers and Producers of Dermal Testing Products
Pharmaceutical Companies
Cosmetic and Personal Care Product Manufacturers
Biotechnology Firms
Industry Associations (e.g., Australian Society of Cosmetic Chemists)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
ChemCentre
Eurofins Scientific
SGS Australia
Intertek Group
Charles River Laboratories
Covance
Q2 Solutions
BioReliance
Labcorp
Toxikon
Medpace
WIL Research
Envigo
Charles Lawrence International
Axiom Laboratories
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia Dermal Toxicity Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia Dermal Toxicity Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia Dermal Toxicity Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Regulatory Requirements
3.1.2 Rising Awareness of Skin Safety
3.1.3 Growth in Cosmetic and Personal Care Industries
3.1.4 Advancements in Testing Technologies
3.2 Market Challenges
3.2.1 High Costs of Testing Procedures
3.2.2 Limited Availability of Skilled Professionals
3.2.3 Stringent Regulatory Compliance
3.2.4 Public Perception and Ethical Concerns
3.3 Market Opportunities
3.3.1 Expansion of E-commerce in Cosmetics
3.3.2 Development of Alternative Testing Methods
3.3.3 Collaborations with Research Institutions
3.3.4 Increasing Demand for Natural Products
3.4 Market Trends
3.4.1 Shift Towards In Vitro Testing
3.4.2 Integration of AI in Toxicity Testing
3.4.3 Focus on Sustainable and Ethical Testing
3.4.4 Growth of Personalized Skincare Solutions
3.5 Government Regulation
3.5.1 Australian Pesticides and Veterinary Medicines Authority (APVMA) Guidelines
3.5.2 National Industrial Chemicals Notification and Assessment Scheme (NICNAS)
3.5.3 Therapeutic Goods Administration (TGA) Regulations
3.5.4 Compliance with OECD Testing Guidelines
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia Dermal Toxicity Testing Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia Dermal Toxicity Testing Market Segmentation
8.1 By Type
8.1.1 In Vitro Testing
8.1.2 In Vivo Testing
8.1.3 Ex Vivo Testing
8.1.4 Others
8.2 By End-User
8.2.1 Cosmetics Industry
8.2.2 Pharmaceutical Industry
8.2.3 Research Institutions
8.2.4 Others
8.3 By Product Type
8.3.1 Skincare Products
8.3.2 Haircare Products
8.3.3 Makeup Products
8.3.4 Others
8.4 By Testing Method
8.4.1 Patch Testing
8.4.2 Repeated Insult Patch Testing (RIPT)
8.4.3 Human Repeat Insult Patch Test (HRIPT)
8.4.4 Others
8.5 By Region
8.5.1 New South Wales
8.5.2 Victoria
8.5.3 Queensland
8.5.4 Others
8.6 By Application
8.6.1 Dermatological Products
8.6.2 Household Products
8.6.3 Industrial Products
8.6.4 Others
8.7 By Regulatory Compliance
8.7.1 TGA Compliance
8.7.2 APVMA Compliance
8.7.3 NICNAS Compliance
8.7.4 Others
9. Australia Dermal Toxicity Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Operational Efficiency
9.2.9 Brand Recognition
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 ChemCentre
9.5.2 Eurofins Scientific
9.5.3 SGS Australia
9.5.4 Intertek Group
9.5.5 Charles River Laboratories
9.5.6 Covance
9.5.7 Q2 Solutions
9.5.8 BioReliance
9.5.9 Labcorp
9.5.10 Toxikon
9.5.11 Medpace
9.5.12 WIL Research
9.5.13 Envigo
9.5.14 Charles Lawrence International
9.5.15 Axiom Laboratories
10. Australia Dermal Toxicity Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government Procurement Policies
10.1.2 Budget Allocations for Toxicity Testing
10.1.3 Collaboration with Private Sector
10.1.4 Evaluation Criteria for Suppliers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in R&D for Toxicity Testing
10.2.2 Budget Trends in Cosmetic Safety
10.2.3 Funding for Regulatory Compliance
10.2.4 Partnerships with Testing Facilities
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Compliance with Regulations
10.3.2 Need for Cost-Effective Testing Solutions
10.3.3 Demand for Faster Testing Turnaround
10.3.4 Concerns Over Ethical Testing Practices
10.4 User Readiness for Adoption
10.4.1 Awareness of Testing Methods
10.4.2 Willingness to Invest in Safety
10.4.3 Training Needs for Staff
10.4.4 Integration with Existing Processes
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Testing Effectiveness
10.5.2 Long-term Cost Savings
10.5.3 Expansion into New Product Lines
10.5.4 Feedback Mechanisms for Continuous Improvement
11. Australia Dermal Toxicity Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategy
2.5 Digital Marketing Approaches
2.6 Trade Show Participation
2.7 Customer Engagement Tactics
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Partnerships with Distributors
3.6 Logistics and Supply Chain Management
3.7 Inventory Management Solutions
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Value-Based Pricing Models
4.6 Discounting Strategies
4.7 Bundling Opportunities
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
5.4 Feedback from End-Users
5.5 Innovation Opportunities
5.6 Market Entry Barriers
5.7 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Community Engagement Initiatives
6.5 Customer Education Programs
6.6 Relationship Management Tools
6.7 Personalization Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Unique Selling Points
7.4 Customer-Centric Innovations
7.5 Competitive Advantages
7.6 Market Differentiation Strategies
7.7 Long-term Value Creation
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Market Research Activities
8.5 Training and Development Programs
8.6 Quality Assurance Processes
8.7 Stakeholder Engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from Australian regulatory bodies on dermal toxicity standards
- Review of scientific literature and journals focusing on dermal toxicity testing methodologies
- Examination of market reports and white papers from industry associations related to dermatological products
Primary Research
- Interviews with toxicologists and dermatologists specializing in dermal toxicity testing
- Surveys with laboratory managers in accredited testing facilities across Australia
- Focus groups with product developers in the cosmetics and pharmaceutical sectors
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from regulatory updates, industry reports, and expert opinions
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national health expenditure related to dermatological treatments
- Segmentation of the market by application areas such as pharmaceuticals, cosmetics, and personal care products
- Incorporation of trends in consumer awareness and regulatory changes affecting dermal toxicity testing
Bottom-up Modeling
- Collection of data on testing volumes from leading dermal toxicity testing laboratories
- Cost analysis based on service pricing for various testing methodologies
- Estimation of market size through volume x cost calculations for different product categories
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as regulatory changes and market growth rates
- Scenario modeling based on potential shifts in consumer preferences and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Dermal Testing | 100 | Toxicologists, Regulatory Affairs Specialists |
| Cosmetic Product Testing | 80 | Product Development Managers, Quality Assurance Officers |
| Personal Care Product Testing | 70 | Research Scientists, Compliance Managers |
| Contract Testing Laboratories | 60 | Laboratory Directors, Operations Managers |
| Regulatory Bodies and Associations | 50 | Policy Makers, Industry Analysts |
Frequently Asked Questions
What is the current value of the Australia Dermal Toxicity Testing Market?
The Australia Dermal Toxicity Testing Market is valued at approximately USD 81.6 million, reflecting a significant growth driven by increasing regulatory scrutiny and consumer awareness regarding product safety and efficacy in the cosmetic and pharmaceutical sectors.