About the Report
Base Year 2024Listen to the audio summary
Kuwait Dermal Toxicity Testing Market Overview
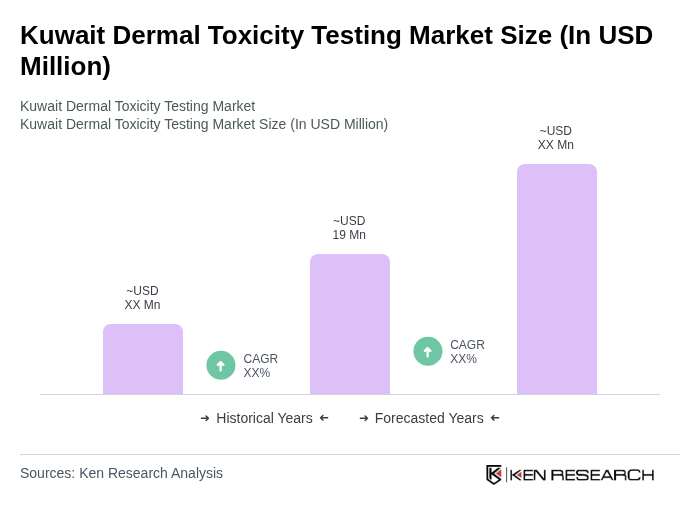
- The Kuwait Dermal Toxicity Testing Market is valued at approximately USD 19 million, based on a five-year historical analysis. This growth is primarily driven by increasing regulatory requirements for safety testing in the pharmaceutical and cosmetic industries, alongside rising consumer awareness regarding product safety and efficacy. The demand for reliable toxicity testing methods has surged, leading to a robust market environment. The global dermal toxicity testing market expansion, driven by regulatory bans on animal testing in cosmetics and accelerated acceptance of in-vitro and in-silico assays by regulatory bodies, continues to influence regional market dynamics.
- Kuwait City is the dominant hub for the Dermal Toxicity Testing Market due to its concentration of pharmaceutical and cosmetic companies, as well as research institutions. The city's strategic location and advanced healthcare infrastructure facilitate collaboration between industry players and regulatory bodies, enhancing the overall market dynamics.
- Kuwait's regulatory framework mandates comprehensive dermal toxicity testing for cosmetic and pharmaceutical products before market entry, aligning with international standards established by organizations such as the OECD and the European Chemicals Agency. This regulatory alignment ensures consumer safety and fosters a more responsible and transparent market environment, with particular emphasis on skin irritation and sensitization testing protocols.
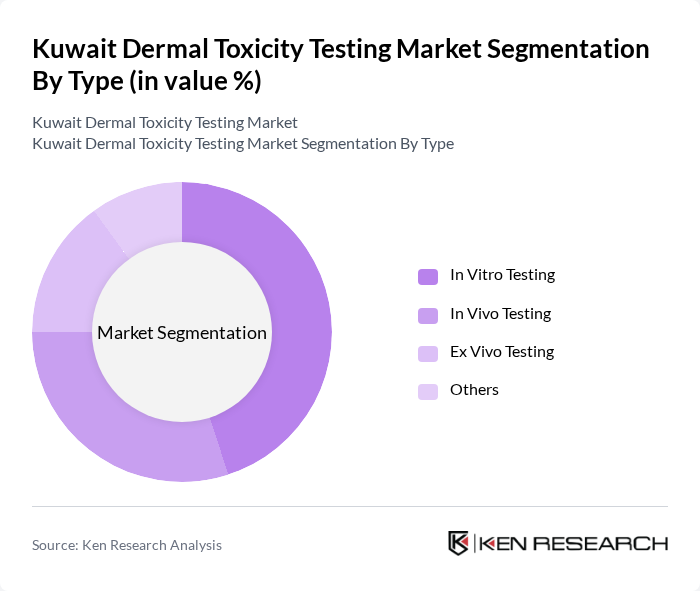
Kuwait Dermal Toxicity Testing Market Segmentation
By Type:The market is segmented into various testing types, including In Vitro Testing, In Vivo Testing, Ex Vivo Testing, and Others. In Vitro Testing is currently the leading sub-segment due to its cost-effectiveness and ethical advantages over animal testing. The increasing adoption of alternative testing methods is driving growth in this segment, as companies seek to comply with regulatory standards while minimizing animal use. Globally, in-vitro reconstructed human epidermis (RHE) systems hold the largest market share, with in-silico computational assays emerging as the fastest-growing segment.
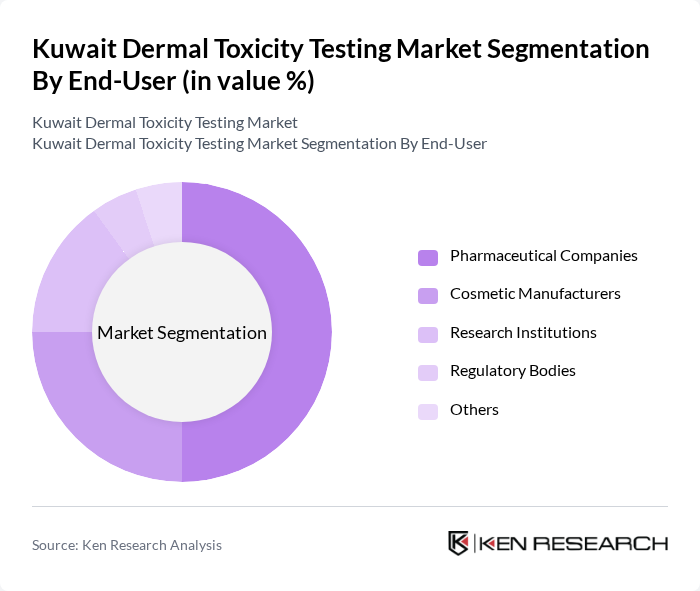
By End-User:The end-user segmentation includes Pharmaceutical Companies, Cosmetic Manufacturers, Research Institutions, Regulatory Bodies, and Others. Pharmaceutical Companies dominate this segment, driven by stringent regulatory requirements for drug safety and efficacy testing. The increasing focus on dermatological products and personalized medicine is further propelling the demand for dermal toxicity testing among these companies. The outsourcing of toxicity testing services to specialized contract research organizations continues to expand, reflecting industry trends toward specialized expertise and cost optimization.
Kuwait Dermal Toxicity Testing Market Competitive Landscape
The Kuwait Dermal Toxicity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Eurofins Scientific, SGS SA, Intertek Group plc, Charles River Laboratories, Covance Inc., BioReliance, Toxikon Corporation, WIL Research, Medpace, Labcorp Drug Development, Q2 Solutions, Envigo, Charles Lawrence International, AccuBioTech, ToxServices LLC contribute to innovation, geographic expansion, and service delivery in this space.
Kuwait Dermal Toxicity Testing Market Industry Analysis
Growth Drivers
- Increasing Regulatory Requirements for Safety Testing:The Kuwait government has intensified its regulatory framework, mandating that all cosmetic products undergo rigorous safety testing. In future, the Ministry of Health is expected to enforce compliance with new guidelines, impacting over 1,300 cosmetic products annually. This regulatory push is driven by a commitment to consumer safety, with the aim of reducing skin-related health issues, which have seen a 17% increase in reported cases over the past three years.
- Rising Awareness of Skin-Related Health Issues:Public awareness campaigns in Kuwait have highlighted the risks associated with harmful chemicals in cosmetics, leading to a 22% increase in demand for dermal toxicity testing services. The Kuwait Health Authority reported that skin diseases accounted for 14% of all health complaints in future. This growing concern is prompting both consumers and manufacturers to prioritize safety, thereby driving the demand for comprehensive testing solutions.
- Growth in the Cosmetics and Personal Care Industry:The cosmetics market in Kuwait is projected to reach $1.7 billion in future, reflecting a robust growth trajectory. This expansion is fueled by a burgeoning middle class and increased disposable income, which has risen by 10% since 2022. As more brands enter the market, the need for reliable dermal toxicity testing becomes critical to ensure product safety and compliance, further propelling market growth.
Market Challenges
- High Costs Associated with Testing Procedures:The financial burden of dermal toxicity testing can be significant, with costs ranging from $6,000 to $16,000 per product. This high expense poses a barrier for smaller companies, limiting their ability to compete in the market. As the demand for testing increases, companies may struggle to allocate sufficient budgets, potentially leading to non-compliance with safety regulations and risking consumer health.
- Limited Availability of Skilled Professionals:The Kuwait dermal toxicity testing sector faces a shortage of qualified professionals, with only 350 certified toxicologists available in the country. This scarcity hampers the ability of testing facilities to meet the growing demand for services. As the industry expands, the lack of skilled labor could lead to delays in testing processes, impacting product launches and compliance timelines for manufacturers.
Kuwait Dermal Toxicity Testing Market Future Outlook
The Kuwait dermal toxicity testing market is poised for significant evolution, driven by technological advancements and changing consumer preferences. The shift towards in vitro testing methods is expected to enhance efficiency and reduce costs, while the integration of AI in testing processes will streamline operations. Additionally, as consumers increasingly demand transparency and cruelty-free products, companies will need to adapt their testing protocols to align with these expectations, ensuring compliance with both local and international standards.
Market Opportunities
- Expansion of the Pharmaceutical Sector:The pharmaceutical industry in Kuwait is projected to grow by 12% in future, creating a substantial demand for dermal toxicity testing services. This growth presents an opportunity for testing facilities to diversify their offerings and cater to pharmaceutical companies seeking to ensure the safety of their products, thereby enhancing market potential.
- Increasing Demand for Cruelty-Free Testing Methods:With a rising consumer preference for ethical products, the demand for cruelty-free testing methods is expected to surge. In future, approximately 35% of consumers in Kuwait are anticipated to prioritize cruelty-free certifications when purchasing cosmetics, prompting manufacturers to seek compliant testing solutions that align with these values, thus opening new market avenues.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | In Vitro Testing In Vivo Testing Ex Vivo Testing Others |
| By End-User | Pharmaceutical Companies Cosmetic Manufacturers Research Institutions Regulatory Bodies Others |
| By Application | Skin Irritation Testing Skin Sensitization Testing Phototoxicity Testing Others |
| By Region | Central Kuwait Southern Kuwait Northern Kuwait Others |
| By Testing Method | Patch Testing Repeated Insult Patch Testing (RIPT) Human Repeat Insult Patch Test (HRIPT) Others |
| By Product Type | Skincare Products Haircare Products Makeup Products Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Kuwait Ministry of Health, Kuwait Environment Public Authority)
Pharmaceutical and Cosmetic Manufacturers
Dermatology Clinics and Research Facilities
Contract Research Organizations (CROs)
Health and Safety Regulatory Agencies
Biotechnology Firms
Quality Assurance and Control Agencies
Players Mentioned in the Report:
Eurofins Scientific
SGS SA
Intertek Group plc
Charles River Laboratories
Covance Inc.
BioReliance
Toxikon Corporation
WIL Research
Medpace
Labcorp Drug Development
Q2 Solutions
Envigo
Charles Lawrence International
AccuBioTech
ToxServices LLC
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Kuwait Dermal Toxicity Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Kuwait Dermal Toxicity Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Kuwait Dermal Toxicity Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing regulatory requirements for safety testing
3.1.2 Rising awareness of skin-related health issues
3.1.3 Growth in the cosmetics and personal care industry
3.1.4 Advancements in testing technologies
3.2 Market Challenges
3.2.1 High costs associated with testing procedures
3.2.2 Limited availability of skilled professionals
3.2.3 Stringent regulatory compliance requirements
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of the pharmaceutical sector
3.3.2 Increasing demand for cruelty-free testing methods
3.3.3 Collaborations with research institutions
3.3.4 Growth in e-commerce for cosmetic products
3.4 Market Trends
3.4.1 Shift towards in vitro testing methods
3.4.2 Integration of AI and machine learning in testing
3.4.3 Focus on sustainable and eco-friendly products
3.4.4 Rising consumer demand for transparency in testing
3.5 Government Regulation
3.5.1 Implementation of GCC regulations on cosmetic safety
3.5.2 Updates to the Kuwait Public Health Law
3.5.3 Guidelines from the Ministry of Health
3.5.4 International standards compliance (ISO, OECD)
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Kuwait Dermal Toxicity Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Kuwait Dermal Toxicity Testing Market Segmentation
8.1 By Type
8.1.1 In Vitro Testing
8.1.2 In Vivo Testing
8.1.3 Ex Vivo Testing
8.1.4 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Cosmetic Manufacturers
8.2.3 Research Institutions
8.2.4 Regulatory Bodies
8.2.5 Others
8.3 By Application
8.3.1 Skin Irritation Testing
8.3.2 Skin Sensitization Testing
8.3.3 Phototoxicity Testing
8.3.4 Others
8.4 By Region
8.4.1 Central Kuwait
8.4.2 Southern Kuwait
8.4.3 Northern Kuwait
8.4.4 Others
8.5 By Testing Method
8.5.1 Patch Testing
8.5.2 Repeated Insult Patch Testing (RIPT)
8.5.3 Human Repeat Insult Patch Test (HRIPT)
8.5.4 Others
8.6 By Product Type
8.6.1 Skincare Products
8.6.2 Haircare Products
8.6.3 Makeup Products
8.6.4 Others
8.7 By Policy Support
8.7.1 Government Grants
8.7.2 Tax Incentives
8.7.3 Research Funding
8.7.4 Others
9. Kuwait Dermal Toxicity Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Average Order Value
9.2.8 Cost of Customer Acquisition
9.2.9 Return on Investment (ROI)
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Eurofins Scientific
9.5.2 SGS SA
9.5.3 Intertek Group plc
9.5.4 Charles River Laboratories
9.5.5 Covance Inc.
9.5.6 BioReliance
9.5.7 Toxikon Corporation
9.5.8 WIL Research
9.5.9 Medpace
9.5.10 Labcorp Drug Development
9.5.11 Q2 Solutions
9.5.12 Envigo
9.5.13 Charles Lawrence International
9.5.14 AccuBioTech
9.5.15 ToxServices LLC
10. Kuwait Dermal Toxicity Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Commerce and Industry
10.1.3 Ministry of Environment
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Testing Facilities
10.2.2 Budget Allocation for R&D
10.2.3 Expenditure on Compliance and Safety
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Regulatory Compliance Challenges
10.3.2 Cost Management Issues
10.3.3 Quality Assurance Concerns
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of Testing Methods
10.4.2 Training and Skill Development
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Effectiveness
10.5.2 Feedback Mechanisms
10.5.3 Opportunities for Scaling
10.5.4 Others
11. Kuwait Dermal Toxicity Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing literature on dermal toxicity testing methodologies and regulations in Kuwait
- Review of market reports and publications from health and safety organizations
- Examination of government and regulatory body guidelines related to dermal toxicity testing
Primary Research
- Interviews with dermatologists and toxicologists specializing in skin-related health issues
- Surveys with laboratory managers and technicians involved in dermal toxicity testing
- Focus groups with industry stakeholders, including pharmaceutical and cosmetic companies
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from regulatory bodies, industry experts, and market trends
- Sanity checks through peer reviews and feedback from academic professionals in toxicology
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on Kuwait's healthcare expenditure on dermatological services
- Segmentation of the market by end-user categories such as pharmaceuticals, cosmetics, and research institutions
- Incorporation of trends in consumer awareness and regulatory changes affecting dermal toxicity testing
Bottom-up Modeling
- Collection of data on the number of testing facilities and their operational capacities in Kuwait
- Estimation of service pricing based on laboratory service rates and testing volumes
- Calculation of market size based on the volume of tests conducted and average pricing per test
Forecasting & Scenario Analysis
- Development of predictive models using historical data on dermatological product launches and testing requirements
- Scenario analysis based on potential regulatory changes and advancements in testing technologies
- Creation of baseline, optimistic, and pessimistic forecasts for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Industry Testing | 45 | Regulatory Affairs Managers, Quality Control Specialists |
| Cosmetic Product Safety Testing | 38 | Product Development Managers, Safety Assessors |
| Academic Research Institutions | 32 | Research Scientists, Laboratory Directors |
| Government Regulatory Bodies | 28 | Policy Makers, Compliance Officers |
| Contract Research Organizations (CROs) | 35 | Project Managers, Clinical Research Coordinators |
Frequently Asked Questions
What is the current value of the Kuwait Dermal Toxicity Testing Market?
The Kuwait Dermal Toxicity Testing Market is valued at approximately USD 19 million, reflecting a significant growth driven by increasing regulatory requirements and consumer awareness regarding product safety in the pharmaceutical and cosmetic industries.