About the Report
Base Year 2024Listen to the audio summary
New Zealand Dermal Toxicity Testing Market Overview
- The New Zealand Dermal Toxicity Testing Market is valued at USD 20 million, based on a five-year historical analysis. This growth is primarily driven by increasing regulatory requirements for safety testing in the pharmaceutical and cosmetic industries, alongside rising consumer awareness regarding product safety and efficacy, and a shift towards alternative in vitro methods due to ethical considerations and technological advancements in toxicity testing. The demand for reliable toxicity testing methods has surged, leading to advancements in testing technologies and methodologies.
- Auckland and Wellington are the dominant cities in the New Zealand Dermal Toxicity Testing Market due to their concentration of pharmaceutical and cosmetic companies, as well as research institutions. These cities benefit from a robust infrastructure, skilled workforce, and strong government support for research and development, making them attractive hubs for innovation in dermal toxicity testing.
- The Hazardous Substances and New Organisms Act 1996, issued by the New Zealand Environmental Protection Authority (EPA), mandates comprehensive safety assessments for hazardous substances including those applied dermally, requiring approvals with defined exposure thresholds, classification for skin corrosion or irritation, and compliance testing standards prior to market entry. This regulation emphasizes the importance of dermal toxicity testing, ensuring that products are thoroughly evaluated for potential skin-related adverse effects, thereby enhancing consumer safety and confidence.
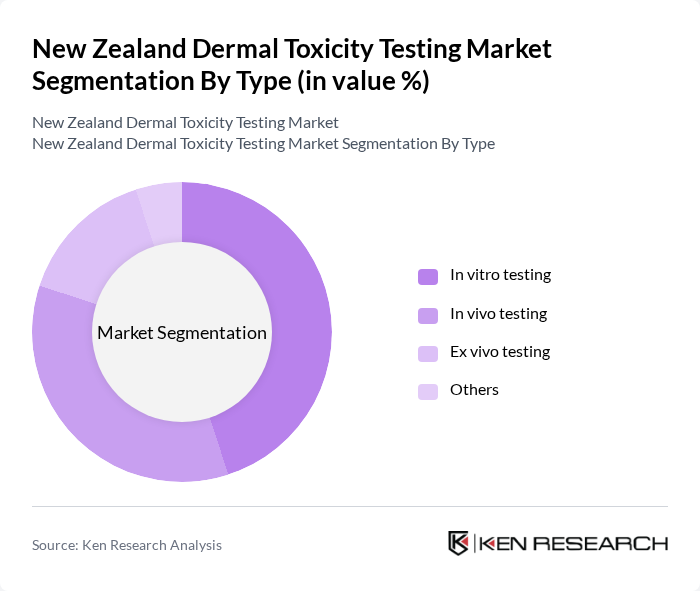
New Zealand Dermal Toxicity Testing Market Segmentation
By Type:The market is segmented into various testing types, including in vitro testing, in vivo testing, ex vivo testing, and others. In vitro testing is gaining traction due to its ethical advantages and cost-effectiveness, while in vivo testing remains essential for comprehensive safety assessments. The ex vivo testing segment is also emerging, providing valuable insights into skin reactions without the need for live subjects. The "Others" category includes alternative methods that are being explored for their potential in toxicity testing.
By End-User:The end-user segmentation includes pharmaceutical companies, cosmetic manufacturers, research institutions, regulatory bodies, and others. Pharmaceutical companies are the largest segment, driven by stringent regulatory requirements for drug safety. Cosmetic manufacturers are also significant users, focusing on consumer safety and product efficacy. Research institutions play a crucial role in developing new testing methodologies, while regulatory bodies ensure compliance with safety standards. The "Others" category encompasses various smaller entities involved in toxicity testing.

New Zealand Dermal Toxicity Testing Market Competitive Landscape
The New Zealand Dermal Toxicity Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Labcorp, Charles River Laboratories, Eurofins Scientific, Covance, SGS SA, Intertek Group, Q2 Solutions, BioReliance, Medpace, Toxikon, Wuxi AppTec, BSL Bioservice, Envigo, Asterand Bioscience, AccuBioTech contribute to innovation, geographic expansion, and service delivery in this space.
New Zealand Dermal Toxicity Testing Market Industry Analysis
Growth Drivers
- Increasing Regulatory Requirements for Safety Testing:The New Zealand government has implemented stringent regulations, including the Health and Safety at Work Act, which mandates comprehensive safety testing for chemicals and cosmetics. In future, the regulatory framework is expected to enforce over 200 compliance checks annually, driving demand for dermal toxicity testing services. This regulatory environment ensures that companies prioritize safety, leading to an estimated increase in testing demand by 15% year-on-year, reflecting a commitment to consumer protection and product safety.
- Rising Consumer Awareness Regarding Product Safety:Consumer awareness regarding the safety of cosmetic and personal care products has surged, with 78% of New Zealanders expressing concern over product ingredients in recent surveys. This heightened awareness is prompting companies to invest in dermal toxicity testing to ensure compliance with consumer expectations. As a result, the demand for reliable testing services is projected to grow, with an estimated increase of 20% in inquiries for safety testing in future, reflecting a shift towards transparency and accountability in product formulation.
- Growth in the Cosmetics and Personal Care Industry:The New Zealand cosmetics market is projected to reach NZD 1.3 billion in future, driven by a growing population and increasing disposable income. This growth is accompanied by a rising demand for innovative and safe cosmetic products, necessitating rigorous dermal toxicity testing. As brands expand their product lines, the need for comprehensive safety assessments is expected to rise, leading to a 25% increase in testing services required by cosmetic manufacturers in future, highlighting the industry's commitment to safety and efficacy.
Market Challenges
- High Costs Associated with Dermal Toxicity Testing:The financial burden of dermal toxicity testing can be significant, with costs ranging from NZD 5,000 to NZD 55,000 per test, depending on the complexity and required methodologies. This high cost can deter smaller companies from conducting necessary testing, potentially leading to non-compliance with safety regulations. In future, it is estimated that 30% of small to medium enterprises may forgo testing due to budget constraints, posing a challenge to overall market growth and safety standards.
- Limited Availability of Skilled Professionals:The dermal toxicity testing sector faces a shortage of qualified professionals, with only 200 certified toxicologists available in New Zealand as of 2023. This scarcity limits the capacity of testing laboratories to meet increasing demand, potentially leading to longer turnaround times and reduced service quality. In future, it is projected that the industry will require an additional 50 toxicologists to keep pace with market needs, highlighting the urgent need for educational initiatives and workforce development in this specialized field.
New Zealand Dermal Toxicity Testing Market Future Outlook
The future of the New Zealand dermal toxicity testing market appears promising, driven by advancements in testing technologies and a growing emphasis on consumer safety. As regulatory frameworks evolve, companies are increasingly adopting in vitro testing methods, which are not only cost-effective but also align with the rising demand for cruelty-free testing. Furthermore, the integration of artificial intelligence in testing processes is expected to enhance efficiency and accuracy, paving the way for innovative solutions that meet both regulatory and consumer expectations in future.
Market Opportunities
- Expansion of the Pharmaceutical Sector:The pharmaceutical industry in New Zealand is projected to grow to NZD 1.6 billion by future, creating significant opportunities for dermal toxicity testing services. As pharmaceutical companies develop new drugs, the demand for comprehensive safety assessments will increase, providing a lucrative market for testing laboratories to expand their services and capabilities.
- Increasing Demand for Cruelty-Free Testing Methods:With 65% of consumers in New Zealand preferring cruelty-free products, there is a growing market for alternative testing methods that do not involve animal testing. This trend presents an opportunity for testing facilities to innovate and offer cruelty-free testing solutions, aligning with consumer preferences and enhancing brand reputation in a competitive market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | In vitro testing In vivo testing Ex vivo testing Others |
| By End-User | Pharmaceutical companies Cosmetic manufacturers Research institutions Regulatory bodies Others |
| By Application | Skin irritation testing Sensitization testing Toxicokinetics Others |
| By Technology | D skin models Organ-on-a-chip technology High-throughput screening Others |
| By Industry Vertical | Healthcare Cosmetics Chemicals Others |
| By Region | North Island South Island |
| By Policy Support | Government grants for research Tax incentives for R&D Regulatory support for innovation Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Environmental Protection Authority, Ministry of Health)
Pharmaceutical and Cosmetic Manufacturers
Biotechnology Companies
Contract Research Organizations (CROs)
Dermatological Product Developers
Health and Safety Regulatory Agencies
Insurance Companies and Risk Assessment Firms
Players Mentioned in the Report:
Labcorp
Charles River Laboratories
Eurofins Scientific
Covance
SGS SA
Intertek Group
Q2 Solutions
BioReliance
Medpace
Toxikon
Wuxi AppTec
BSL Bioservice
Envigo
Asterand Bioscience
AccuBioTech
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. New Zealand Dermal Toxicity Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 New Zealand Dermal Toxicity Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. New Zealand Dermal Toxicity Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing regulatory requirements for safety testing
3.1.2 Rising consumer awareness regarding product safety
3.1.3 Growth in the cosmetics and personal care industry
3.1.4 Advancements in testing technologies
3.2 Market Challenges
3.2.1 High costs associated with dermal toxicity testing
3.2.2 Limited availability of skilled professionals
3.2.3 Stringent regulatory frameworks
3.2.4 Competition from alternative testing methods
3.3 Market Opportunities
3.3.1 Expansion of the pharmaceutical sector
3.3.2 Increasing demand for cruelty-free testing methods
3.3.3 Collaborations with research institutions
3.3.4 Growth in e-commerce for cosmetic products
3.4 Market Trends
3.4.1 Shift towards in vitro testing methods
3.4.2 Integration of AI and machine learning in testing
3.4.3 Focus on sustainable and eco-friendly products
3.4.4 Rising investment in R&D for innovative testing solutions
3.5 Government Regulation
3.5.1 New Zealand's Health and Safety at Work Act
3.5.2 Cosmetic Products Group Standard
3.5.3 Environmental Protection Authority guidelines
3.5.4 Animal Welfare Act compliance requirements
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. New Zealand Dermal Toxicity Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. New Zealand Dermal Toxicity Testing Market Segmentation
8.1 By Type
8.1.1 In vitro testing
8.1.2 In vivo testing
8.1.3 Ex vivo testing
8.1.4 Others
8.2 By End-User
8.2.1 Pharmaceutical companies
8.2.2 Cosmetic manufacturers
8.2.3 Research institutions
8.2.4 Regulatory bodies
8.2.5 Others
8.3 By Application
8.3.1 Skin irritation testing
8.3.2 Sensitization testing
8.3.3 Toxicokinetics
8.3.4 Others
8.4 By Technology
8.4.1 3D skin models
8.4.2 Organ-on-a-chip technology
8.4.3 High-throughput screening
8.4.4 Others
8.5 By Industry Vertical
8.5.1 Healthcare
8.5.2 Cosmetics
8.5.3 Chemicals
8.5.4 Others
8.6 By Region
8.6.1 North Island
8.6.2 South Island
8.7 By Policy Support
8.7.1 Government grants for research
8.7.2 Tax incentives for R&D
8.7.3 Regulatory support for innovation
8.7.4 Others
9. New Zealand Dermal Toxicity Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Development Cycle Time
9.2.8 Compliance Rate with Regulatory Standards
9.2.9 Customer Satisfaction Score
9.2.10 Innovation Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Labcorp
9.5.2 Charles River Laboratories
9.5.3 Eurofins Scientific
9.5.4 Covance
9.5.5 SGS SA
9.5.6 Intertek Group
9.5.7 Q2 Solutions
9.5.8 BioReliance
9.5.9 Medpace
9.5.10 Toxikon
9.5.11 Wuxi AppTec
9.5.12 BSL Bioservice
9.5.13 Envigo
9.5.14 Asterand Bioscience
9.5.15 AccuBioTech
10. New Zealand Dermal Toxicity Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget allocation for safety testing
10.1.2 Decision-making processes
10.1.3 Preferred suppliers and partnerships
10.1.4 Compliance with national standards
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in testing facilities
10.2.2 Funding for R&D initiatives
10.2.3 Expenditure on technology upgrades
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Regulatory compliance challenges
10.3.2 Cost management issues
10.3.3 Access to advanced testing methods
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of testing benefits
10.4.2 Training and skill development needs
10.4.3 Infrastructure readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of testing effectiveness
10.5.2 Expansion into new product lines
10.5.3 Long-term cost savings
10.5.4 Others
11. New Zealand Dermal Toxicity Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
1.6 Key partnerships identification
1.7 Operational framework design
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing tactics
2.6 Event marketing opportunities
2.7 Feedback mechanisms
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Direct sales approaches
3.5 Partnership with distributors
3.6 Logistics and supply chain management
3.7 Inventory management strategies
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing comparison
4.4 Value-based pricing strategies
4.5 Discount and promotion strategies
4.6 Customer feedback on pricing
4.7 Price elasticity analysis
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Emerging trends exploration
5.4 Product development opportunities
5.5 Market entry barriers
5.6 Customer pain points
5.7 Future demand forecasting
6. Customer Relationship
6.1 Loyalty programs design
6.2 After-sales service strategies
6.3 Customer engagement initiatives
6.4 Feedback collection methods
6.5 Relationship management tools
6.6 Community building efforts
6.7 Customer education programs
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Customer-centric approach
7.5 Competitive advantages
7.6 Value delivery mechanisms
7.7 Brand positioning strategies
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Marketing campaigns
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries identification
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
10.6 Strategic fit evaluation
10.7 Long-term sustainability considerations
11. Capital and Timeline Estimation
11.1 Capital requirements analysis
11.2 Timelines for market entry
11.3 Funding sources exploration
11.4 Financial projections
11.5 Budget allocation strategies
11.6 Cost management plans
11.7 Risk mitigation strategies
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk management frameworks
12.4 Control mechanisms
12.5 Strategic alignment
12.6 Long-term vision
12.7 Stakeholder interests
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability strategies
13.3 Profit margin optimization
13.4 Revenue diversification
13.5 Cost reduction initiatives
13.6 Financial health monitoring
13.7 Investment return analysis
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Research collaborations
14.6 Technology partners
14.7 Supply chain partners
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for key activities
15.2.2 Milestone tracking
15.2.3 Performance evaluation
15.2.4 Stakeholder updates
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from New Zealand's Environmental Protection Authority (EPA)
- Review of scientific literature on dermal toxicity testing methodologies and regulations
- Examination of market trends and forecasts from industry publications and journals
Primary Research
- Interviews with dermatologists and toxicologists specializing in dermal toxicity
- Surveys with regulatory bodies and compliance officers in the cosmetics and chemical sectors
- Field interviews with R&D managers at leading dermal testing laboratories
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and focus groups
- Triangulation of data from regulatory updates, market reports, and expert opinions
- Sanity checks through peer reviews and feedback from industry stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national health and safety regulations impacting dermal testing
- Analysis of the growth of the cosmetics and personal care industry in New Zealand
- Incorporation of government initiatives promoting safe testing practices
Bottom-up Modeling
- Collection of data on testing volumes from leading dermal toxicity testing facilities
- Operational cost analysis based on service pricing and testing methodologies
- Volume x cost calculations for various testing services offered in the market
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors like regulatory changes and consumer safety awareness
- Scenario modeling based on potential shifts in market demand and technological advancements
- Baseline, optimistic, and pessimistic projections for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cosmetics Industry Testing | 100 | Product Safety Managers, Regulatory Affairs Specialists |
| Pharmaceutical Dermal Testing | 80 | Clinical Research Coordinators, Toxicology Experts |
| Consumer Product Safety Testing | 70 | Quality Assurance Managers, Compliance Officers |
| Academic Research Institutions | 60 | Research Scientists, University Professors |
| Regulatory Bodies | 50 | Policy Makers, Environmental Health Officials |
Frequently Asked Questions
What is the current value of the New Zealand Dermal Toxicity Testing Market?
The New Zealand Dermal Toxicity Testing Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This growth is driven by increasing regulatory requirements and consumer awareness regarding product safety in the pharmaceutical and cosmetic industries.