Region:Middle East
Author(s):Geetanshi
Product Code:KRAC4429
Pages:96
Published On:October 2025
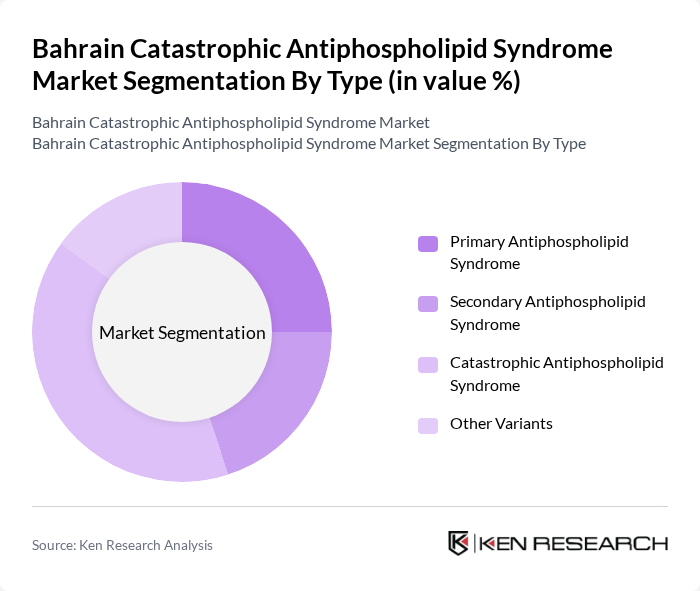
By Type:The market is segmented into various types, including Primary Antiphospholipid Syndrome, Secondary Antiphospholipid Syndrome, Catastrophic Antiphospholipid Syndrome, and Other Variants. Among these, Catastrophic Antiphospholipid Syndrome is the most critical due to its severe implications and the urgent need for effective treatment. The increasing awareness of this life-threatening condition has led to a rise in demand for specialized care and treatment options, making it a focal point in the market. Recent advances in diagnostic and therapeutic approaches have further differentiated catastrophic APS as a distinct clinical priority .
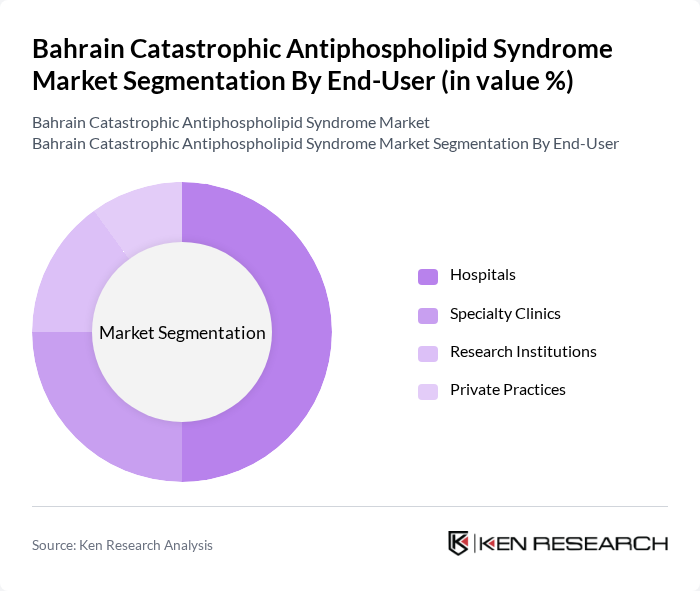
By End-User:The market is categorized by end-users, including Hospitals, Specialty Clinics, Research Institutions, and Private Practices. Hospitals are the leading end-user segment, primarily due to their comprehensive facilities and access to advanced treatment options. The increasing number of patients requiring hospitalization for severe cases of catastrophic antiphospholipid syndrome drives this segment's growth, as hospitals are equipped to provide specialized care and emergency services. This mirrors global market trends, where hospitals account for over 60% of catastrophic APS treatments .
The Bahrain Catastrophic Antiphospholipid Syndrome Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Sanofi S.A., Bayer AG, Bristol Myers Squibb Company, AbbVie Inc., Novartis AG, GlaxoSmithKline plc, Merck & Co., Inc., AstraZeneca plc, and Eli Lilly and Company contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain Catastrophic Antiphospholipid Syndrome market appears promising, driven by ongoing advancements in medical technology and increased healthcare funding. As the government continues to prioritize healthcare improvements, the integration of telemedicine and personalized medicine approaches is expected to enhance patient management in future. Furthermore, collaborations with international research organizations will likely foster innovation, leading to the development of new therapeutic agents and improved patient outcomes in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Primary Antiphospholipid Syndrome Secondary Antiphospholipid Syndrome Catastrophic Antiphospholipid Syndrome Other Variants |
| By End-User | Hospitals Specialty Clinics Research Institutions Private Practices |
| By Treatment Type | Anticoagulants Corticosteroids Immunosuppressants Therapeutic Plasmapheresis Intravenous Immunoglobulin (IVIG) |
| By Distribution Channel | Direct Sales Online Pharmacies Retail Pharmacies Hospital Pharmacies |
| By Patient Demographics | Adults Children Elderly Pregnant Women |
| By Geographic Distribution | Urban Areas Rural Areas Coastal Regions |
| By Research and Development Focus | Clinical Trials Drug Development Patient Registries Basic Research |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers in APS Treatment | 100 | Hematologists, Rheumatologists |
| Patient Experience with Catastrophic APS | 70 | Patients diagnosed with APS, Caregivers |
| Pharmaceutical Stakeholders | 45 | Pharmaceutical Representatives, Medical Affairs Managers |
| Health Policy Makers | 40 | Government Health Officials, Policy Analysts |
| Insurance Providers | 40 | Health Insurance Underwriters, Claims Adjusters |
The Bahrain Catastrophic Antiphospholipid Syndrome market is valued at approximately USD 30 million, reflecting a five-year historical analysis that indicates growth driven by the rising prevalence of autoimmune diseases and advancements in diagnostic technologies.