Region:Middle East
Author(s):Dev
Product Code:KRAC4737
Pages:81
Published On:October 2025
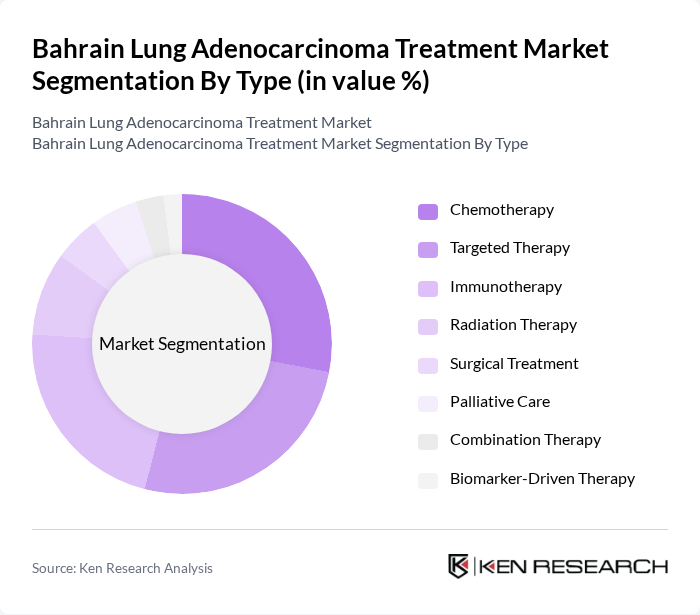
By Type:The treatment market is segmented into Chemotherapy, Targeted Therapy, Immunotherapy, Radiation Therapy, Surgical Treatment, Palliative Care, Combination Therapy, and Biomarker-Driven Therapy. Chemotherapy and targeted therapy remain foundational for advanced-stage patients, while immunotherapy is increasingly adopted for its efficacy in improving survival rates. Radiation therapy and surgical treatment are typically reserved for localized disease, and biomarker-driven therapy is expanding due to the growing availability of molecular diagnostics. Palliative care and combination therapy support patient quality of life and optimize outcomes in complex cases.
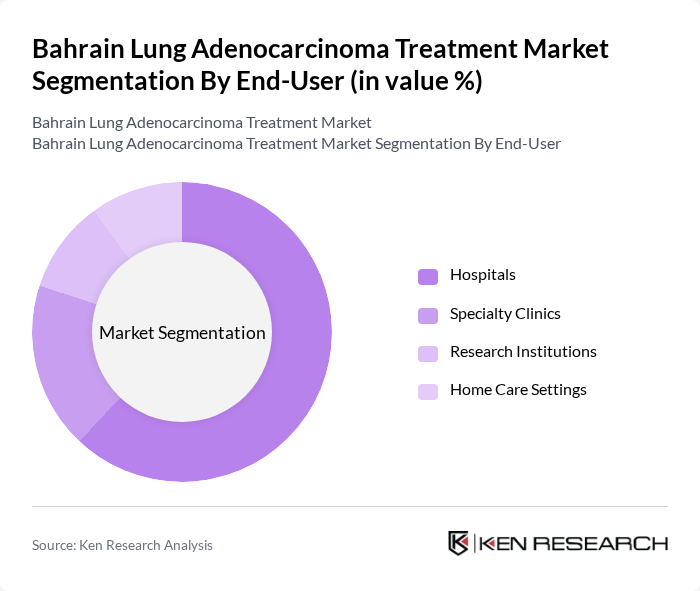
By End-User:The market is segmented by end-users: Hospitals, Specialty Clinics, Research Institutions, and Home Care Settings. Hospitals account for the largest share, driven by their comprehensive oncology services and access to multidisciplinary teams. Specialty clinics and research institutions are increasingly involved in clinical trials and advanced therapies, while home care settings are expanding for palliative and supportive care.
The Bahrain Lung Adenocarcinoma Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Roche Holding AG, AstraZeneca PLC, Merck & Co., Inc., Novartis AG, Bristol-Myers Squibb Company, Eli Lilly and Company, Johnson & Johnson (Janssen Pharmaceuticals), Amgen Inc., GSK plc (GlaxoSmithKline plc), Sanofi S.A., Takeda Pharmaceutical Company Limited, Bayer AG, AbbVie Inc., Astellas Pharma Inc., Boehringer Ingelheim GmbH, Hikma Pharmaceuticals PLC, Gulf Pharmaceutical Industries (Julphar), Mundipharma International Limited, National Health Regulatory Authority (NHRA) Bahrain (as key regulator) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the lung adenocarcinoma treatment market in Bahrain appears promising, driven by ongoing advancements in medical technology and increased healthcare investments. The government’s commitment to enhancing healthcare infrastructure is expected to facilitate better access to innovative treatments. Additionally, the integration of telemedicine and AI in diagnostics is likely to improve early detection rates, ultimately leading to better patient outcomes and a more robust treatment landscape in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Chemotherapy Targeted Therapy Immunotherapy Radiation Therapy Surgical Treatment Palliative Care Combination Therapy Biomarker-Driven Therapy |
| By End-User | Hospitals Specialty Clinics Research Institutions Home Care Settings |
| By Treatment Stage | Early Stage Locally Advanced Stage Metastatic Stage |
| By Patient Demographics | Age Group (Adults, Elderly) Gender (Male, Female) |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Geographic Distribution | Urban Areas Rural Areas |
| By Insurance Coverage | Private Insurance Public Insurance Out-of-Pocket |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologist Insights | 50 | Medical Oncologists, Radiation Oncologists |
| Patient Experience Surveys | 100 | Lung Adenocarcinoma Patients, Caregivers |
| Hospital Administration Feedback | 50 | Hospital Administrators, Oncology Department Heads |
| Pharmaceutical Insights | 40 | Pharmaceutical Representatives, Medical Science Liaisons |
| Health Insurance Perspectives | 40 | Health Insurance Analysts, Policy Makers |
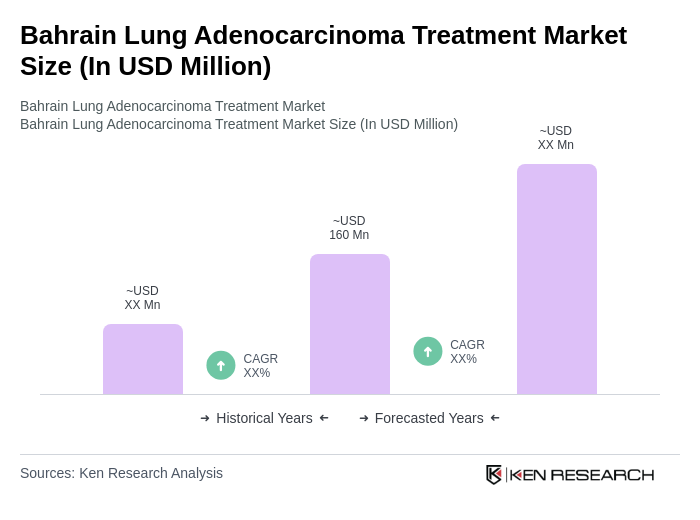
The Bahrain Lung Adenocarcinoma Treatment Market is valued at approximately USD 160 million, reflecting a significant investment in healthcare and advancements in treatment options, driven by the rising prevalence of lung cancer and the adoption of innovative therapies.