Region:Middle East
Author(s):Dev
Product Code:KRAC3484
Pages:96
Published On:October 2025
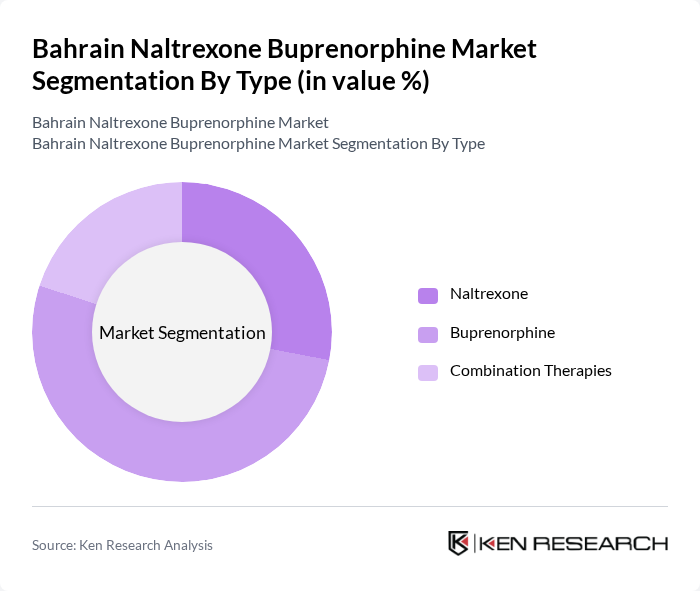
By Type:The market is segmented into Naltrexone, Buprenorphine, and Combination Therapies. Among these, Buprenorphine remains the leading sub-segment due to its proven clinical effectiveness in managing withdrawal symptoms and cravings associated with opioid dependence. The increasing preference for Buprenorphine in treatment protocols is driven by its favorable safety profile, lower risk of misuse compared to full agonists, and the growing acceptance of medication-assisted treatment among healthcare providers .
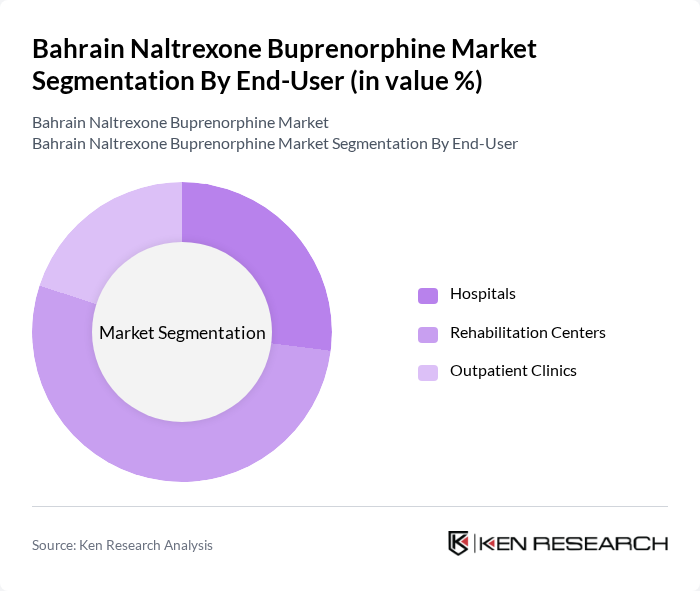
By End-User:The market is categorized into Hospitals, Rehabilitation Centers, and Outpatient Clinics. Rehabilitation Centers dominate this segment, as they are specifically designed to provide comprehensive treatment and support for individuals battling addiction. The increasing number of specialized facilities, the expansion of private rehabilitation services, and the growing recognition of the importance of structured recovery programs contribute to the prominence of this sub-segment .
The Bahrain Naltrexone Buprenorphine Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Indivior PLC, Alkermes PLC, Teva Pharmaceutical Industries Ltd., Viatris Inc. (formerly Mylan N.V.), Hikma Pharmaceuticals PLC, Sandoz International GmbH (Novartis), Aurobindo Pharma Ltd., Sun Pharmaceutical Industries Ltd., Mallinckrodt Pharmaceuticals, Lannett Company, Inc., Amgen Inc., GSK (GlaxoSmithKline) PLC, Novartis AG, Merck & Co., Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain Naltrexone Buprenorphine market appears promising, driven by ongoing government support and increasing public awareness of addiction treatment. As healthcare systems evolve, the integration of telemedicine and digital health solutions is expected to enhance access to treatment. Furthermore, the development of new drug formulations tailored to individual needs will likely improve treatment outcomes, fostering a more robust market environment for addiction therapies in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Naltrexone Buprenorphine Combination Therapies |
| By End-User | Hospitals Rehabilitation Centers Outpatient Clinics |
| By Treatment Setting | Inpatient Treatment Outpatient Treatment |
| By Distribution Channel | Direct Sales (to hospitals/clinics) Retail Pharmacies Online Pharmacies |
| By Patient Demographics | Adults Adolescents |
| By Treatment Duration | Short-term Treatment Long-term Treatment |
| By Others | Experimental Treatments Support Services (e.g., counseling, follow-up) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Doctors, Addiction Specialists, Pharmacists |
| Patients Undergoing Treatment | 80 | Individuals in Opioid Recovery Programs |
| Pharmaceutical Distributors | 40 | Supply Chain Managers, Sales Representatives |
| Policy Makers | 40 | Government Health Officials, Regulatory Authorities |
| Healthcare Administrators | 40 | Hospital Managers, Clinic Directors |
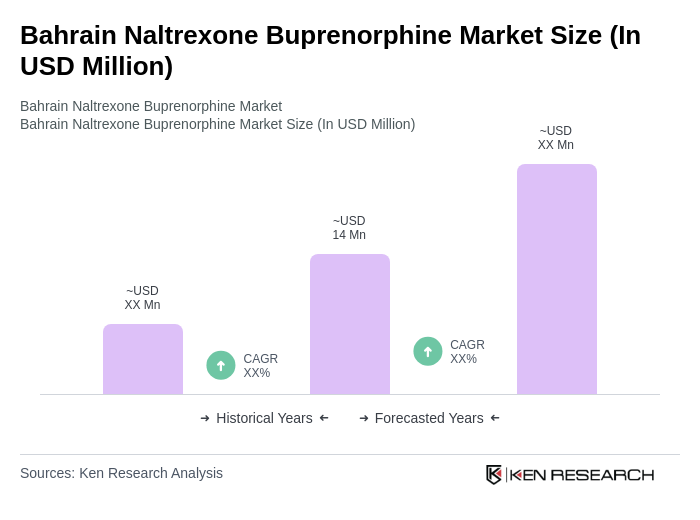
The Bahrain Naltrexone Buprenorphine Market is valued at approximately USD 14 million, reflecting a growing demand for opioid dependence treatments driven by increased awareness and the prevalence of substance use disorders in the region.