Region:Middle East
Author(s):Rebecca
Product Code:KRAD5026
Pages:86
Published On:December 2025
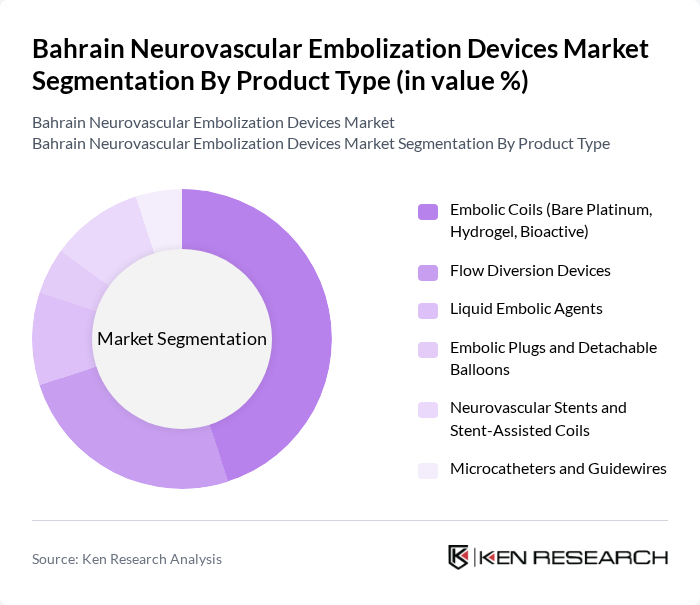
By Product Type:The product type segmentation includes various categories of neurovascular embolization devices that cater to different medical needs. The dominant sub-segment in this category is Embolic Coils, which are widely used for treating intracranial aneurysms and arteriovenous malformations due to their proven safety profile, effectiveness, and minimal invasiveness in endovascular procedures. Flow Diversion Devices and Liquid Embolic Agents are also gaining traction as they offer innovative solutions for complex, wide-neck, or giant aneurysms and other challenging vascular malformations where traditional coiling alone may be suboptimal. The increasing adoption of these devices in hospitals and specialized centers, supported by growing neurointerventional capability across leading tertiary centers in the Gulf region, is driving their market share.
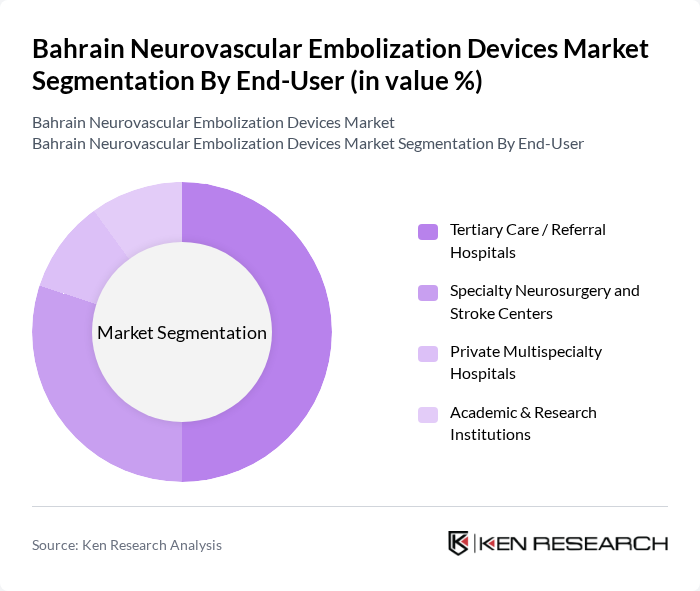
By End-User:The end-user segmentation highlights the various healthcare facilities utilizing neurovascular embolization devices. Tertiary Care/Referral Hospitals lead this segment due to their advanced imaging capabilities (including CT, MRI, and biplane angiography), availability of hybrid catheterization laboratories, and multidisciplinary stroke and neurosurgery teams required to perform complex endovascular procedures. Specialty Neurosurgery and Stroke Centers are also significant users, as they focus on treating complex neurovascular conditions and are often designated as referral hubs for acute ischemic and hemorrhagic stroke management in Bahrain and neighboring countries. The increasing number of private multispecialty hospitals and academic institutions investing in advanced neuroimaging, interventional radiology, and neurovascular programs further supports the growth of this market segment in Bahrain, in line with broader Middle East trends toward expanding high-acuity specialty care.
The Bahrain Neurovascular Embolization Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Stryker Corporation, Cerenovus (Johnson & Johnson MedTech), Boston Scientific Corporation, Terumo Corporation, Penumbra, Inc., MicroVention, Inc. (Terumo Group), Balt Extrusion SAS, Phenox GmbH, B. Braun Melsungen AG, Integra LifeSciences, Merit Medical Systems, Inc., Asahi Intecc Co., Ltd., Cook Medical LLC, Abbott Laboratories contribute to innovation, geographic expansion, and service delivery in this space, reflecting the broader global competitive landscape for neurovascular embolization and coiling devices.
The future of the Bahrain neurovascular embolization devices market appears promising, driven by ongoing advancements in medical technology and increasing healthcare investments. As the government continues to enhance healthcare infrastructure, more facilities are expected to adopt innovative devices. Additionally, the growing trend towards minimally invasive procedures will likely encourage the development of new products, improving patient outcomes and expanding market reach. The integration of artificial intelligence in medical devices may further revolutionize treatment approaches, enhancing diagnostic accuracy and procedural efficiency.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Embolic Coils (Bare Platinum, Hydrogel, Bioactive) Flow Diversion Devices Liquid Embolic Agents Embolic Plugs and Detachable Balloons Neurovascular Stents and Stent-Assisted Coils Microcatheters and Guidewires |
| By End-User | Tertiary Care / Referral Hospitals Specialty Neurosurgery and Stroke Centers Private Multispecialty Hospitals Academic & Research Institutions |
| By Clinical Application | Intracranial Aneurysm Coiling and Flow Diversion Arteriovenous Malformations (AVMs) and Fistulas Ischemic Stroke and Carotid / Intracranial Stenosis Tumor and Dural Arteriovenous Fistula Embolization Others (Traumatic and Iatrogenic Vascular Lesions) |
| By Distribution Channel | Direct Tender / Institutional Sales Local Distributors and Agents Group Purchasing & Regional Procurement Platforms |
| By Governorate | Capital Governorate Northern Governorate Southern Governorate Muharraq Governorate |
| By Patient Demographics | Age Group (Pediatric, Adult, Geriatric) Gender (Male, Female) Nationality (Bahraini, Expatriate) |
| By Procedure Setting & Technology | Inpatient Angiography Suite Procedures Hybrid OR / Advanced Neurointerventional Suites Use of 3D Rotational Angiography and Advanced Imaging |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 60 | Neurosurgeons, Interventional Radiologists |
| Hospital Procurement Teams | 40 | Procurement Managers, Supply Chain Coordinators |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
| Medical Device Distributors | 50 | Sales Managers, Product Specialists |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
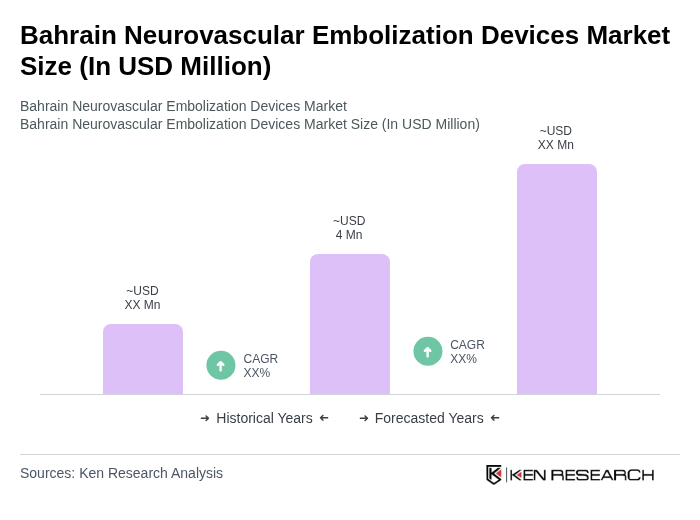
The Bahrain Neurovascular Embolization Devices Market is valued at approximately USD 4 million, reflecting a small share of the global market, influenced by the country's population size and healthcare expenditure trends in the Middle East.