About the Report
Base Year 2024France Cold Chain for Pharma & Vaccine Distribution Market Overview
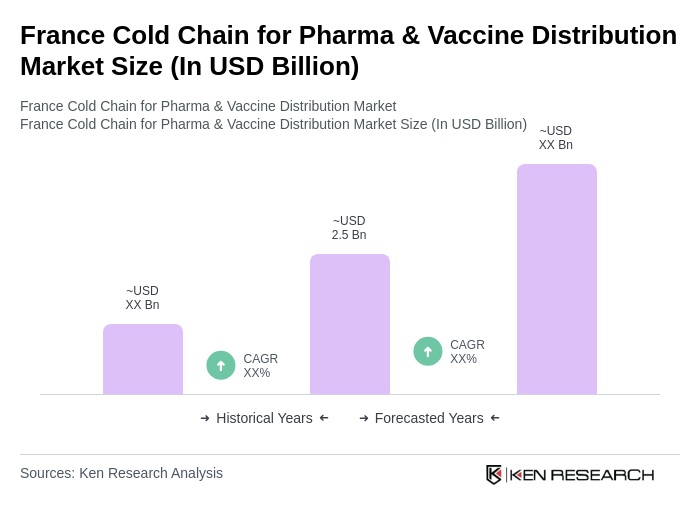
- The France Cold Chain for Pharma & Vaccine Distribution Market is valued at USD 2.5 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for temperature-sensitive pharmaceuticals and vaccines, particularly in the wake of the COVID-19 pandemic, which has heightened awareness of the importance of effective cold chain logistics in healthcare.
- Key cities such as Paris, Lyon, and Marseille dominate the market due to their strategic locations, advanced infrastructure, and proximity to major healthcare facilities and pharmaceutical manufacturers. These urban centers serve as critical hubs for distribution, ensuring timely delivery of essential medical products across the country.
- In 2023, the French government implemented stringent regulations mandating that all pharmaceutical products requiring refrigeration must be transported and stored within specified temperature ranges. This regulation aims to enhance the safety and efficacy of medications and vaccines, thereby reinforcing the importance of compliance within the cold chain logistics sector.
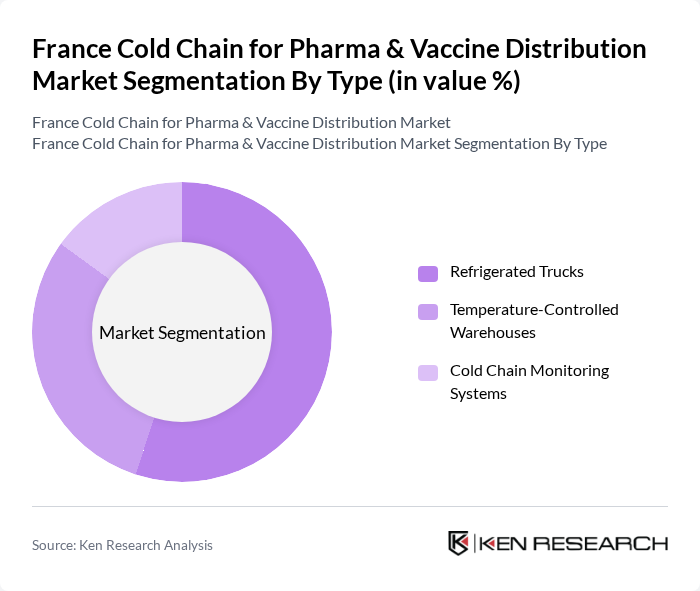
France Cold Chain for Pharma & Vaccine Distribution Market Segmentation
By Type:The cold chain logistics market is segmented into three main types: Refrigerated Trucks, Temperature-Controlled Warehouses, and Cold Chain Monitoring Systems. Among these, Refrigerated Trucks are the most dominant segment due to their essential role in the transportation of temperature-sensitive products. The increasing demand for efficient and reliable transportation solutions has led to significant investments in refrigerated vehicle fleets, ensuring compliance with stringent temperature regulations.
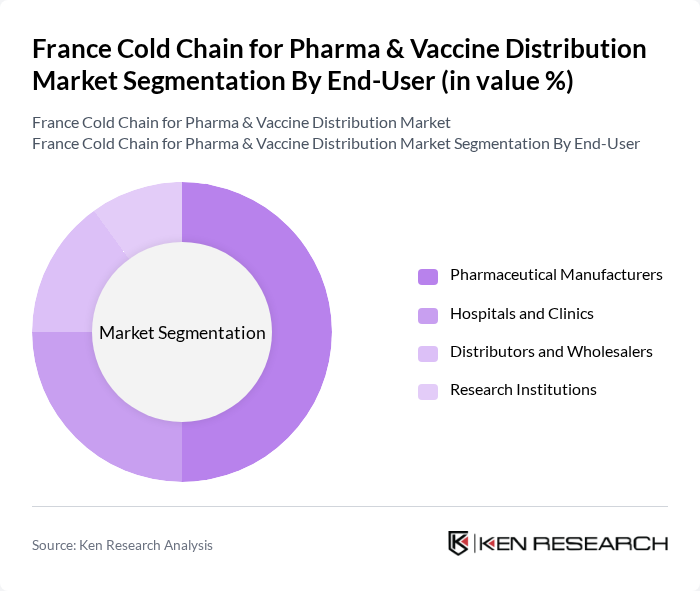
By End-User:The end-user segmentation includes Pharmaceutical Manufacturers, Hospitals and Clinics, Distributors and Wholesalers, and Research Institutions. Pharmaceutical Manufacturers represent the largest segment, driven by the need for reliable cold chain solutions to maintain the integrity of their products during distribution. The growing focus on biopharmaceuticals and personalized medicine further fuels the demand for specialized cold chain logistics tailored to the unique requirements of these products.
France Cold Chain for Pharma & Vaccine Distribution Market Competitive Landscape
The France Cold Chain for Pharma & Vaccine Distribution Market is characterized by a dynamic mix of regional and international players. Leading participants such as DHL Supply Chain, Kuehne + Nagel, DB Schenker, UPS Healthcare, FedEx, Geodis, XPO Logistics, Cardinal Health, AmerisourceBergen, Thermo Fisher Scientific, Lineage Logistics, Maersk, Panalpina, Agility Logistics, CEVA Logistics contribute to innovation, geographic expansion, and service delivery in this space.
France Cold Chain for Pharma & Vaccine Distribution Market Industry Analysis
Growth Drivers
- Increasing Demand for Biologics:The French pharmaceutical market is projected to reach €50 billion by 2024, driven by a surge in biologics, which accounted for 30% of total drug sales in 2023. This growth is fueled by the rising prevalence of chronic diseases, with over 22 million patients in France requiring biologic therapies. Consequently, the demand for efficient cold chain logistics to maintain the integrity of these temperature-sensitive products is paramount, necessitating advanced cold chain solutions.
- Expansion of Vaccine Distribution Networks:France's vaccination efforts have intensified, with over 90 million vaccine doses administered in 2023 alone. The government aims to enhance vaccine distribution networks, particularly for mRNA vaccines, which require strict temperature controls. This expansion is supported by a €1.5 billion investment in healthcare logistics, emphasizing the need for robust cold chain systems to ensure timely and safe delivery of vaccines across the nation.
- Technological Advancements in Cold Chain Logistics:The cold chain logistics sector in France is witnessing significant technological advancements, with an estimated €600 million allocated for innovation in 2024. The adoption of IoT devices and real-time monitoring systems is expected to enhance temperature control and traceability. These technologies are crucial for maintaining compliance with stringent regulations, as they help mitigate risks associated with temperature excursions, thereby ensuring product safety and efficacy.
Market Challenges
- High Operational Costs:The operational costs associated with cold chain logistics in France are projected to exceed €2.2 billion in 2024. Factors contributing to these high costs include energy expenses, maintenance of specialized equipment, and compliance with regulatory standards. These financial burdens can hinder smaller logistics providers from competing effectively, potentially leading to a consolidation of services within the industry, which may limit options for pharmaceutical companies.
- Infrastructure Limitations:Despite advancements, France's cold chain infrastructure still faces significant limitations, particularly in rural areas. Approximately 32% of the population resides in regions with inadequate cold storage facilities, impacting the distribution of temperature-sensitive pharmaceuticals. The lack of investment in infrastructure development, estimated at €1.2 billion needed in future, poses a challenge to ensuring consistent and reliable cold chain services across the country.
France Cold Chain for Pharma & Vaccine Distribution Market Future Outlook
The future of the cold chain logistics market in France is poised for transformation, driven by technological innovations and regulatory advancements. As the demand for biologics and vaccines continues to rise, the industry will likely see increased investments in smart technologies and infrastructure improvements. Additionally, the integration of AI in supply chain management will enhance operational efficiency, while sustainability initiatives will shape practices, ensuring compliance with environmental regulations and meeting consumer expectations for greener solutions.
Market Opportunities
- Growth in E-commerce for Pharmaceuticals:The e-commerce sector for pharmaceuticals in France is expected to grow to €6 billion by 2024, driven by consumer demand for online access to medications. This trend presents a significant opportunity for cold chain logistics providers to develop specialized solutions that ensure the safe transport of temperature-sensitive products directly to consumers' homes, enhancing service delivery and customer satisfaction.
- Increasing Investment in Cold Chain Infrastructure:With the French government allocating €1.5 billion for healthcare logistics improvements, there is a substantial opportunity for cold chain providers to expand their infrastructure. This investment will facilitate the development of advanced storage and transportation facilities, enabling better service delivery and compliance with stringent regulations, ultimately enhancing the overall efficiency of the pharmaceutical supply chain.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Refrigerated Trucks Temperature-Controlled Warehouses Cold Chain Monitoring Systems |
| By End-User | Pharmaceutical Manufacturers Hospitals and Clinics Distributors and Wholesalers Research Institutions |
| By Distribution Mode | Direct Distribution Third-Party Logistics E-commerce Platforms |
| By Application | Vaccines Biopharmaceuticals Clinical Trials |
| By Sales Channel | Online Sales Offline Sales |
| By Price Range | Premium Mid-Range Budget |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Agence Nationale de Sécurité du Médicament et des Produits de Santé)
Pharmaceutical Manufacturers
Vaccine Producers
Cold Chain Logistics Providers
Healthcare Providers and Hospitals
Pharmaceutical Wholesalers
Industry Associations (e.g., Leem - Les Entreprises du Médicament)
Players Mentioned in the Report:
DHL Supply Chain
Kuehne + Nagel
DB Schenker
UPS Healthcare
FedEx
Geodis
XPO Logistics
Cardinal Health
AmerisourceBergen
Thermo Fisher Scientific
Lineage Logistics
Maersk
Panalpina
Agility Logistics
CEVA Logistics
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. France Cold Chain for Pharma & Vaccine Distribution Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 France Cold Chain for Pharma & Vaccine Distribution Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. France Cold Chain for Pharma & Vaccine Distribution Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Biologics
3.1.2 Expansion of Vaccine Distribution Networks
3.1.3 Technological Advancements in Cold Chain Logistics
3.1.4 Regulatory Compliance Requirements
3.2 Market Challenges
3.2.1 High Operational Costs
3.2.2 Infrastructure Limitations
3.2.3 Stringent Regulatory Standards
3.2.4 Risk of Temperature Excursions
3.3 Market Opportunities
3.3.1 Growth in E-commerce for Pharmaceuticals
3.3.2 Increasing Investment in Cold Chain Infrastructure
3.3.3 Adoption of IoT and Smart Technologies
3.3.4 Expansion into Emerging Markets
3.4 Market Trends
3.4.1 Shift Towards Sustainable Practices
3.4.2 Integration of AI in Supply Chain Management
3.4.3 Rise of Third-Party Logistics Providers
3.4.4 Focus on Real-Time Monitoring Solutions
3.5 Government Regulation
3.5.1 EU Good Distribution Practice (GDP) Guidelines
3.5.2 French National Cold Chain Regulations
3.5.3 Compliance with WHO Standards
3.5.4 Environmental Regulations on Refrigerants
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. France Cold Chain for Pharma & Vaccine Distribution Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. France Cold Chain for Pharma & Vaccine Distribution Market Segmentation
8.1 By Type
8.1.1 Refrigerated Trucks
8.1.2 Temperature-Controlled Warehouses
8.1.3 Cold Chain Monitoring Systems
8.2 By End-User
8.2.1 Pharmaceutical Manufacturers
8.2.2 Hospitals and Clinics
8.2.3 Distributors and Wholesalers
8.2.4 Research Institutions
8.3 By Distribution Mode
8.3.1 Direct Distribution
8.3.2 Third-Party Logistics
8.3.3 E-commerce Platforms
8.4 By Application
8.4.1 Vaccines
8.4.2 Biopharmaceuticals
8.4.3 Clinical Trials
8.5 By Sales Channel
8.5.1 Online Sales
8.5.2 Offline Sales
8.6 By Price Range
8.6.1 Premium
8.6.2 Mid-Range
8.6.3 Budget
8.7 Others
9. France Cold Chain for Pharma & Vaccine Distribution Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Operational Efficiency Ratio
9.2.7 Pricing Strategy
9.2.8 Service Level Agreements Compliance
9.2.9 Inventory Turnover Rate
9.2.10 Delivery Performance Metrics
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 DHL Supply Chain
9.5.2 Kuehne + Nagel
9.5.3 DB Schenker
9.5.4 UPS Healthcare
9.5.5 FedEx
9.5.6 Geodis
9.5.7 XPO Logistics
9.5.8 Cardinal Health
9.5.9 AmerisourceBergen
9.5.10 Thermo Fisher Scientific
9.5.11 Lineage Logistics
9.5.12 Maersk
9.5.13 Panalpina
9.5.14 Agility Logistics
9.5.15 CEVA Logistics
10. France Cold Chain for Pharma & Vaccine Distribution Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Research and Innovation
10.1.3 Ministry of Economy and Finance
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Cold Storage Facilities
10.2.2 Budget Allocation for Transportation
10.2.3 Funding for Technology Upgrades
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Manufacturers
10.3.2 Hospitals
10.3.3 Distributors
10.4 User Readiness for Adoption
10.4.1 Awareness of Cold Chain Solutions
10.4.2 Training and Skill Development
10.4.3 Infrastructure Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Performance Metrics Improvement
10.5.3 Scalability of Solutions
11. France Cold Chain for Pharma & Vaccine Distribution Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from pharmaceutical and logistics associations in France
- Review of government publications on cold chain regulations and compliance standards
- Examination of market trends and forecasts from reputable market research firms
Primary Research
- Interviews with logistics managers at pharmaceutical companies and vaccine manufacturers
- Surveys with cold chain service providers and distributors in the pharmaceutical sector
- Field interviews with regulatory experts and compliance officers in the healthcare industry
Validation & Triangulation
- Cross-validation of data through multiple sources including trade publications and expert insights
- Triangulation of findings from primary interviews with secondary data trends
- Sanity checks conducted through expert panel reviews and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total cold chain logistics spending in the pharmaceutical sector in France
- Segmentation of market size by product type, including vaccines and temperature-sensitive drugs
- Incorporation of growth rates based on historical data and future projections
Bottom-up Modeling
- Collection of operational data from leading cold chain logistics providers
- Estimation of costs associated with temperature-controlled storage and transportation
- Volume and cost analysis based on service offerings and market demand
Forecasting & Scenario Analysis
- Development of forecasting models based on current market dynamics and regulatory changes
- Scenario analysis considering potential disruptions in supply chains and demand fluctuations
- Projections for market growth under various economic conditions through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Cold Chain Logistics | 150 | Logistics Managers, Supply Chain Directors |
| Vaccine Distribution Networks | 100 | Operations Managers, Distribution Coordinators |
| Temperature-Controlled Storage Facilities | 80 | Facility Managers, Compliance Officers |
| Regulatory Compliance in Cold Chain | 70 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Pharmaceutical Supply Chain Innovations | 90 | R&D Managers, Technology Officers |
Frequently Asked Questions
What is the current value of the France Cold Chain for Pharma & Vaccine Distribution Market?
The France Cold Chain for Pharma & Vaccine Distribution Market is valued at approximately USD 2.5 billion, reflecting significant growth driven by the increasing demand for temperature-sensitive pharmaceuticals and vaccines, especially following the COVID-19 pandemic.