Region:Middle East
Author(s):Dev
Product Code:KRAC2636
Pages:88
Published On:October 2025
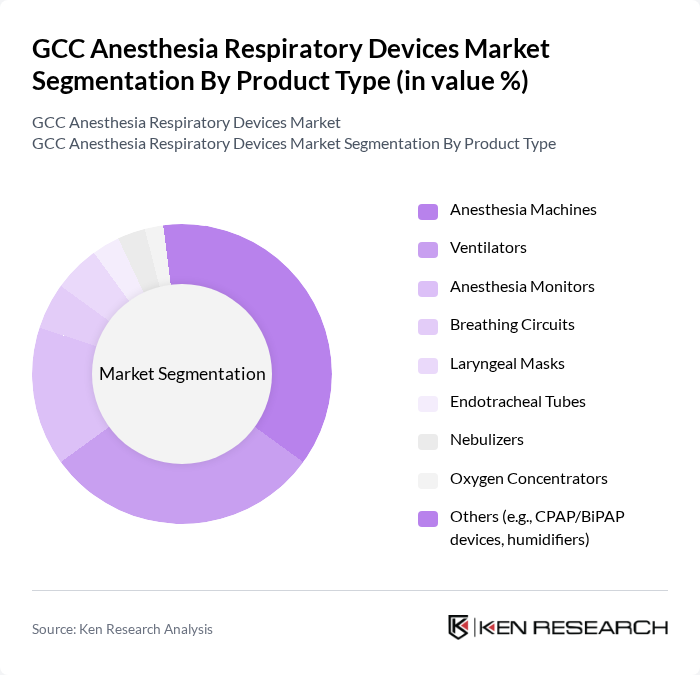
By Product Type:The product type segmentation encompasses a comprehensive range of devices essential for anesthesia and respiratory care. Subsegments include Anesthesia Machines, Ventilators, Anesthesia Monitors, Breathing Circuits, Laryngeal Masks, Endotracheal Tubes, Nebulizers, Oxygen Concentrators, and Others (such as CPAP/BiPAP devices and humidifiers). Among these, Anesthesia Machines and Ventilators are the most prominent, reflecting their critical role in surgical procedures and respiratory support. Recent trends highlight the adoption of AI-integrated anesthesia workstations and next-generation ventilators with personalized configuration, supporting improved patient outcomes and workflow efficiency .
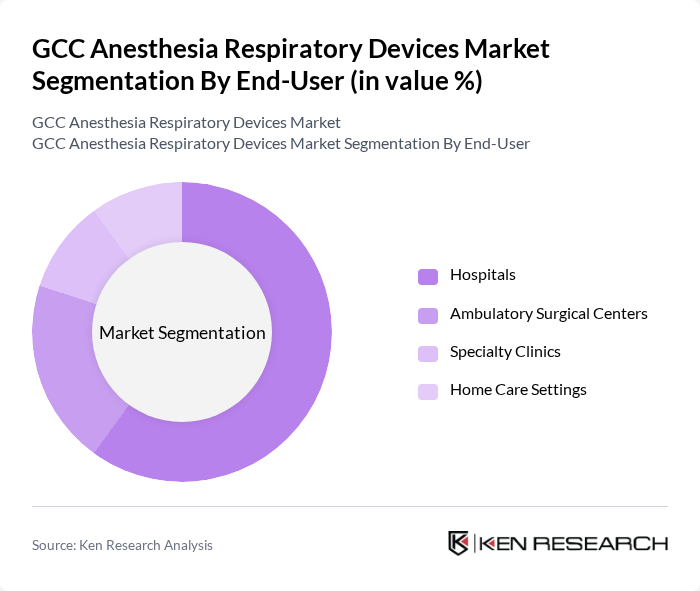
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, and Home Care Settings. Hospitals dominate this segment due to the high volume of surgical procedures and the need for advanced anesthesia and respiratory devices in critical care settings. Ambulatory Surgical Centers are experiencing notable growth, driven by the increasing trend toward outpatient surgeries and the adoption of minimally invasive techniques. Specialty clinics and home care settings are also expanding, reflecting the shift toward decentralized healthcare delivery and chronic disease management .
The GCC Anesthesia Respiratory Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Philips Healthcare (Koninklijke Philips N.V.), GE HealthCare Technologies Inc., Drägerwerk AG & Co. KGaA, Smiths Medical (ICU Medical, Inc.), Baxter International Inc., Nihon Kohden Corporation, Hillrom (Baxter International Inc.), Teleflex Incorporated, Fisher & Paykel Healthcare Corporation Limited, Getinge AB, ZOLL Medical Corporation (Asahi Kasei Corporation), Vyaire Medical, Inc., Ambu A/S, Intersurgical Ltd., Mindray Medical International Limited, Hamilton Medical AG, Becton, Dickinson and Company (BD) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC anesthesia respiratory devices market appears promising, driven by technological advancements and an increasing focus on patient safety. The integration of artificial intelligence in anesthesia devices is expected to enhance monitoring capabilities, improving patient outcomes. Additionally, the shift towards minimally invasive procedures will likely increase the demand for portable anesthesia devices, further shaping the market landscape. As healthcare infrastructure expands, the region is poised for significant growth in this sector.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Anesthesia Machines Ventilators Anesthesia Monitors Breathing Circuits Laryngeal Masks Endotracheal Tubes Nebulizers Oxygen Concentrators Others (e.g., CPAP/BiPAP devices, humidifiers) |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Home Care Settings |
| By Application | General Surgery Cardiac Surgery Orthopedic Surgery Neurological Surgery Respiratory Therapy |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Price Range | Low Range Mid Range High Range |
| By Policy Support | Subsidies for medical devices Tax incentives for manufacturers Grants for research and development Regulatory support for market entry |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Anesthesia Device Procurement | 100 | Procurement Managers, Hospital Administrators |
| Clinical Usage of Respiratory Devices | 80 | Anesthesiologists, Respiratory Therapists |
| Market Trends in Medical Device Distribution | 70 | Medical Device Distributors, Sales Representatives |
| Regulatory Compliance and Standards | 60 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Healthcare Facility Infrastructure | 60 | Facility Managers, Biomedical Engineers |
The GCC Anesthesia Respiratory Devices Market is valued at approximately USD 1.1 billion, reflecting a robust growth trajectory driven by advancements in technology and increasing healthcare demands in the region.