Region:Middle East
Author(s):Rebecca
Product Code:KRAD4894
Pages:99
Published On:December 2025
By Product Type:The product type segmentation includes various categories of catheter securement devices that cater to different medical needs. The subsegments are as follows:
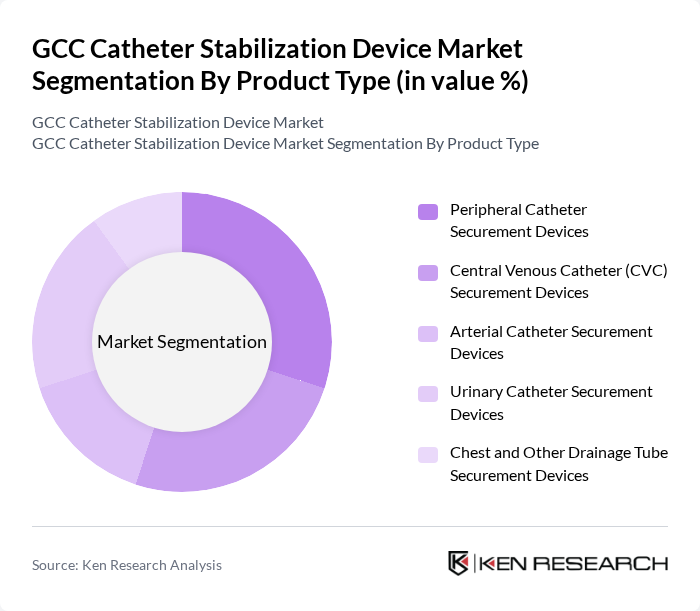
The Peripheral Catheter Securement Devices segment is currently leading the market due to their widespread use in hospitals and outpatient settings. These devices are essential for ensuring the stability of peripheral catheters, which are commonly used for intravenous therapy. The increasing number of outpatient procedures and the growing emphasis on patient comfort and safety are driving the demand for these devices. Additionally, advancements in skin-friendly adhesive materials and ergonomic designs have improved the effectiveness and ease of use of peripheral securement devices, further solidifying their market dominance.
By End-User:The end-user segmentation includes various healthcare settings where catheter stabilization devices are utilized. The subsegments are as follows:
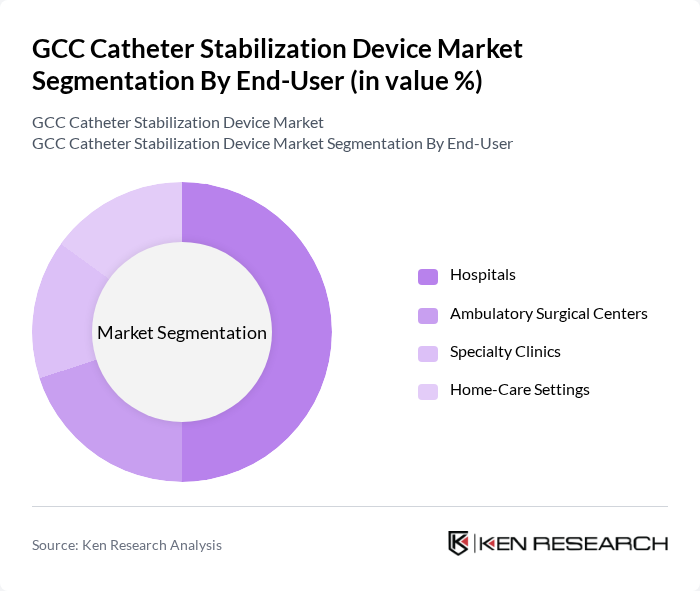
Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is primarily due to the high volume of surgical procedures performed in hospitals, which require effective catheter stabilization to minimize complications. The increasing focus on patient safety and infection control in hospital settings has led to a greater adoption of advanced catheter stabilization devices. Furthermore, hospitals are investing in innovative technologies to enhance patient care, and the shift toward home-based and self-catheterization care is elevating the need for secure catheter fixation across diverse care settings, further driving the demand for these devices.
The GCC Catheter Stabilization Device Market is characterized by a dynamic mix of regional and international players. Leading participants such as 3M Company, B. Braun Melsungen AG, Baxter International Inc., Cardinal Health Inc., ConvaTec Group plc, Merit Medical Systems Inc., Smiths Medical (ICU Medical Inc.), Medtronic plc, Teleflex Incorporated, C. R. Bard (Becton, Dickinson and Company), Centurion Medical Products Corporation, M.C. Johnson Company Inc., Vygon SA, Johnson & Johnson (including Ethicon), Amecath Medical Technologies contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC catheter stabilization device market appears promising, driven by ongoing advancements in technology and a growing emphasis on patient-centered care. As healthcare providers increasingly prioritize infection control and patient comfort, the integration of smart technologies into catheter stabilization devices is expected to gain traction. Furthermore, the expansion of telemedicine and remote monitoring solutions will likely enhance the management of chronic diseases, creating a favorable environment for innovative product development and market growth.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Peripheral Catheter Securement Devices Central Venous Catheter (CVC) Securement Devices Arterial Catheter Securement Devices Urinary Catheter Securement Devices Chest and Other Drainage Tube Securement Devices |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Home-Care Settings |
| By Application | Cardiovascular Procedures Urological Procedures Respiratory Procedures General Surgery and Other Procedures |
| By Material | Silicone-Based Securement Devices Hydrocolloid and Acrylic Adhesive Devices Foam and Fabric-Based Devices Others |
| By Distribution Channel | Direct Tenders to Hospitals and Ministries Local Distributors and Importers Group Purchasing Organizations (GPOs) Online and Other Channels |
| By Country | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain |
| By Pricing Tier | Premium International Brands Mid-Priced Value Brands Economy and Private-Label Brands Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 45 | Procurement Managers, Supply Chain Coordinators |
| Outpatient Clinics | 35 | Clinic Managers, Nursing Supervisors |
| Medical Device Distributors | 30 | Sales Representatives, Distribution Managers |
| Healthcare Regulatory Bodies | 20 | Regulatory Affairs Specialists, Compliance Officers |
| Clinical Research Organizations | 25 | Clinical Researchers, Project Managers |
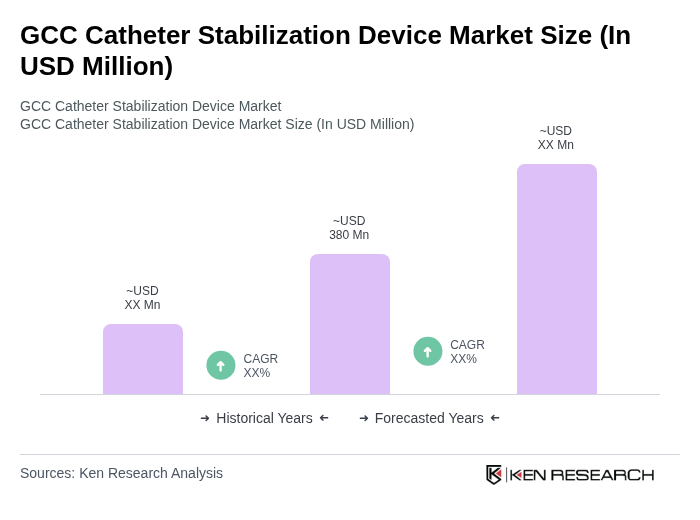
The GCC Catheter Stabilization Device Market is valued at approximately USD 380 million, driven by the increasing prevalence of chronic diseases, surgical procedures, and advancements in medical technology that enhance patient safety and comfort.