Region:Middle East
Author(s):Geetanshi
Product Code:KRAC3771
Pages:86
Published On:October 2025
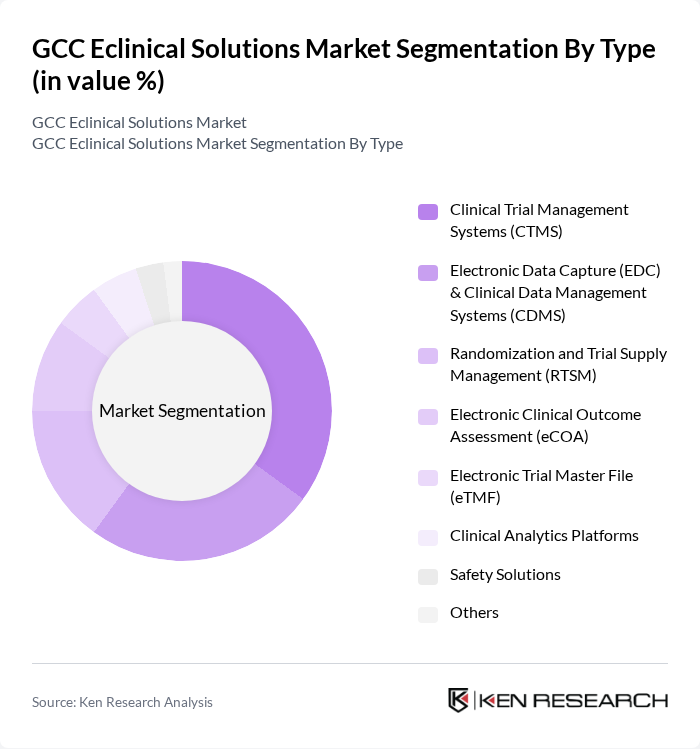
By Type:The market is segmented into various types, including Clinical Trial Management Systems (CTMS), Electronic Data Capture (EDC) & Clinical Data Management Systems (CDMS), Randomization and Trial Supply Management (RTSM), Electronic Clinical Outcome Assessment (eCOA), Electronic Trial Master File (eTMF), Clinical Analytics Platforms, Safety Solutions, and Others. Among these, Clinical Trial Management Systems (CTMS) is the leading sub-segment, driven by the increasing complexity of clinical trials, the need for efficient management of trial data and processes, and the growing adoption of integrated platforms for real-time monitoring and regulatory compliance .
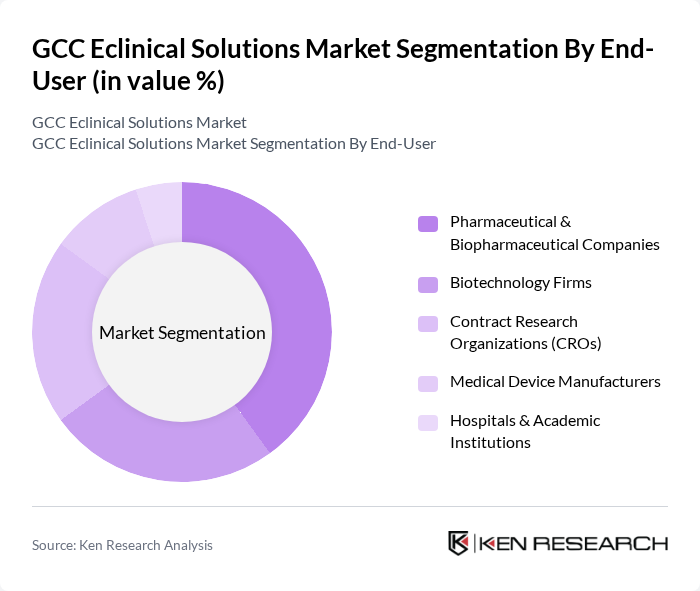
By End-User:The end-user segmentation includes Pharmaceutical & Biopharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Medical Device Manufacturers, and Hospitals & Academic Institutions. Pharmaceutical & Biopharmaceutical Companies dominate this segment due to their extensive involvement in clinical trials, the need for robust eclinical solutions to manage complex data and regulatory requirements, and the increasing trend of outsourcing clinical research to specialized organizations .
The GCC Eclinical Solutions Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Inc., Oracle Corporation, Veeva Systems Inc., Parexel International Corporation, Labcorp (Covance Inc.), Clario (formerly ERT and BioClinica), CRF Health (now part of Clario), Medpace Holdings, Inc., PPD, Inc. (part of Thermo Fisher Scientific), Syneos Health, Inc., ICON plc, Charles River Laboratories International, Inc., WCG Clinical, KCR S.A., DATATRAK International, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC eClinical solutions market appears promising, driven by technological advancements and a shift towards patient-centric care. As healthcare providers increasingly embrace decentralized clinical trials, the demand for innovative eClinical solutions will likely rise. Furthermore, the integration of artificial intelligence in clinical research is expected to enhance data analysis and decision-making processes, paving the way for more efficient and effective clinical trials in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Trial Management Systems (CTMS) Electronic Data Capture (EDC) & Clinical Data Management Systems (CDMS) Randomization and Trial Supply Management (RTSM) Electronic Clinical Outcome Assessment (eCOA) Electronic Trial Master File (eTMF) Clinical Analytics Platforms Safety Solutions Others |
| By End-User | Pharmaceutical & Biopharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Medical Device Manufacturers Hospitals & Academic Institutions |
| By Application | Oncology Trials Cardiovascular Trials Infectious Disease Trials Neurology Trials Rare Disease Trials Others |
| By Deployment Mode | Cloud-Based Solutions (SaaS) Web-Hosted (On-Demand) Solutions On-Premise Solutions |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Sales Channel | Direct Sales Distributors Online Sales |
| By Pricing Model | Subscription-Based Pricing Pay-Per-Use Pricing One-Time License Fee |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 60 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Healthcare IT Solutions | 50 | IT Directors, eClinical Solution Architects |
| Patient Management Systems | 40 | Healthcare Administrators, Patient Care Coordinators |
| Data Management in Clinical Research | 45 | Data Analysts, Clinical Research Associates |
| Regulatory Compliance in eClinical Solutions | 40 | Compliance Officers, Quality Assurance Managers |
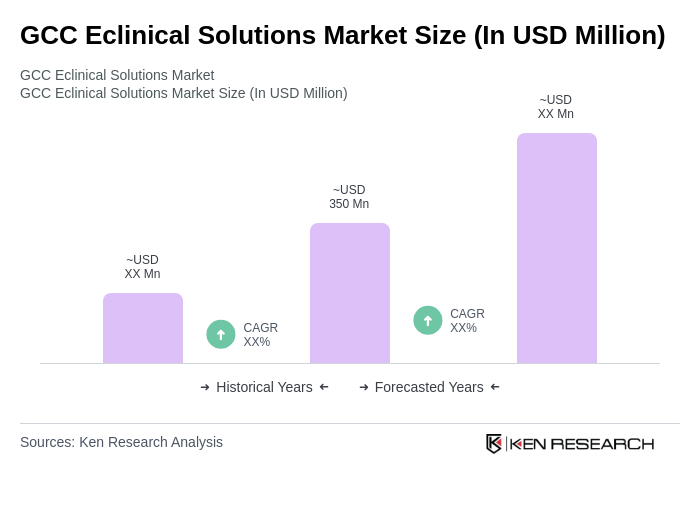
The GCC Eclinical Solutions Market is valued at approximately USD 350 million, reflecting a significant growth driven by the increasing demand for efficient clinical trial management and the adoption of advanced digital technologies in healthcare.