Region:Middle East
Author(s):Shubham
Product Code:KRAC9001
Pages:95
Published On:November 2025
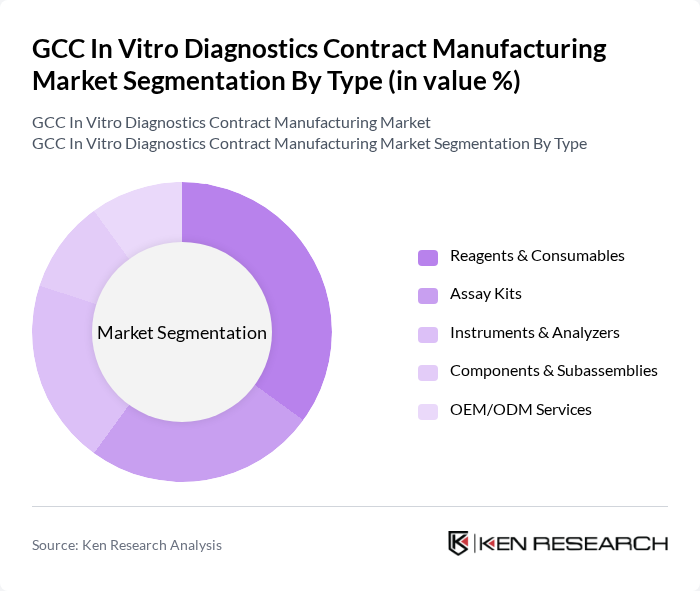
By Type:The market is segmented into Reagents & Consumables, Assay Kits, Instruments & Analyzers, Components & Subassemblies, and OEM/ODM Services. Each of these subsegments plays a crucial role in the overall market dynamics, with demand patterns shaped by the shift toward rapid diagnostics, increased use of molecular and immunoassay platforms, and the need for flexible, scalable manufacturing solutions among diagnostic companies .
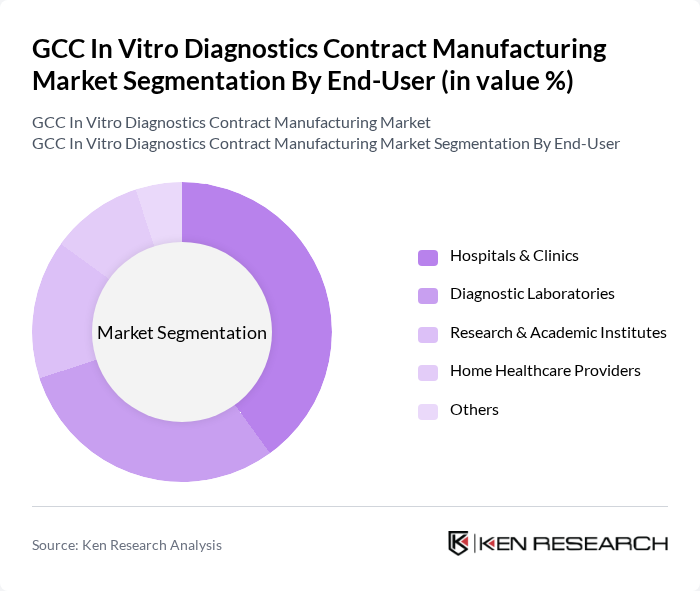
By End-User:The end-user segmentation includes Hospitals & Clinics, Diagnostic Laboratories, Research & Academic Institutes, Home Healthcare Providers, and Others. Each segment has unique requirements and preferences that influence the demand for in vitro diagnostic products and services. Hospitals and clinics remain the largest consumers due to high patient throughput and broad diagnostic needs, while diagnostic laboratories drive demand for specialized and high-throughput solutions .
The GCC In Vitro Diagnostics Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Al Hayat Pharmaceuticals (UAE), Gulf Medical Co. Ltd. (Saudi Arabia), Al Zahrawi Medical (UAE/Saudi Arabia), BioGenix Lab (UAE), Julphar Gulf Pharmaceutical Industries (UAE), National Reference Laboratory (UAE), Al Borg Diagnostics (Saudi Arabia), Siemens Healthineers, Roche Diagnostics, Abbott Laboratories, Thermo Fisher Scientific, Bio-Rad Laboratories, QIAGEN, Mindray Medical International Limited, Sysmex Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC in vitro diagnostics contract manufacturing market appears promising, driven by the increasing integration of digital health solutions and the growing emphasis on preventive healthcare. As telemedicine and remote diagnostics gain traction, manufacturers are likely to adapt their offerings to meet evolving consumer needs. Additionally, the ongoing investment in healthcare infrastructure across the GCC will further bolster the demand for innovative diagnostic solutions, positioning the region as a key player in the global market.
| Segment | Sub-Segments |
|---|---|
| By Type | Reagents & Consumables Assay Kits Instruments & Analyzers Components & Subassemblies OEM/ODM Services |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Research & Academic Institutes Home Healthcare Providers Others |
| By Application | Infectious Disease Testing Oncology/Cancer Diagnostics Cardiology Diagnostics Genetic & Molecular Testing Others |
| By Distribution Channel | Direct to OEMs Distributors/Wholesalers Online B2B Platforms Others |
| By Region | Saudi Arabia United Arab Emirates (UAE) Qatar Kuwait Oman |
| By Technology | Molecular Diagnostics Immunoassays Clinical Chemistry Hematology Point-of-Care Testing Others |
| By Regulatory Compliance | ISO 13485 Certification CE Marking SFDA/MOH Approval (GCC-specific) FDA Approval Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 60 | Laboratory Managers, Quality Control Officers |
| Hospital Procurement Departments | 50 | Procurement Managers, Supply Chain Directors |
| Diagnostic Equipment Manufacturers | 40 | Product Development Managers, Sales Executives |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
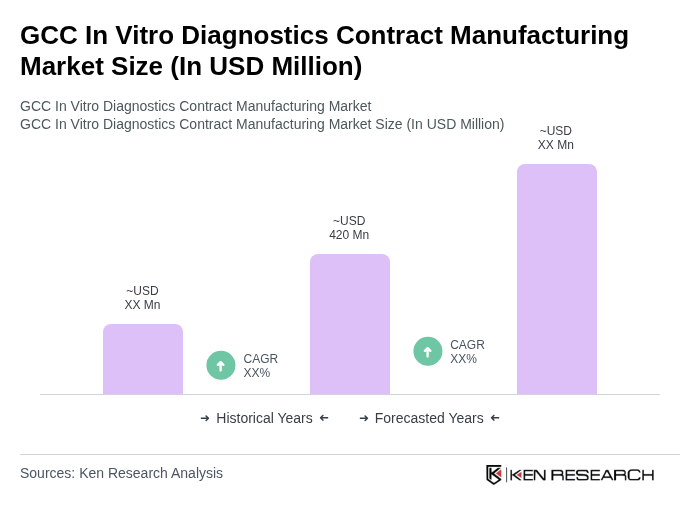
The GCC In Vitro Diagnostics Contract Manufacturing Market is valued at approximately USD 420 million. This valuation reflects the growing demand for advanced diagnostic solutions driven by chronic disease prevalence and healthcare advancements in the region.