Region:Middle East
Author(s):Geetanshi
Product Code:KRAD6027
Pages:81
Published On:December 2025
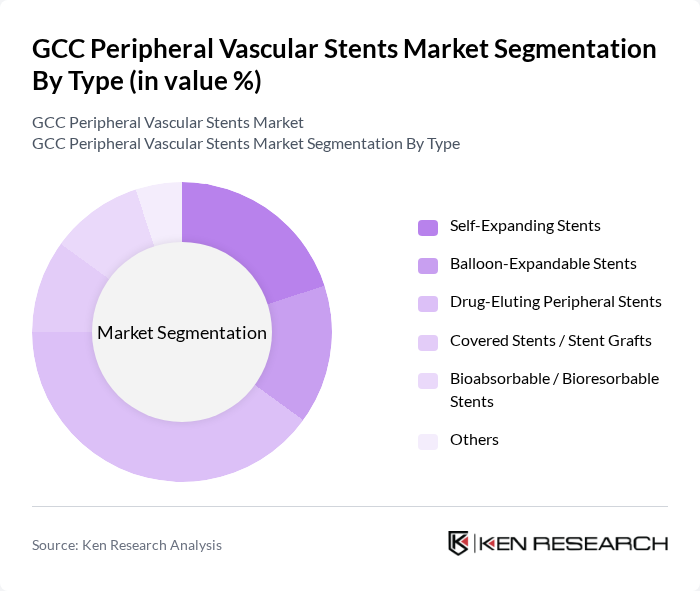
By Type:The market is segmented into various types of stents, including Self-Expanding Stents, Balloon-Expandable Stents, Drug-Eluting Peripheral Stents, Covered Stents / Stent Grafts, Bioabsorbable / Bioresorbable Stents, and Others. This typology is consistent with global peripheral stent classifications used in major market and clinical studies. Among these, Drug-Eluting Peripheral Stents are gaining traction due to their ability to reduce the risk of restenosis and improve long?term vessel patency, making them a preferred choice for many healthcare providers, in line with global trends where drug?eluting stents hold the largest revenue share in peripheral stents. The increasing awareness of the benefits of these stents, along with the shift toward evidence?based endovascular therapy and broader reimbursement for advanced stent technologies in tertiary urban hospitals, is driving their adoption, particularly in urban areas with advanced healthcare facilities.
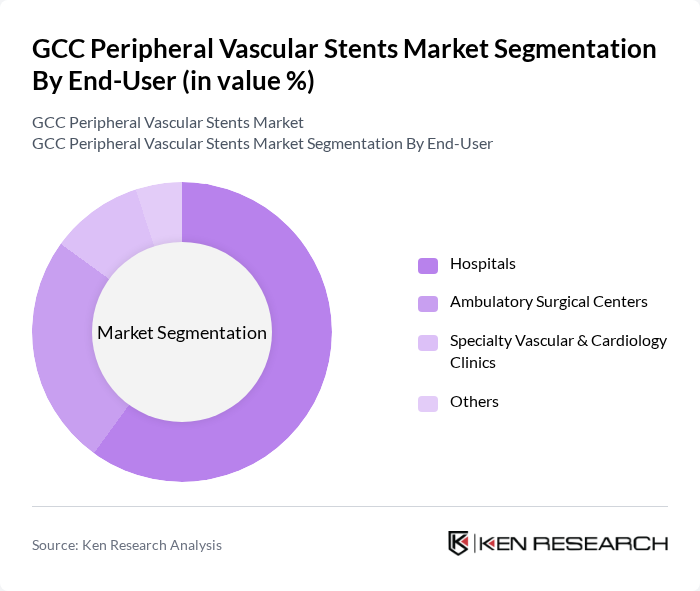
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Vascular & Cardiology Clinics, and Others. This segmentation aligns with global peripheral vascular device and stent market analyses, which consistently identify hospitals, ambulatory surgical centers, and specialty clinics as the primary sites of use. Hospitals are the dominant end-user segment, primarily due to their capacity to provide comprehensive care, complex endovascular and hybrid surgical procedures, and intensive care support; global data show hospitals hold the largest share of peripheral vascular device procedures. The increasing number of patients requiring vascular interventions in hospitals, expansion of catheterization labs and vascular units, and the concentration of interventional radiologists and vascular surgeons in these facilities are driving the demand for stents, as these centers are equipped with the necessary imaging technology, inventory, and expertise to handle complex peripheral artery disease cases.
The GCC Peripheral Vascular Stents Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, B. Braun Melsungen AG, Cook Medical LLC, Terumo Corporation, Cardinal Health, Inc., W. L. Gore & Associates, Inc. (Gore Medical), BIOTRONIK SE & Co. KG, Merit Medical Systems, Inc., Endologix LLC, Koninklijke Philips N.V. (Philips Image-Guided Therapy Devices), Stryker Corporation, Alvimedica Medical Technologies Inc., Biosensors International Group, Ltd. contribute to innovation, geographic expansion, and service delivery in this space, mirroring their strong global positions in peripheral stents and broader peripheral vascular devices.
The future of the GCC peripheral vascular stents market appears promising, driven by ongoing technological advancements and an increasing focus on patient-centric care. As healthcare infrastructure expands, particularly in underserved areas, access to vascular interventions is expected to improve. Additionally, the integration of digital health technologies will enhance patient monitoring and outcomes, further propelling market growth. Stakeholders must remain agile to adapt to evolving regulatory landscapes and patient needs in this dynamic environment.
| Segment | Sub-Segments |
|---|---|
| By Type | Self-Expanding Stents Balloon-Expandable Stents Drug-Eluting Peripheral Stents Covered Stents / Stent Grafts Bioabsorbable / Bioresorbable Stents Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Vascular & Cardiology Clinics Others |
| By Material | Nitinol Stainless Steel Cobalt-Chromium Alloys Polymer-Based / Bioabsorbable Materials Others |
| By Delivery Method | Balloon-Expandable Stents Self-Expanding Stents Others |
| By Application | Peripheral Artery Disease of Lower Extremities (Fem-Pop, Iliac, Infrapopliteal) Aortic & Iliac Aneurysm Repair Carotid Artery Disease Renal & Mesenteric Artery Disease Others |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Distribution Channel | Direct Sales to Hospitals and Health Systems Local & Regional Distributors Group Purchasing Organizations (GPOs) & Tender-Based Procurement Online / E-Procurement Platforms Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiovascular Surgeons | 60 | Interventional Cardiologists, Vascular Surgeons |
| Hospital Procurement Managers | 50 | Supply Chain Managers, Purchasing Directors |
| Medical Device Sales Representatives | 40 | Sales Managers, Territory Representatives |
| Healthcare Policy Makers | 40 | Health Economists, Regulatory Affairs Specialists |
| Clinical Researchers | 40 | Clinical Trial Coordinators, Research Scientists |
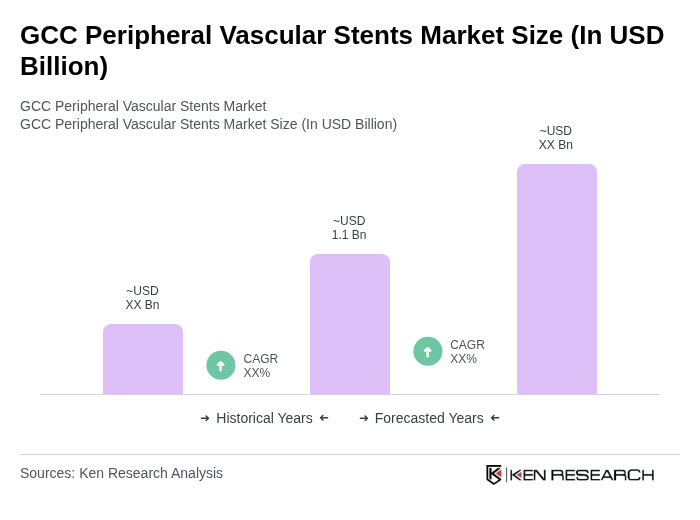
The GCC Peripheral Vascular Stents Market is valued at approximately USD 1.1 billion, reflecting significant growth driven by the rising prevalence of peripheral artery diseases and advancements in stent technology within the region.