Region:Middle East
Author(s):Shubham
Product Code:KRAC4248
Pages:89
Published On:October 2025
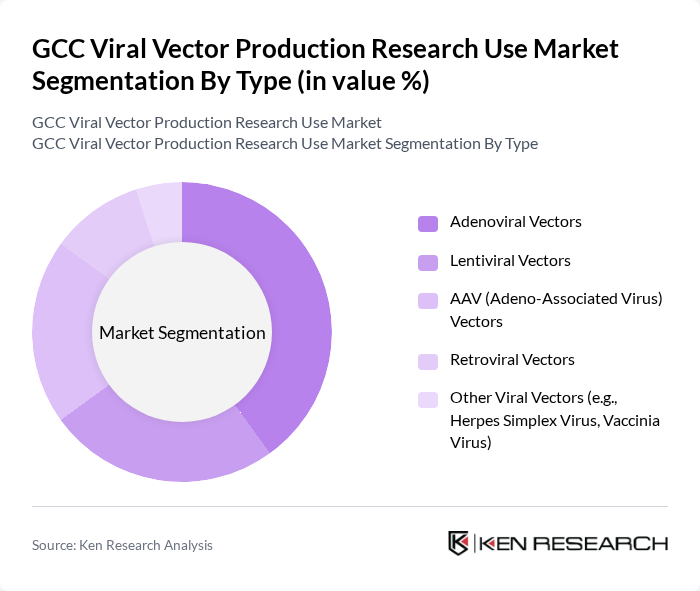
By Type:The market is segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV (Adeno-Associated Virus) Vectors, Retroviral Vectors, and Other Viral Vectors (e.g., Herpes Simplex Virus, Vaccinia Virus). Among these,Adenoviral Vectorsare leading the market due to their high transduction efficiency and ability to accommodate large genetic payloads, making them ideal for gene therapy applications.Lentiviral Vectorsare also gaining traction, particularly in the field of oncology and gene editing, due to their stable integration into the host genome. These trends are consistent with global advancements in vector design and manufacturing, as well as increased clinical trial activity in gene and cell therapy .
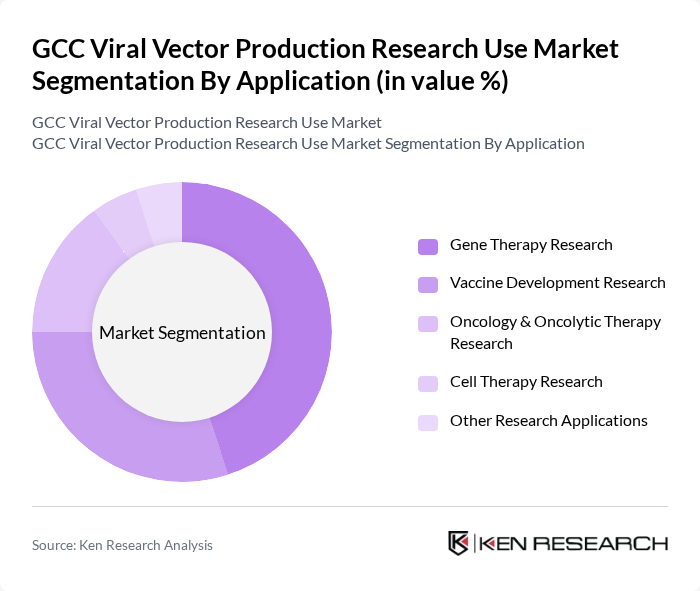
By Application:The applications of viral vectors in the GCC market include Gene Therapy Research, Vaccine Development Research, Oncology & Oncolytic Therapy Research, Cell Therapy Research, and Other Research Applications.Gene Therapy Researchis the dominant application segment, driven by the increasing incidence of genetic disorders and the growing focus on personalized medicine.Vaccine Development Researchis also significant, especially in light of recent global health challenges, leading to increased investments in viral vector-based vaccines. The market is further supported by the expansion of clinical trials and the integration of viral vectors in regenerative medicine and immunotherapy research .
The GCC Viral Vector Production Research Use Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Thermo Fisher Scientific Inc. (Brammer Bio), FUJIFILM Diosynth Biotechnologies, Oxford Biomedica plc, Catalent Inc., Merck KGaA, Cobra Biologics (Charles River Laboratories), Wuxi Biologics, Takara Bio Inc., Batavia Biosciences, Miltenyi Biotec GmbH, UniQure N.V., FinVector Oy, Genezen Laboratories, Biovian Oy contribute to innovation, geographic expansion, and service delivery in this space.
The GCC viral vector production market is poised for significant advancements, driven by ongoing research and technological innovations. As personalized medicine gains traction, the demand for tailored gene therapies will likely increase, prompting further investment in production capabilities. Additionally, collaborations between biotech firms and academic institutions are expected to foster innovation, enhancing the region's competitive edge. With a focus on sustainable practices and scalable production methods, the market is set to evolve, addressing both current challenges and future needs effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV (Adeno-Associated Virus) Vectors Retroviral Vectors Other Viral Vectors (e.g., Herpes Simplex Virus, Vaccinia Virus) |
| By Application | Gene Therapy Research Vaccine Development Research Oncology & Oncolytic Therapy Research Cell Therapy Research Other Research Applications |
| By End-User | Academic & Research Institutes Pharmaceutical & Biotechnology Companies Contract Development and Manufacturing Organizations (CDMOs) Hospitals & Clinical Laboratories |
| By Distribution Channel | Direct Sales Online Sales Distributors/Wholesalers |
| By Region | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain Rest of GCC |
| By Research Type | Preclinical Research Clinical Research Translational Research |
| By Funding Source | Government Grants Private Investments Academic & Institutional Funding International Collaborations |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Developers | 60 | R&D Directors, Product Managers |
| Vaccine Manufacturers | 50 | Production Managers, Quality Assurance Leads |
| Contract Manufacturing Organizations (CMOs) | 40 | Operations Directors, Business Development Managers |
| Academic Research Institutions | 40 | Principal Investigators, Lab Managers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
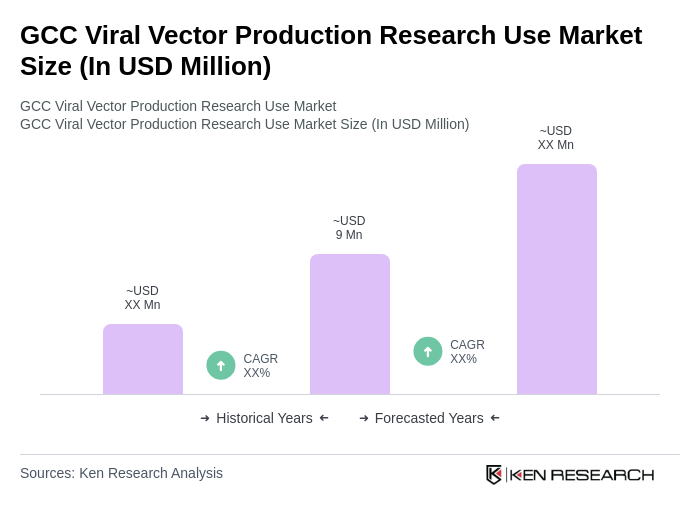
The GCC Viral Vector Production Research Use Market is valued at approximately USD 9 million. This valuation reflects the increasing demand for gene therapies, advancements in biotechnology, and significant investments in research and development within the region.