Region:Middle East
Author(s):Geetanshi
Product Code:KRAC9368
Pages:91
Published On:November 2025
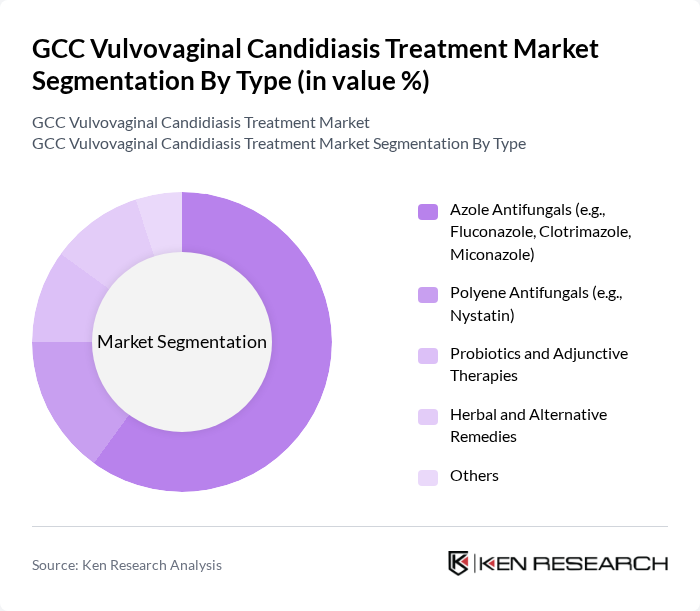
By Type:The market is segmented into various types of antifungal treatments, which include Azole Antifungals, Polyene Antifungals, Probiotics and Adjunctive Therapies, Herbal and Alternative Remedies, and Others. Among these, Azole Antifungals, such as Fluconazole and Clotrimazole, dominate the market due to their widespread use and effectiveness in treating vulvovaginal candidiasis. The convenience of oral and topical formulations has led to increased consumer preference for these products.
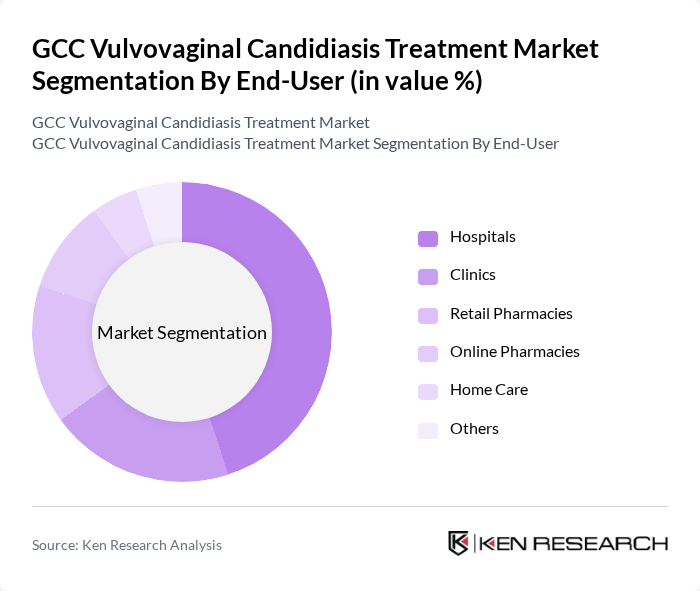
By End-User:The end-user segmentation includes Hospitals, Clinics, Retail Pharmacies, Online Pharmacies, Home Care, and Others. Hospitals are the leading end-users due to their capacity to provide comprehensive care and access to a wide range of antifungal treatments. The increasing number of outpatient visits and the growing trend of self-medication through pharmacies also contribute to the market dynamics.
The GCC Vulvovaginal Candidiasis Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer Inc., Johnson & Johnson, GlaxoSmithKline plc (GSK), Bayer AG, Merck & Co., Inc., Abbott Laboratories, Novartis AG, Sanofi S.A., Astellas Pharma Inc., Teva Pharmaceutical Industries Ltd., Hikma Pharmaceuticals PLC, Julphar (Gulf Pharmaceutical Industries), Tabuk Pharmaceuticals Manufacturing Co., SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Sandoz (a Novartis division) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC vulvovaginal candidiasis treatment market appears promising, driven by increasing healthcare investments and a focus on women's health. With the GCC governments projected to increase healthcare spending by 10% in future, innovative treatment options and telemedicine services are expected to flourish. Additionally, the growing emphasis on preventive healthcare will likely lead to enhanced patient engagement and education, fostering a more proactive approach to managing vulvovaginal candidiasis in the region.
| Segment | Sub-Segments |
|---|---|
| By Type | Azole Antifungals (e.g., Fluconazole, Clotrimazole, Miconazole) Polyene Antifungals (e.g., Nystatin) Probiotics and Adjunctive Therapies Herbal and Alternative Remedies Others |
| By End-User | Hospitals Clinics Retail Pharmacies Online Pharmacies Home Care Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Geography | Saudi Arabia United Arab Emirates (UAE) Qatar Kuwait Oman Bahrain |
| By Treatment Duration | Short-term Treatment (?7 days) Long-term/Recurrent Treatment (>7 days or maintenance) |
| By Patient Demographics | Age Group (18-30, 31-45, 46-60, 60+) Socioeconomic Status Pregnancy Status Others |
| By Product Formulation | Oral Tablets/Capsules Topical Creams/Ointments Vaginal Suppositories/Pessaries Vaginal Tablets Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gynecological Clinics | 100 | Gynecologists, Nurse Practitioners |
| Pharmacy Chains | 80 | Pharmacists, Pharmacy Managers |
| Patient Focus Groups | 50 | Women aged 18-50 with candidiasis experience |
| Healthcare Policy Experts | 40 | Health Economists, Policy Advisors |
| Clinical Researchers | 60 | Clinical Researchers, Medical Professors |
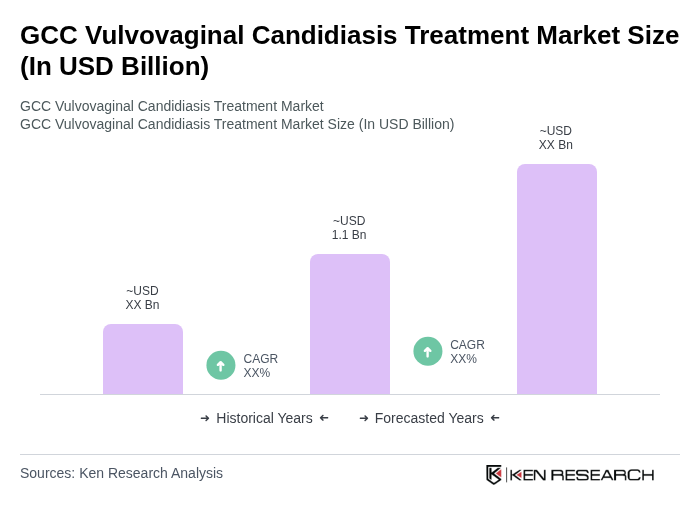
The GCC Vulvovaginal Candidiasis Treatment Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by the increasing prevalence of vulvovaginal candidiasis and advancements in treatment options available in the region.