Region:Global
Author(s):Shubham
Product Code:KRAA8734
Pages:89
Published On:November 2025
By Offering:
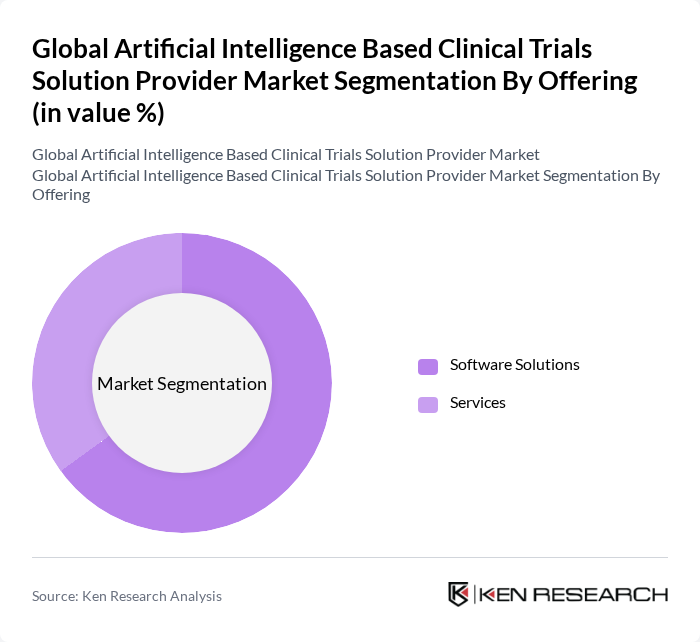
The offering segment includes two primary subsegments: Software Solutions and Services. Software Solutions encompass Clinical Trial Management Systems, Electronic Data Capture, Predictive Analytics Tools, Patient Recruitment & Retention Platforms, and Real-World Evidence Platforms. Services include Consulting, Data Management & Analysis, Regulatory Compliance, and Training & Support. Among these, Software Solutions are dominating the market due to the increasing demand for automated systems that enhance data accuracy and streamline trial processes. The trend towards digital transformation in clinical trials is driving the adoption of these software solutions, making them essential for efficient trial management.
By Process:
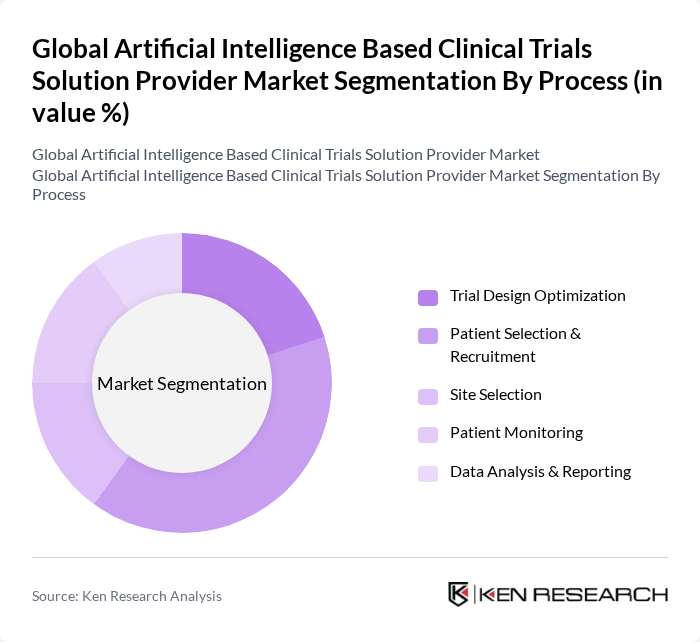
This segment includes Trial Design Optimization, Patient Selection & Recruitment, Site Selection, Patient Monitoring, and Data Analysis & Reporting. The Patient Selection & Recruitment process is currently leading the market, driven by the need for more targeted and efficient recruitment strategies that leverage AI capabilities. As clinical trials become more complex, the ability to quickly identify and enroll suitable participants is critical for success. This trend is further supported by advancements in data analytics and machine learning, which enhance the precision of patient selection. AI technologies are refining patient selection by examining electronic health records and genetic information, ensuring prompt identification of appropriate candidates and minimizing trial durations and expenses.
The Global Artificial Intelligence Based Clinical Trials Solution Provider Market is characterized by a dynamic mix of regional and international players. Leading participants such as IBM Watson Health, Medidata Solutions (Dassault Systèmes), Oracle Health Sciences, Parexel International, Covance (Labcorp Drug Development), Syneos Health, Veeva Systems, CRF Health (Signant Health), BioClinica (now part of ERT/Clario), TrialSpark, Science 37, Medable, Deep 6 AI, Antidote Technologies, ClinOne, ConcertAI, Sema4 (now GeneDx), Saama Technologies, Philips Healthcare, Intel Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of AI-based clinical trials is poised for significant transformation, driven by technological advancements and evolving regulatory frameworks. As organizations increasingly embrace virtual and decentralized trials, the integration of AI will enhance patient engagement and streamline processes. Furthermore, the focus on personalized medicine will likely accelerate the development of tailored therapies, supported by AI-driven insights. This convergence of technology and patient-centric approaches will redefine clinical trial methodologies, fostering innovation and improving outcomes in the healthcare sector.
| Segment | Sub-Segments |
|---|---|
| By Offering | Software Solutions (e.g., Clinical Trial Management Systems, Electronic Data Capture, Predictive Analytics Tools, Patient Recruitment & Retention Platforms, Real-World Evidence Platforms) Services (e.g., Consulting, Data Management & Analysis, Regulatory Compliance, Training & Support) |
| By Process | Trial Design Optimization Patient Selection & Recruitment Site Selection Patient Monitoring Data Analysis & Reporting |
| By Application | Oncology Trials Neurological Disease Trials Cardiovascular Disease Trials Metabolic Disease Trials Infectious Disease Trials Immunology Disease Trials Others |
| By End-User | Pharmaceutical Companies Biotechnology Companies Contract Research Organizations (CROs) Academic & Research Institutions Others |
| By Technology | Machine Learning Deep Learning Natural Language Processing Computer Vision Robotic Process Automation Other Technologies |
| By Clinical Trial Phase | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Clinical Trials | 100 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Research Initiatives | 80 | Biotech Researchers, Project Leads |
| Medical Device Trials | 60 | Product Development Managers, Clinical Affairs Directors |
| AI Technology Providers | 50 | AI Developers, Business Development Executives |
| Healthcare Regulatory Bodies | 40 | Regulatory Compliance Officers, Policy Makers |
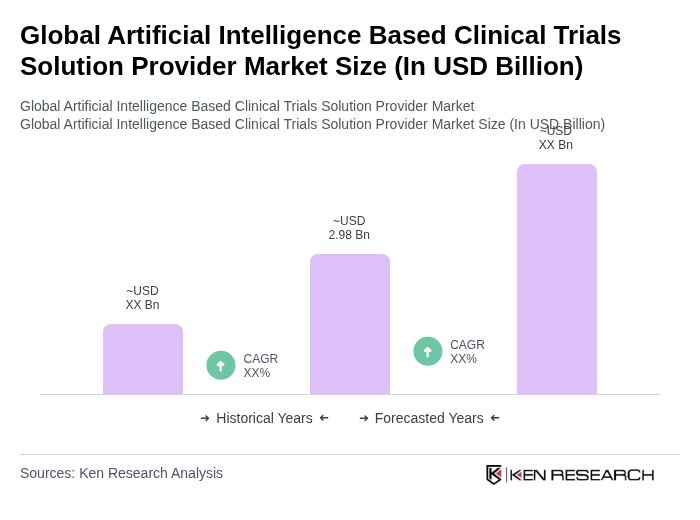
The Global Artificial Intelligence Based Clinical Trials Solution Provider Market is valued at approximately USD 2.98 billion, reflecting significant growth driven by the increasing adoption of AI technologies in healthcare and the demand for faster drug development processes.