About the Report
Base Year 2024Global Bacterial Vaginosis Diagnostics Market Overview
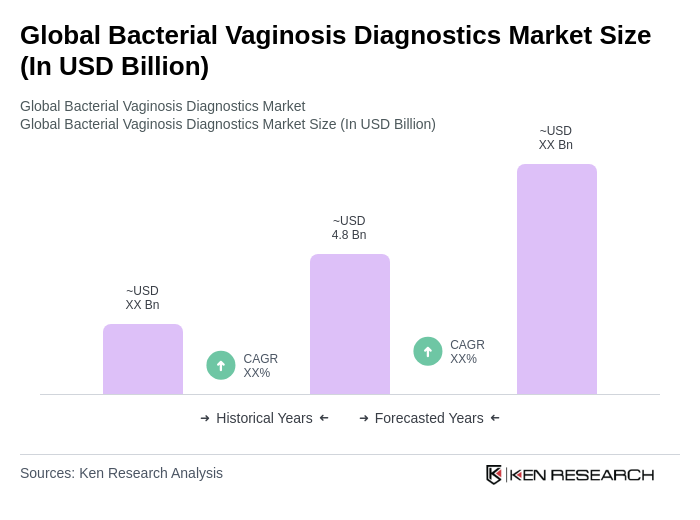
- The Global Bacterial Vaginosis Diagnostics Market is valued at USD 4.8 billion, based on a five-year historical analysis. This market size reflects the substantial economic burden associated with the diagnosis and treatment of bacterial vaginosis, as well as the costs of related complications such as preterm birth and increased susceptibility to HIV. Growth is primarily driven by the high global prevalence of bacterial vaginosis among women (23–29%), rising awareness of women's health issues, and ongoing advancements in diagnostic technologies. Additional growth drivers include increased healthcare expenditure and a growing demand for rapid, accurate diagnostic tests, particularly in both developed and emerging markets .
- Key players in this market include the United States, Germany, and Japan, which maintain dominance due to advanced healthcare infrastructure, significant investment in research and development, and a relatively high prevalence of bacterial vaginosis. The presence of leading diagnostic companies and a strong focus on women's health initiatives further reinforce their market leadership .
- In 2023, the U.S. Food and Drug Administration (FDA) strengthened regulatory requirements for diagnostic tests, mandating rigorous validation processes for all bacterial vaginosis diagnostics. These measures are designed to ensure the accuracy and reliability of test results, thereby enhancing patient safety and improving treatment outcomes .
Global Bacterial Vaginosis Diagnostics Market Segmentation
By Type:The market is segmented into various types of diagnostic tests, including PCR-based tests, culture tests, rapid diagnostic tests, microscopy tests, immunoassays, and others. PCR-based tests are gaining significant traction due to their high sensitivity and specificity, making them the preferred choice for accurate diagnosis. Culture tests, while traditional, remain widely used in clinical settings for confirmation and research purposes. Rapid diagnostic tests are increasingly popular due to their convenience, speed, and suitability for point-of-care testing, reflecting the growing trend toward decentralized healthcare delivery .

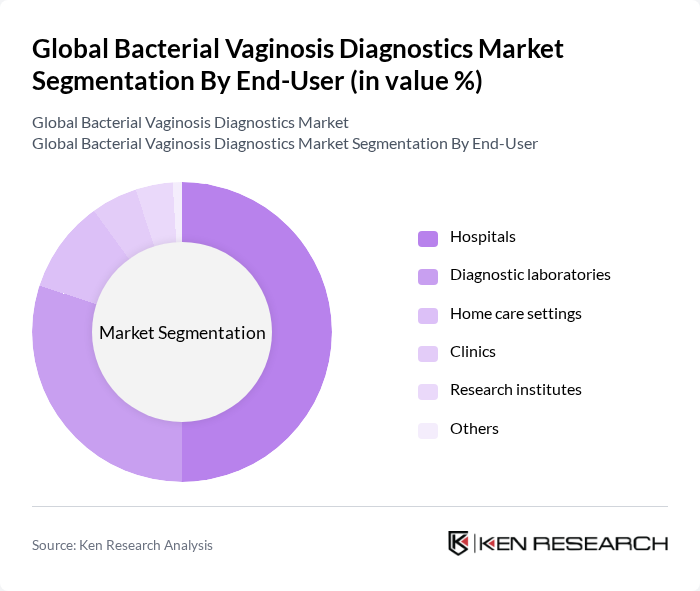
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, home care settings, clinics, research institutes, and others. Hospitals are the leading end-users due to their comprehensive facilities and the need for accurate diagnostics in patient care. Diagnostic laboratories also play a crucial role, equipped with advanced technologies for high-throughput testing and confirmatory analysis. Home care settings are emerging as a significant segment, driven by the increasing adoption of at-home testing solutions and patient-centric healthcare models. Clinics and research institutes contribute to market growth through routine screening and ongoing research activities .
Global Bacterial Vaginosis Diagnostics Market Competitive Landscape
The Global Bacterial Vaginosis Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Hologic, Inc., BD (Becton, Dickinson and Company), Roche Diagnostics, Abbott Laboratories, Cepheid (Danaher Corporation), Thermo Fisher Scientific Inc., QuidelOrtho Corporation, bioMérieux SA, Siemens Healthineers AG, GenMark Diagnostics, Inc. (Roche), QIAGEN N.V., Sekisui Diagnostics, LabCorp (Laboratory Corporation of America Holdings), Quest Diagnostics Incorporated, DiaSorin S.p.A., Seegene Inc., Sansure Biotech Inc., altona Diagnostics GmbH, R-Biopharm AG contribute to innovation, geographic expansion, and service delivery in this space.
Global Bacterial Vaginosis Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Bacterial Vaginosis:The World Health Organization estimates that bacterial vaginosis affects approximately23% to 29% of women globally, with higher rates in certain regions. In the United States alone, around21 million women aged 14–49 have bacterial vaginosis at any given time. This rising prevalence drives the demand for effective diagnostic solutions, as healthcare providers seek to address the growing number of cases. The increasing incidence is expected to propel the market for bacterial vaginosis diagnostics significantly in the coming years.
- Rising Awareness About Women's Health:According to a 2023 report by the National Women's Health Network, awareness campaigns have led to a 40% increase in women seeking healthcare services related to reproductive health. This heightened awareness is crucial in promoting early diagnosis and treatment of bacterial vaginosis. As educational initiatives continue to expand, more women are likely to recognize symptoms and seek diagnostic testing, further driving market growth in the diagnostics sector.
- Advancements in Diagnostic Technologies:The global investment in healthcare technology is projected to reach$10 trillion in future, with significant advancements in diagnostic tools for bacterial vaginosis. Innovations such as molecular diagnostics and rapid testing kits are enhancing accuracy and speed. For instance, the introduction of PCR-based tests has reduced diagnosis time to under two hours, improving patient outcomes. These technological advancements are expected to stimulate market growth by providing more efficient diagnostic options.
Market Challenges
- High Cost of Advanced Diagnostic Tools:The cost of advanced diagnostic tools for bacterial vaginosis can exceed$1,000, which poses a significant barrier to widespread adoption, especially in low-income regions. Many healthcare facilities struggle to afford these technologies, limiting access to accurate diagnostics. This financial constraint can hinder the overall growth of the market, as healthcare providers may opt for less effective, lower-cost alternatives that do not provide the same level of accuracy.
- Lack of Awareness in Developing Regions:In many developing regions, awareness of bacterial vaginosis and its implications remains low, with only15%of women aware of the condition, according to a 2023 study by the Global Health Organization. This lack of awareness leads to underdiagnosis and undertreatment, stifling market growth. Without educational initiatives and outreach programs, the potential for diagnostic solutions to reach those in need remains limited, impacting overall market expansion.
Global Bacterial Vaginosis Diagnostics Market Future Outlook
The future of the bacterial vaginosis diagnostics market appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, while the shift towards home-based testing solutions will cater to the growing demand for convenience. Additionally, the rise of telemedicine services will facilitate remote consultations, further supporting the market's expansion. These trends indicate a dynamic landscape for bacterial vaginosis diagnostics in the coming years.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets present significant growth opportunities, with a projected increase in healthcare spending reaching$1.5 trillion in future. Companies can capitalize on this trend by introducing affordable diagnostic solutions tailored to local needs, thereby increasing market penetration and improving health outcomes in these regions.
- Development of Rapid Testing Kits:The demand for rapid testing kits is on the rise, with a projected market value of$2 billion in future. Developing innovative, user-friendly kits that provide quick results can enhance patient compliance and satisfaction, creating a lucrative opportunity for manufacturers in the bacterial vaginosis diagnostics market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | PCR-based tests Culture tests Rapid diagnostic tests Microscopy tests Immunoassays Others |
| By End-User | Hospitals Diagnostic laboratories Home care settings Clinics Research institutes Others |
| By Distribution Channel | Direct sales Online sales Distributors Retail pharmacies Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Test Method | Molecular diagnostics Immunoassays Nucleic acid amplification tests (NAATs) Gram stain Others |
| By Sample Type | Vaginal swabs Urine samples Blood samples Cervical samples Others |
| By Pricing Range | Low-cost tests Mid-range tests Premium tests Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers of Diagnostic Devices
Distributors and Retailers of Medical Supplies
Healthcare Providers and Clinics
Pharmaceutical Companies
Industry Associations (e.g., American Society for Microbiology)
Health Insurance Companies
Players Mentioned in the Report:
Hologic, Inc.
BD (Becton, Dickinson and Company)
Roche Diagnostics
Abbott Laboratories
Cepheid (Danaher Corporation)
Thermo Fisher Scientific Inc.
QuidelOrtho Corporation
bioMerieux SA
Siemens Healthineers AG
GenMark Diagnostics, Inc. (Roche)
QIAGEN N.V.
Sekisui Diagnostics
LabCorp (Laboratory Corporation of America Holdings)
Quest Diagnostics Incorporated
DiaSorin S.p.A.
Seegene Inc.
Sansure Biotech Inc.
altona Diagnostics GmbH
R-Biopharm AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Bacterial Vaginosis Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Bacterial Vaginosis Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Bacterial Vaginosis Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of bacterial vaginosis
3.1.2 Rising awareness about women's health
3.1.3 Advancements in diagnostic technologies
3.1.4 Growing demand for point-of-care testing
3.2 Market Challenges
3.2.1 High cost of advanced diagnostic tools
3.2.2 Lack of awareness in developing regions
3.2.3 Regulatory hurdles in product approvals
3.2.4 Limited reimbursement policies
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Development of rapid testing kits
3.3.3 Collaborations with healthcare providers
3.3.4 Increasing investment in R&D
3.4 Market Trends
3.4.1 Shift towards home-based testing solutions
3.4.2 Integration of AI in diagnostics
3.4.3 Focus on personalized medicine
3.4.4 Rise in telemedicine services
3.5 Government Regulation
3.5.1 Stricter guidelines for diagnostic accuracy
3.5.2 Enhanced surveillance of infectious diseases
3.5.3 Support for women's health initiatives
3.5.4 Incentives for innovative diagnostic solutions
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Bacterial Vaginosis Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Bacterial Vaginosis Diagnostics Market Segmentation
8.1 By Type
8.1.1 PCR-based tests
8.1.2 Culture tests
8.1.3 Rapid diagnostic tests
8.1.4 Microscopy tests
8.1.5 Immunoassays
8.1.6 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic laboratories
8.2.3 Home care settings
8.2.4 Clinics
8.2.5 Research institutes
8.2.6 Others
8.3 By Distribution Channel
8.3.1 Direct sales
8.3.2 Online sales
8.3.3 Distributors
8.3.4 Retail pharmacies
8.3.5 Others
8.4 By Region
8.4.1 North America
8.4.2 Europe
8.4.3 Asia-Pacific
8.4.4 Latin America
8.4.5 Middle East & Africa
8.5 By Test Method
8.5.1 Molecular diagnostics
8.5.2 Immunoassays
8.5.3 Nucleic acid amplification tests (NAATs)
8.5.4 Gram stain
8.5.5 Others
8.6 By Sample Type
8.6.1 Vaginal swabs
8.6.2 Urine samples
8.6.3 Blood samples
8.6.4 Cervical samples
8.6.5 Others
8.7 By Pricing Range
8.7.1 Low-cost tests
8.7.2 Mid-range tests
8.7.3 Premium tests
8.7.4 Others
9. Global Bacterial Vaginosis Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Market Share (%)
9.2.3 Revenue (USD million)
9.2.4 Revenue Growth Rate (%)
9.2.5 Product Portfolio Breadth (Number of BV diagnostic products)
9.2.6 Geographic Presence (Number of countries/regions)
9.2.7 R&D Investment (% of revenue)
9.2.8 Regulatory Approvals (Number of BV-related diagnostics approved)
9.2.9 Time-to-Market for New Products (months)
9.2.10 Distribution Network Strength (qualitative/quantitative)
9.2.11 Customer Base (Number of healthcare providers served)
9.2.12 Partnerships & Collaborations (Number of active partnerships)
9.2.13 Pricing Strategy (Average selling price per test)
9.2.14 Brand Recognition Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Hologic, Inc.
9.5.2 BD (Becton, Dickinson and Company)
9.5.3 Roche Diagnostics
9.5.4 Abbott Laboratories
9.5.5 Cepheid (Danaher Corporation)
9.5.6 Thermo Fisher Scientific Inc.
9.5.7 QuidelOrtho Corporation
9.5.8 bioMérieux SA
9.5.9 Siemens Healthineers AG
9.5.10 GenMark Diagnostics, Inc. (Roche)
9.5.11 QIAGEN N.V.
9.5.12 Sekisui Diagnostics
9.5.13 LabCorp (Laboratory Corporation of America Holdings)
9.5.14 Quest Diagnostics Incorporated
9.5.15 DiaSorin S.p.A.
9.5.16 Seegene Inc.
9.5.17 Sansure Biotech Inc.
9.5.18 altona Diagnostics GmbH
9.5.19 R-Biopharm AG
10. Global Bacterial Vaginosis Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Government health departments
10.1.2 Public health organizations
10.1.3 Non-profit organizations
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in diagnostic facilities
10.2.2 Funding for women's health programs
10.2.3 Budget allocation for research and development
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to affordable diagnostics
10.3.2 Need for rapid testing solutions
10.3.3 Challenges in accurate diagnosis
10.4 User Readiness for Adoption
10.4.1 Awareness of bacterial vaginosis
10.4.2 Acceptance of new diagnostic methods
10.4.3 Training and education needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of diagnostic effectiveness
10.5.2 Expansion into new markets
10.5.3 Long-term cost savings analysis
11. Global Bacterial Vaginosis Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue model exploration
1.4 Customer segmentation analysis
1.5 Competitive landscape overview
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience identification
2.4 Communication strategies
2.5 Digital marketing initiatives
3. Distribution Plan
3.1 Urban retail strategies
3.2 Rural NGO tie-ups
3.3 Online distribution channels
3.4 Partnerships with healthcare providers
3.5 Logistics and supply chain management
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands analysis
4.3 Competitor pricing comparison
4.4 Value-based pricing strategies
4.5 Discount and promotion strategies
5. Unmet Demand & Latent Needs
5.1 Category gaps identification
5.2 Consumer segments analysis
5.3 Product development opportunities
5.4 Market entry barriers
5.5 Customer feedback mechanisms
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback and improvement processes
6.5 Community building initiatives
7. Value Proposition
7.1 Sustainability initiatives
7.2 Integrated supply chains
7.3 Cost-effectiveness analysis
7.4 Customer-centric approach
7.5 Innovation in product offerings
8. Key Activities
8.1 Regulatory compliance
8.2 Branding efforts
8.3 Distribution setup
8.4 Training and development
8.5 Market research and analysis
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix considerations
9.1.2 Pricing band analysis
9.1.3 Packaging strategies
9.2 Export Entry Strategy
9.2.1 Target countries selection
9.2.2 Compliance roadmap development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk assessment
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines for market entry
11.3 Financial projections
11.4 Resource allocation
11.5 Milestone tracking
12. Control vs Risk Trade-Off
12.1 Ownership considerations
12.2 Partnerships evaluation
12.3 Risk management strategies
12.4 Control mechanisms
12.5 Exit strategies
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
13.3 Profit margin analysis
13.4 Revenue forecasting
13.5 Cost management strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition targets
14.4 Strategic alliances
14.5 Collaborative research opportunities
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity tracking
15.2.2 Milestone achievements
15.2.3 Performance evaluation
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports and white papers on bacterial vaginosis diagnostics
- Review of academic journals and clinical studies related to diagnostic methods and technologies
- Examination of regulatory guidelines and approvals from health authorities such as the FDA and EMA
Primary Research
- Interviews with gynecologists and healthcare professionals specializing in women's health
- Surveys with laboratory managers and diagnostic test manufacturers
- Focus groups with patients to understand their experiences and preferences regarding diagnostics
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and stakeholder feedback
- Triangulation of data from clinical studies, market reports, and expert opinions
- Sanity checks through peer reviews and consultations with industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on global healthcare expenditure on women's health
- Segmentation of the market by diagnostic type, including molecular tests, culture tests, and rapid tests
- Incorporation of trends in bacterial vaginosis prevalence and awareness campaigns
Bottom-up Modeling
- Collection of sales data from leading diagnostic test manufacturers and distributors
- Estimation of market share based on product offerings and regional presence
- Volume and pricing analysis to derive revenue projections for each diagnostic category
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as healthcare access, population demographics, and disease prevalence
- Scenario modeling based on potential changes in healthcare policies and diagnostic technology advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 100 | Gynecologists, Family Physicians |
| Diagnostic Laboratories | 60 | Laboratory Managers, Technicians |
| Patients with Bacterial Vaginosis | 50 | Women aged 18-45, Recently Diagnosed Patients |
| Pharmaceutical Representatives | 40 | Sales Representatives, Product Managers |
| Health Policy Makers | 40 | Public Health Officials, Regulatory Experts |
Frequently Asked Questions
What is the current value of the Global Bacterial Vaginosis Diagnostics Market?
The Global Bacterial Vaginosis Diagnostics Market is valued at approximately USD 4.8 billion, reflecting the economic burden associated with diagnosing and treating bacterial vaginosis, as well as related complications such as preterm birth and increased susceptibility to HIV.